Scalable Clean Production of 2-Fluoro-4-nitrobenzonitrile for Kinase Inhibitor Manufacturing
The pharmaceutical industry's relentless pursuit of potent tyrosine kinase irreversible inhibitors and protein kinase inhibitors has placed a spotlight on the efficient manufacturing of key building blocks, specifically 2-fluoro-4-nitrobenzonitrile. As detailed in the groundbreaking patent CN103694137A, a novel production method has been developed that fundamentally restructures the traditional synthetic pathway to address critical bottlenecks in environmental sustainability and energy consumption. This technology represents a paradigm shift for manufacturers seeking to optimize their supply chains for high-value oncology and cardiovascular therapeutics. By re-engineering the diazotization-bromination stage, the process not only mitigates the generation of hazardous acidic waste but also drastically lowers the thermal energy requirements traditionally associated with cryogenic reactions. For global procurement leaders and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while adhering to increasingly strict green chemistry mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-4-nitrobenzonitrile has been plagued by significant operational inefficiencies, particularly during the conversion of the aniline precursor to the bromo-intermediate. Traditional protocols necessitate rigorous temperature control, typically maintaining reaction conditions between 0°C and 5°C to prevent the decomposition of the unstable diazonium salt. This requirement imposes a heavy burden on production facilities, demanding extensive refrigeration infrastructure that drives up both capital expenditure and ongoing utility costs. Furthermore, the conventional aqueous acidic media used in these reactions generates an exorbitant volume of waste; for every single ton of 2-fluoro-4-nitrobromobenzene produced, approximately 20 to 30 tons of acidic waste liquid are created. This massive effluent stream presents a formidable challenge for waste treatment facilities, creating environmental compliance risks and escalating the total cost of ownership for the manufacturing process. The combination of high energy intensity for cooling and the logistical nightmare of waste disposal has long rendered this a suboptimal route for large-scale commercial production.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent introduces a robust, high-temperature diazotization-bromination system that operates effectively between 40°C and 80°C. This substantial increase in operating temperature eliminates the dependency on energy-intensive freezing equipment, thereby streamlining the thermal management of the reactor. The process utilizes a biphasic system involving ethylene dichloride and water, which facilitates the immediate extraction of the organic product, stabilizing the reaction mixture and allowing for higher thermal tolerance. Consequently, the volume of acidic waste liquid is significantly curtailed, dropping to a range of 15.5 to 22.7 tons per ton of product, representing a tangible improvement in atom economy and environmental footprint. Moreover, the integration of a "one-pot" strategy merges multiple operational steps, reducing the complexity of the workflow and minimizing the potential for human error or material loss during transfer. This approach not only enhances safety but also aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing by simplifying plant operations.
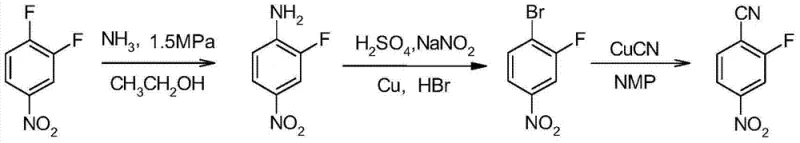
Mechanistic Insights into Improved Diazotization-Bromination
The core innovation lies in the precise manipulation of the reaction medium and catalytic components during the Sandmeyer-type transformation. By employing a specific molar ratio of hydrobromic acid to copper powder (ranging from 5.0:1 to 15.0:1) and introducing sodium nitrite slowly into the heated mixture, the system maintains a steady generation of the diazonium species which is immediately consumed by the bromide source. The presence of ethylene dichloride acts as a crucial phase-transfer agent, extracting the lipophilic 2-fluoro-4-nitrobromobenzene as it forms, which shifts the equilibrium forward and protects the product from hydrolysis or side reactions that typically occur in purely aqueous acidic environments at elevated temperatures. This biphasic dynamic is key to enabling the reaction to proceed safely at 40-80°C, a condition that would be catastrophic in a standard homogeneous aqueous setup. The subsequent cyanation step utilizes N-methyl pyrrolidone (NMP) as a high-boiling polar aprotic solvent, which effectively dissolves cuprous cyanide and facilitates the nucleophilic substitution of the bromine atom with the cyano group at temperatures around 150-160°C.
Impurity control is rigorously managed through the optimization of stoichiometry and workup procedures. The patent specifies that the crude 2-fluoro-4-nitrobromobenzene obtained is a brown crystal with a content exceeding 99.0% (GC), indicating minimal formation of phenolic byproducts or diaryl ethers which are common impurities in nucleophilic aromatic substitutions. The final recrystallization from toluene ensures that the finished 2-fluoro-4-nitrobenzonitrile achieves a purity of greater than 99.0% (HPLC), appearing as distinct yellow crystals. The ability to recover solvents like ethylene dichloride and toluene via vacuum distillation further purifies the system for subsequent batches, creating a closed-loop efficiency that prevents the accumulation of trace contaminants over time. This level of purity is non-negotiable for high-purity pharmaceutical intermediates destined for clinical applications, where even trace genotoxic impurities can halt drug development programs.
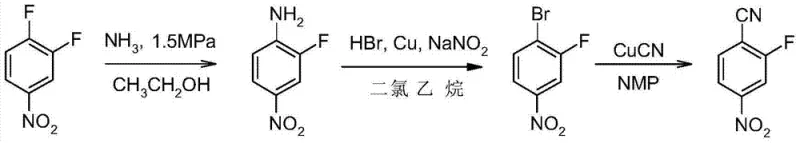
How to Synthesize 2-Fluoro-4-nitrobenzonitrile Efficiently
The execution of this synthesis requires careful attention to the sequential addition of reagents and temperature profiling to maximize yield and safety. The process begins with the preparation of the amine precursor, followed by the critical high-temperature bromination, and concludes with the cyanation. Operators must ensure strict adherence to the molar ratios of copper catalysts and halide sources to maintain the catalytic cycle. The following guide outlines the standardized operational parameters derived from the patent examples, serving as a baseline for process engineers aiming to implement this technology. For the complete, step-by-step laboratory protocol including exact addition rates and quenching procedures, please refer to the detailed instructions below.
- Prepare 2-fluoro-4-nitroaniline by reacting 3,4-difluoronitrobenzene with ammonia solution and copper oxide catalyst under pressure.
- Perform the improved diazotization-bromination in a biphasic system of ethylene dichloride and water at 40-80°C using HBr, Cu powder, and NaNO2.
- Execute the cyanation reaction using cuprous cyanide in NMP solvent at elevated temperatures to yield the final nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain executives and procurement managers, the adoption of this improved synthesis route offers compelling strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the drastic simplification of the utility infrastructure required for production. By removing the necessity for cryogenic cooling systems, facilities can repurpose expensive refrigeration capacity or avoid installing it altogether, leading to substantial capital savings and reduced maintenance overheads. Furthermore, the reduction in waste volume directly translates to lower disposal fees and reduced regulatory scrutiny, ensuring a more stable and uninterrupted supply continuity. The ability to recover valuable byproducts like sodium bromide through bipolar membrane electrodialysis adds an additional revenue stream or cost offset, enhancing the overall economic viability of the process. These factors combined create a resilient supply chain capable of withstanding market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The elimination of low-temperature constraints significantly lowers energy consumption, as the process no longer relies on power-hungry chillers to maintain 0-5°C conditions. Additionally, the recovery and recycling of solvents such as ethylene dichloride and toluene minimize raw material procurement costs, while the "one-pot" nature of the bromination step reduces labor hours and equipment occupancy time. The qualitative reduction in waste treatment complexity further decreases the operational expenditure associated with environmental compliance and effluent management.
- Enhanced Supply Chain Reliability: The robustness of the high-temperature reaction conditions makes the process less susceptible to minor fluctuations in cooling water availability or ambient temperature variations, ensuring consistent batch-to-batch performance. The use of readily available industrial-grade reagents and the ability to recycle key components mitigate the risk of raw material shortages. This stability is crucial for maintaining long-term contracts with downstream API manufacturers who require guaranteed delivery schedules for their own production planning.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as the biphasic system manages heat dissipation effectively even in large reactors. The significant reduction in acidic waste generation aligns with modern green chemistry principles, facilitating easier permitting and community acceptance of manufacturing sites. The integration of membrane technology for salt recovery demonstrates a commitment to circular economy practices, future-proofing the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-fluoro-4-nitrobenzonitrile using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding process adoption and vendor qualification.
Q: How does the new diazotization process reduce environmental impact?
A: The improved process reduces acidic waste liquid generation from 20-30 tons per ton of product down to 15.5-22.7 tons and allows for sodium bromide recovery via bipolar membrane electrodialysis.
Q: What are the energy advantages of this synthesis route?
A: By raising the reaction temperature from the conventional 0-5°C to 40-80°C, the process eliminates the need for energy-intensive refrigeration equipment during the critical bromination stage.
Q: What purity levels can be achieved with this method?
A: The method consistently yields 2-fluoro-4-nitrobenzonitrile with a content greater than 99.0% (HPLC) after recrystallization, meeting stringent pharmaceutical intermediate standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-4-nitrobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of the CN103694137A process can be fully realized at your required volume. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-fluoro-4-nitrobenzonitrile meets the exacting standards required for kinase inhibitor synthesis. Our commitment to quality assurance means that you receive a product that is not only chemically pure but also free from the process-related impurities that could jeopardize your downstream reactions.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies specific to your facility's constraints. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and reliability of our manufacturing processes before making any commitments. Let us be your strategic partner in delivering high-performance intermediates for the next generation of life-saving medicines.
