Advanced Low-Pressure Synthesis of 2-Chloro-3-Trifluoromethylpyridine for Commercial Scale-Up
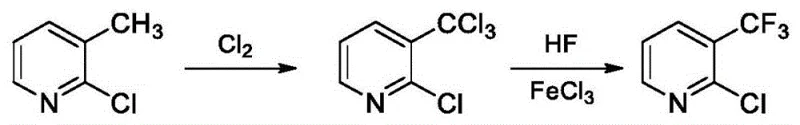
The global demand for fluorinated heterocycles continues to surge, driven by their critical role in next-generation pharmaceuticals and agrochemicals. A pivotal advancement in this sector is detailed in patent CN114292227A, which discloses a highly efficient method for preparing 2-chloro-3-trifluoromethylpyridine. This compound serves as a vital building block for active ingredients such as the herbicide flazasulfuron and the anti-prostate cancer drug apalutamide. The disclosed technology addresses long-standing industrial bottlenecks by utilizing a unique solvent system and a telescoped reaction sequence that eliminates intermediate isolation. By employing 3,4-dichlorotrifluorotoluene as a reaction medium, the process achieves exceptional selectivity during the chlorination phase, while the subsequent fluorination step operates under remarkably mild pressure conditions compared to legacy methods. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted pyridines has been plagued by severe operational hazards and inefficiencies. Traditional routes often rely on starting materials that are either prohibitively expensive or require multi-step derivatization to achieve the desired substitution pattern. For instance, methods utilizing 3-methylpyridine often suffer from poor regioselectivity, yielding significant amounts of the unwanted 2-chloro-5-trifluoromethyl isomer, which complicates purification and drives up waste disposal costs. Furthermore, conventional fluorination protocols typically necessitate extreme reaction conditions, with pressures soaring between 3.0 and 9.0 MPa and temperatures exceeding 160°C. These harsh parameters not only accelerate equipment degradation but also impose stringent safety requirements that limit batch sizes and increase capital expenditure. Additionally, the use of standard chlorinated solvents like chlorobenzene often leads to competitive side reactions where the solvent itself becomes chlorinated, generating difficult-to-remove impurities that compromise the final product quality.
The Novel Approach
The methodology outlined in CN114292227A offers a transformative solution by re-engineering both the solvent environment and the reaction workflow. The core innovation lies in the selection of 3,4-dichlorotrifluorotoluene as the solvent, which demonstrates superior chemical inertness during the radical chlorination of 2-chloro-3-methylpyridine. Unlike traditional solvents, it resists further chlorination, ensuring that the reaction energy is focused exclusively on converting the methyl group to a trichloromethyl group. Moreover, the process introduces a telescoped strategy where the crude chlorination mixture is directly subjected to fluorination without any concentration or purification steps. This "one-pot" philosophy drastically reduces processing time and solvent consumption. The fluorination stage is equally revolutionary, utilizing a controlled two-stage temperature gradient and maintaining system pressure at a safe 1.0-1.5 MPa. This gentle approach not only preserves the integrity of the pyridine ring but also facilitates a straightforward crystallization workup, delivering the target molecule with purity levels reaching 99.4%.
Mechanistic Insights into FeCl3-Catalyzed Halogen Exchange
The chemical elegance of this process is rooted in the precise control of radical and ionic mechanisms across the two distinct stages. In the initial chlorination phase, the reaction proceeds via a free-radical mechanism where chlorine gas attacks the benzylic methyl position of the 2-chloro-3-methylpyridine. The absence of a catalyst in this step is intentional; it prevents premature electrophilic aromatic substitution on the pyridine ring, which would lead to polychlorinated byproducts. The use of a dual temperature gradient—starting at 105-120°C and ramping to 130-145°C—ensures complete conversion to the trichloromethyl intermediate while managing the exothermicity of the chlorine addition. Following this, the introduction of anhydrous hydrogen fluoride in the presence of ferric chloride (FeCl3) shifts the mechanism to a Lewis acid-catalyzed halogen exchange (Halex). The FeCl3 activates the carbon-chlorine bonds of the trichloromethyl group, making them susceptible to nucleophilic attack by fluoride ions. This catalytic cycle is highly efficient, requiring only a molar ratio of 0.01-0.03 relative to the substrate, which minimizes metal contamination in the final product.
Impurity control is rigorously maintained through the specific choice of reaction media and stoichiometry. The patent data highlights that deviating from the optimal molar ratios of chlorine or hydrogen fluoride leads to incomplete conversion or the formation of partially fluorinated species. Crucially, the solvent 3,4-dichlorotrifluorotoluene acts as a thermodynamic sink that stabilizes the transition states without participating in the reaction. Comparative data indicates that using chlorobenzene results in detectable levels of polychlorinated solvent derivatives, which co-elute with the product and require energy-intensive distillation to remove. By contrast, the inert solvent system allows the final product to be isolated simply by cooling the reaction mass to -10°C to -5°C. This crystallization-driven purification leverages the solubility differences between the highly polar trifluoromethyl product and the non-polar solvent, effectively excluding organic impurities and yielding a solid product of exceptional quality suitable for direct use in sensitive pharmaceutical syntheses.
How to Synthesize 2-Chloro-3-Trifluoromethylpyridine Efficiently
The synthesis protocol described in the patent provides a robust framework for manufacturing this critical intermediate at scale. The procedure begins with the dissolution of the starting material in the specialized solvent, followed by a carefully monitored chlorination sequence that avoids the need for light or radical initiators. Once the trichloromethyl intermediate is formed, the process transitions seamlessly into the fluorination phase by introducing the catalyst and hydrogen fluoride. The detailed operational parameters, including specific temperature ramps and pressure controls, are designed to maximize yield while ensuring operator safety. For technical teams looking to implement this route, the following standardized guide outlines the critical process steps derived from the patent examples.
- Dissolve 2-chloro-3-methylpyridine in 3,4-dichlorotrifluorotoluene and perform two-stage radical chlorination at 105-145°C without a catalyst.
- Directly add ferric chloride catalyst to the crude chlorination mixture and initiate fluorination with hydrogen fluoride.
- Execute a two-stage temperature gradient fluorination (105-155°C) at low pressure (1.0-1.5 MPa), followed by cooling crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic advantages beyond mere technical performance. The elimination of intermediate isolation steps fundamentally alters the cost structure of production by reducing labor hours, energy consumption for solvent recovery, and equipment occupancy time. Furthermore, the ability to operate fluorination reactors at significantly lower pressures reduces the specification requirements for reactor vessels, allowing for the use of more cost-effective equipment and lowering maintenance overheads. These efficiencies translate directly into a more resilient supply chain capable of meeting fluctuating market demands without the bottlenecks associated with complex multi-step purifications.
- Cost Reduction in Manufacturing: The telescoped nature of this process eliminates the need to concentrate and purify the 2-chloro-3-trichloromethylpyridine intermediate, which traditionally accounts for a significant portion of processing costs. By avoiding solvent swapping and distillation between steps, the facility saves substantial amounts of energy and reduces the loss of valuable material during transfer operations. Additionally, the reduced consumption of hydrogen fluoride—achieved through optimized stoichiometry rather than massive excess—lowers the cost of raw materials and minimizes the expense associated with neutralizing and disposing of hazardous fluoride waste streams.
- Enhanced Supply Chain Reliability: The starting material, 2-chloro-3-methylpyridine, is a commodity chemical with a stable global supply, reducing the risk of raw material shortages that often plague exotic fluorinated precursors. The robustness of the reaction conditions, particularly the tolerance for slight variations in temperature and pressure within the specified gradients, ensures consistent batch-to-batch quality. This reliability is crucial for long-term contracts with pharmaceutical clients who require strict adherence to quality specifications and uninterrupted delivery schedules for their own API production lines.
- Scalability and Environmental Compliance: Operating at pressures below 1.5 MPa significantly eases the regulatory burden and safety protocols required for scale-up, facilitating faster technology transfer from pilot plant to commercial production. The process generates fewer chlorinated organic byproducts due to the inert solvent system, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly a deciding factor for multinational partners selecting suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and product quality. Understanding these nuances is essential for engineering teams evaluating the feasibility of adopting this route for large-scale manufacturing.
Q: Why is 3,4-dichlorotrifluorotoluene preferred over chlorobenzene solvents in this synthesis?
A: Unlike chlorobenzene or dichlorobenzene, 3,4-dichlorotrifluorotoluene is chemically inert under radical chlorination conditions. This prevents the solvent itself from undergoing side reactions to form polychlorinated impurities, thereby significantly enhancing the purity of the 2-chloro-3-trichloromethylpyridine intermediate and simplifying downstream purification.
Q: How does this patent improve safety compared to traditional fluorination methods?
A: Traditional methods often require extreme pressures ranging from 3.0 to 9.0 MPa and temperatures exceeding 160°C. This novel process operates at a significantly lower pressure of 1.0-1.5 MPa and moderate temperatures below 155°C, drastically reducing the mechanical stress on reactor vessels and minimizing the risk of thermal runaway incidents.
Q: Is intermediate isolation required between the chlorination and fluorination steps?
A: No. A key efficiency driver of this technology is the telescoped nature of the reaction. The crude chlorination liquid is used directly for the subsequent fluorination step after simply adding the catalyst, eliminating the need for concentration, solvent swapping, or purification of the trichloromethyl intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of low-pressure fluorination are fully realized in practice. Our facilities are equipped with advanced corrosion-resistant reactors capable of handling anhydrous hydrogen fluoride safely, and our stringent purity specifications are enforced by rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to secure a stable supply of this high-value intermediate for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your project timelines with reliable, high-quality chemical solutions.
