Advanced Synthesis of 2-Chloro-3-Trifluoromethylpyridine via Byproduct Valorization for Commercial Scale-Up
The chemical manufacturing landscape is increasingly driven by the dual imperatives of economic efficiency and environmental sustainability, a challenge directly addressed by the innovative methodology disclosed in patent CN112441966A. This pivotal intellectual property outlines a sophisticated multi-step synthesis for producing 2-chloro-3-trifluoromethylpyridine, a critical intermediate widely utilized in the agrochemical sector for herbicides such as flazasulfuron. Traditionally, the production of 2,3,6-trichloro-5-trifluoromethylpyridine results in significant quantities of this compound as an over-chlorinated byproduct with limited commercial utility, often destined for waste streams. The patented process ingeniously reverses this narrative by employing a directional conversion strategy that transforms this specific waste stream into a high-value commodity through a sequence of selective reduction, etherification protection, catalytic hydrogenation, hydrolysis, and chlorination. For R&D directors and procurement specialists alike, this represents a paradigm shift from linear consumption to circular resource utilization, offering a robust pathway to secure supply chains while mitigating the environmental footprint associated with fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 2-chloro-3-trifluoromethylpyridine have historically been plagued by significant economic and technical bottlenecks that hinder efficient commercial scale-up. Existing literature, such as CN108586334, describes routes starting from 3-trifluoromethyl-pyridine-2-carboxylic acid, which suffer from relatively low yields around 65% and generate substantial solid waste, thereby inflating disposal costs and reducing overall process mass intensity. Other approaches, including those cited in CN101062915 and CN102875454, rely on the fluorination of methylpyridine derivatives under extremely harsh conditions, often requiring temperatures exceeding 250°C and the use of hazardous reagents like hydrofluoric acid or cobalt chloride catalysts. These high-energy processes not only demand specialized, corrosion-resistant equipment that drives up capital expenditure but also frequently result in poor selectivity, yielding mixtures of 2-chloro-3-trifluoromethylpyridine and its 5-isomer that are difficult and costly to separate. Furthermore, the reliance on expensive starting materials like 2-chloro-3-methylpyridine creates a volatile cost structure that is highly sensitive to upstream petrochemical fluctuations, making long-term supply planning precarious for downstream herbicide manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the process defined in CN112441966A introduces a groundbreaking strategy that leverages the abundance of 2,3,6-trichloro-5-trifluoromethylpyridine, turning a liability into a strategic asset. By initiating the synthesis with this readily available byproduct, the novel approach effectively bypasses the need for expensive fluorination steps and high-temperature gas-phase reactions, operating instead under much milder liquid-phase conditions. The core innovation lies in the precise control of chemoselectivity during the initial reduction phase, where specific chlorine atoms are removed while preserving the trifluoromethyl group and the pyridine ring integrity. This is followed by a protective etherification step that facilitates subsequent hydrogenation, ensuring that the final chlorination yields the desired 2-chloro isomer with exceptional purity levels reaching approximately 99%. This route not only simplifies the purification train by utilizing techniques like steam distillation but also aligns perfectly with green chemistry principles by maximizing atom economy and minimizing the generation of hazardous waste, thereby offering a compelling value proposition for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Selective Reduction and Catalytic Hydrogenation
The chemical elegance of this synthesis is rooted in its five distinct transformation stages, beginning with the critical selective reductive dechlorination of the trichloro starting material. This initial step can be executed via a direct method using inexpensive metals like zinc powder in the presence of acetic acid, or an indirect method involving hydrazine and subsequent oxidation with sodium hypochlorite. In the direct pathway, the metal-acid system generates nascent hydrogen in situ, which selectively targets the chlorine atom at the 2-position due to electronic and steric factors inherent to the pyridine ring, yielding 2,5-dichloro-3-trifluoromethylpyridine with high selectivity around 95%. Following this, the intermediate undergoes nucleophilic aromatic substitution with sodium alkoxide in a lower aliphatic alcohol solvent, installing an alkoxy protecting group at the 2-position. This etherification is crucial as it activates the ring for the subsequent catalytic hydrogenation step, where a palladium-on-carbon catalyst facilitates the removal of the remaining chlorine atom at the 5-position under mild hydrogen pressure, typically between 0.1 MPa and 2.0 MPa.
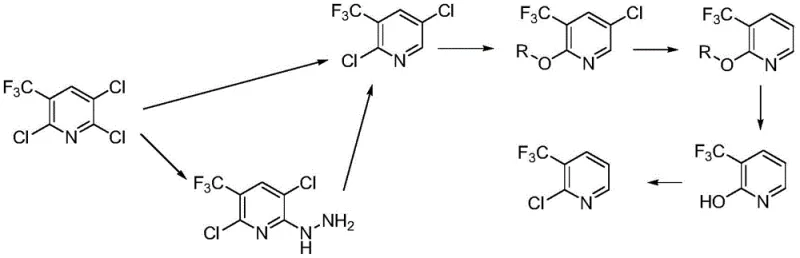
The final stages of the mechanism involve the strategic removal of the protecting group and the reintroduction of the chlorine atom at the desired position. The 2-alkoxy-3-trifluoromethylpyridine intermediate is subjected to azeotropic reflux hydrolysis using concentrated hydrochloric acid, which cleaves the ether bond to reveal the 2-hydroxy-3-trifluoromethylpyridine phenol derivative. This hydrolysis step is meticulously controlled at temperatures around 85-90°C to prevent degradation of the sensitive trifluoromethyl group while ensuring complete conversion. The synthesis concludes with a chlorination reaction using phosphorus oxychloride, a standard yet highly effective reagent for converting pyridones to chloropyridines. This final transformation restores the chlorine atom at the 2-position, completing the cycle and delivering the target 2-chloro-3-trifluoromethylpyridine. The entire sequence demonstrates remarkable impurity control, as evidenced by the ability to recover unreacted starting materials via steam distillation and the high purity of the final distillate, ensuring that the product meets the stringent specifications required for pharmaceutical and agrochemical applications.
How to Synthesize 2-Chloro-3-Trifluoromethylpyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the selective reduction and hydrogenation phases where catalyst activity and stoichiometry play pivotal roles. The process is designed to be operationally robust, utilizing common solvents like methanol and ethanol which can be easily recovered and recycled, further enhancing the economic viability of the method. Operators must monitor the conversion rates closely, especially in the first step, maintaining raw material conversion between 33% and 40% in certain direct reduction modes to maximize selectivity and minimize byproduct formation. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures for each of the five stages, are outlined in the comprehensive guide below to ensure reproducibility and safety during scale-up.
- Perform selective reductive dechlorination of 2,3,6-trichloro-5-trifluoromethylpyridine using zinc/acetic acid or hydrazine to obtain 2,5-dichloro-3-trifluoromethylpyridine.
- React the dichloro intermediate with sodium alkoxide in lower aliphatic alcohol to form the protected 2-alkoxy-5-chloro-3-trifluoromethylpyridine ether.
- Execute catalytic hydrogenation to remove the remaining chlorine atom, followed by acid hydrolysis and final chlorination with phosphorus oxychloride to yield the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers transformative benefits that extend far beyond simple chemical synthesis, fundamentally altering the cost structure and reliability of the supply base. By shifting the raw material basis from expensive, dedicated precursors to a valorized byproduct stream, manufacturers can achieve substantial cost savings that are resilient to market volatility. The elimination of high-temperature gas-phase fluorination steps significantly reduces energy consumption and the need for specialized high-alloy reactors, leading to lower capital and operational expenditures. Furthermore, the use of standard unit operations such as filtration, distillation, and slurry-bed hydrogenation ensures that the process can be rapidly scaled from pilot plants to multi-ton production facilities without requiring exotic infrastructure. This scalability translates directly into enhanced supply chain reliability, as production can be ramped up quickly to meet surging demand for herbicide intermediates without the long lead times associated with constructing new specialized plants.
- Cost Reduction in Manufacturing: The economic model of this process is predicated on the utilization of 2,3,6-trichloro-5-trifluoromethylpyridine, a material that is often available at a fraction of the cost of traditional starting materials like 3-trifluoromethyl-pyridine-2-carboxylic acid. By converting a low-value waste product into a high-margin intermediate, the overall cost of goods sold is drastically reduced, allowing for more competitive pricing in the global agrochemical market. Additionally, the recovery and reuse of solvents and unreacted starting materials through steam distillation further compresses manufacturing costs, ensuring that the process remains economically attractive even when raw material prices fluctuate. The avoidance of precious metal catalysts in the initial reduction step, favoring cheap metals like zinc or iron, also contributes to a leaner cost structure compared to routes relying on continuous noble metal catalysis.
- Enhanced Supply Chain Reliability: Diversifying the source of 2-chloro-3-trifluoromethylpyridine away from a single synthetic route mitigates the risk of supply disruptions caused by feedstock shortages or regulatory changes affecting specific precursors. Since the starting material is a byproduct of another major industrial process, its availability is generally stable and decoupled from the specific demand cycles of the herbicide market. This decoupling provides a buffer against supply shocks, ensuring consistent delivery schedules for downstream customers. Moreover, the robustness of the chemical steps, which tolerate minor variations in reaction conditions without significant yield loss, means that production uptime is maximized, and batch-to-batch consistency is maintained, fostering trust and long-term partnerships with key accounts.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing equipment and conditions that are standard in modern fine chemical facilities. The moderate reaction temperatures and pressures reduce safety risks and simplify engineering controls, facilitating faster regulatory approvals and permitting. From an environmental perspective, the conversion of a hazardous chlorinated byproduct into a useful product aligns with increasingly strict global regulations on waste disposal and carbon emissions. The reduction in solid waste generation and the minimization of hazardous reagent usage simplify wastewater treatment and effluent management, lowering the environmental compliance burden and enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on yield expectations, purity profiles, and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of the process described in CN112441966A?
A: The primary advantage is the valorization of 2,3,6-trichloro-5-trifluoromethylpyridine, a low-value byproduct from other pyridine productions, converting it into the high-value intermediate 2-chloro-3-trifluoromethylpyridine with high purity (approx. 99%) and improved economic efficiency.
Q: How does this method compare to traditional synthesis routes regarding raw materials?
A: Traditional routes often rely on expensive precursors like 3-trifluoromethyl-pyridine-2-carboxylic acid or require harsh fluorination conditions on methylpyridines. This novel method utilizes an abundant chlorinated byproduct, significantly reducing raw material costs and avoiding complex fluorination steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard unit operations such as steam distillation, filtration, and catalytic hydrogenation in slurry bed reactors. The reaction conditions are moderate (e.g., 20-65°C for reduction, atmospheric to low pressure for hydrogenation), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global agrochemical supply chain and are uniquely positioned to leverage technologies like CN112441966A for our clients. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can seamlessly transition this innovative waste-valorization route from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-chloro-3-trifluoromethylpyridine meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking reliable sources of critical building blocks for herbicide production.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this byproduct-derived route. We encourage you to contact us today to索取 specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring that your production needs are met with the utmost efficiency and reliability.
