Advanced Chiral Bisphosphonamide Compounds for Scalable Asymmetric Catalysis and Pharmaceutical Manufacturing
Advanced Chiral Bisphosphonamide Compounds for Scalable Asymmetric Catalysis and Pharmaceutical Manufacturing
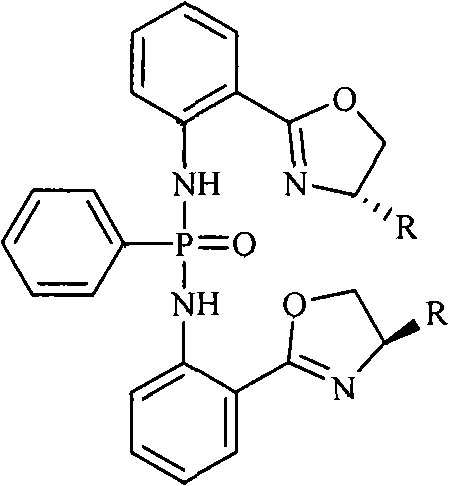
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher enantiomeric purity and more sustainable manufacturing processes in the pharmaceutical sector. Patent CN101885742A introduces a significant advancement in this field through the development of novel chiral bisphosphonamide compounds. These molecules, specifically bis-N-2-[(4S)-4,5-dihydro-4-R-2-oxazolinyl]diphenyl-phenylphosphonamides, represent a robust class of chiral ligands designed to enhance the efficiency of metal-catalyzed transformations. By leveraging a streamlined synthetic pathway that utilizes zinc chloride as a key catalyst, this technology addresses critical bottlenecks associated with traditional ligand synthesis, such as complex purification and low overall yields. For R&D directors and procurement specialists seeking a reliable chiral catalyst supplier, understanding the structural nuances and synthetic accessibility of these compounds is paramount for integrating them into next-generation drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phosphine-oxazoline ligands has been plagued by several inherent inefficiencies that hinder their widespread adoption in large-scale manufacturing. Traditional routes often rely on precious metal catalysts or require stringent cryogenic conditions to maintain stereochemical integrity, which drastically increases operational expenditures and energy consumption. Furthermore, many conventional methods involve multi-step sequences with intermediate isolation procedures that lead to substantial material loss, often resulting in overall yields that are commercially unviable for bulk production. The reliance on sensitive reagents also complicates supply chain logistics, as specialized storage and handling protocols are necessary to prevent degradation. These factors collectively contribute to extended lead times and elevated costs, creating significant barriers for pharmaceutical companies aiming to optimize their cost reduction in asymmetric synthesis manufacturing strategies.
The Novel Approach
In contrast, the methodology outlined in CN101885742A offers a transformative solution by employing a zinc chloride-catalyzed cyclization strategy that operates under anhydrous and oxygen-free reflux conditions. This approach eliminates the need for expensive noble metals during the ligand construction phase, thereby simplifying the impurity profile and reducing the burden on downstream purification systems. The process is characterized by its robustness, utilizing common industrial solvents such as chlorobenzene and toluene, which are readily available and cost-effective. By consolidating the synthesis into a concise two-step sequence, the novel approach minimizes unit operations and enhances the overall throughput of the manufacturing line. This efficiency not only accelerates the timeline from laboratory bench to pilot plant but also ensures a more consistent supply of high-purity materials, addressing the critical needs of supply chain heads focused on continuity and reliability.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization and Phosphonylation
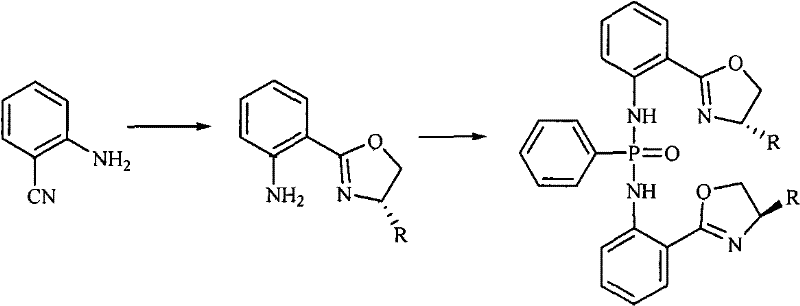
The core innovation of this technology lies in the precise mechanistic control exerted during the formation of the chiral oxazoline ring, which serves as the stereodetermining element of the final catalyst. The reaction between 2-cyanoaniline and various L-amino alcohols, facilitated by ZnCl2, proceeds through a coordinated activation of the nitrile group, promoting nucleophilic attack by the hydroxyl moiety of the amino alcohol. This cyclization occurs with high regioselectivity and retention of configuration at the chiral center, ensuring that the stereochemical information from the starting amino alcohol is faithfully transferred to the heterocyclic scaffold. The subsequent phosphonylation step involves the reaction of the resulting amine intermediate with phenyl phosphonyl dichloride in the presence of triethylamine, which acts as an acid scavenger to drive the equilibrium towards the desired bisphosphonamide product. This dual-step mechanism is meticulously optimized to prevent racemization, a common pitfall in chiral synthesis that can compromise the efficacy of the final catalytic application.
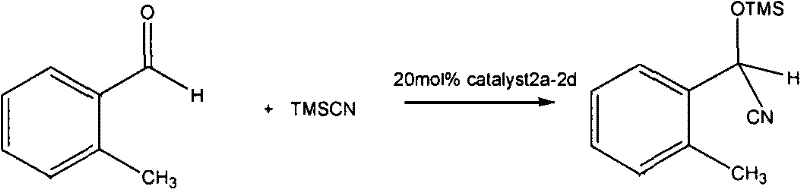
Furthermore, the structural versatility of these compounds allows for fine-tuning of steric and electronic properties by varying the R group on the oxazoline ring, which can be derived from different amino alcohols such as L-leucinol or L-phenylalaninol. This modularity enables chemists to tailor the catalyst for specific transformations, such as the nitrile silylation reaction demonstrated in the patent data, where exceptional enantioselectivity is observed. The ability to achieve up to 99% ee in model reactions underscores the potency of these ligands in inducing asymmetry, making them invaluable tools for the synthesis of complex chiral intermediates. For technical teams evaluating new methodologies, the combination of mechanistic clarity and practical performance provides a compelling case for adopting this chemistry in their process development workflows.
How to Synthesize Chiral Bisphosphonamide Efficiently
The practical implementation of this synthetic route is designed to be accessible for both laboratory research and industrial scale-up, requiring standard equipment and widely available reagents. The process begins with the preparation of the chiral oxazoline intermediate, followed by its conversion into the final phosphonamide derivative through a controlled condensation reaction. Detailed operational parameters, including solvent ratios, temperature profiles, and workup procedures, are critical to maximizing yield and purity. While the general framework is robust, specific attention must be paid to maintaining anhydrous conditions throughout the sequence to prevent hydrolysis of the reactive phosphonyl chloride species. The following guide outlines the standardized protocol derived from the patent examples, serving as a foundational reference for process chemists aiming to replicate or adapt this chemistry for their specific production needs.
- React 2-cyanoaniline with L-amino alcohol using ZnCl2 catalyst in chlorobenzene under reflux for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via chloroform extraction and column chromatography to ensure high optical purity.
- Condense the purified intermediate with phenyl phosphonyl dichloride in toluene and triethylamine under reflux to yield the target bisphosphonamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral bisphosphonamide synthesis route offers substantial strategic benefits for organizations focused on optimizing their supply chain and reducing manufacturing overheads. The elimination of precious metal catalysts in the ligand synthesis phase directly translates to lower raw material costs and reduced dependency on volatile commodity markets associated with metals like palladium or rhodium. Additionally, the use of common organic solvents simplifies waste management and solvent recovery processes, contributing to a more sustainable and environmentally compliant operation. These factors collectively enhance the economic viability of producing high-value chiral intermediates, allowing companies to maintain competitive pricing while adhering to strict quality standards. For procurement managers, this translates into a more predictable cost structure and reduced risk of supply disruptions caused by specialized reagent shortages.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis significantly reduces the number of unit operations required compared to traditional multi-step ligand preparations. By avoiding the use of expensive transition metal catalysts and minimizing the need for complex chromatographic purifications through efficient crystallization or extraction techniques, the overall production cost is drastically lowered. This efficiency allows for better margin management and provides flexibility in pricing strategies for downstream pharmaceutical clients seeking cost-effective solutions for their API synthesis.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 2-cyanoaniline and standard L-amino alcohols ensures a stable and resilient supply chain. Unlike proprietary reagents that may be sourced from single suppliers, these bulk chemicals are produced by multiple vendors globally, mitigating the risk of bottlenecks. Furthermore, the robustness of the reaction conditions means that production can be maintained consistently without frequent adjustments, ensuring timely delivery of materials to meet tight project deadlines and clinical trial schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard reflux setups that can be easily translated to larger reactor volumes. The absence of highly toxic reagents and the ability to recycle solvents like toluene and chlorobenzene align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations facilitates smoother regulatory approvals and enhances the corporate sustainability profile, which is increasingly important for stakeholders and investors in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these chiral bisphosphonamide compounds. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the technology's capabilities. Whether you are concerned about reaction scope, purification methods, or scalability, the answers below offer a comprehensive overview to assist in your decision-making process. We encourage technical teams to review these details carefully to assess the fit for their specific synthetic challenges.
Q: What are the primary advantages of this ZnCl2-catalyzed route?
A: The use of ZnCl2 allows for efficient cyclization under relatively mild reflux conditions, avoiding expensive transition metals and simplifying downstream purification processes significantly.
Q: What is the enantioselectivity performance of these catalysts?
A: In nitrile silylation reactions, these compounds demonstrate excellent enantioselectivity, achieving up to 99% ee, making them highly suitable for chiral drug synthesis.
Q: Is this process scalable for industrial production?
A: Yes, the two-step protocol utilizes common solvents like chlorobenzene and toluene and avoids ultra-low temperature requirements, facilitating straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisphosphonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral catalysts play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our infrastructure is designed to handle complex chemistries safely and consistently, providing our partners with the confidence they need to advance their projects without supply chain interruptions.
We invite you to collaborate with us to explore how this advanced chiral bisphosphonamide technology can enhance your synthetic processes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your operations. Let us be your partner in achieving excellence in asymmetric synthesis and pharmaceutical manufacturing.
