Advanced Synthesis of Chiral Bisphosphonamide Ligands for Commercial Asymmetric Catalysis
Advanced Synthesis of Chiral Bisphosphonamide Ligands for Commercial Asymmetric Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for high-purity chiral intermediates in the pharmaceutical sector. Patent CN101607970A introduces a robust methodology for preparing chiral bisphosphonamide compounds, specifically bis N-2-[(4S)-4,5-dihydro-4-R-2-oxazolinyl]diphenyl-phenylphosphonamide derivatives. These compounds serve as exceptional chiral ligands, demonstrating superior catalytic activity and high enantioselectivity in the asymmetric synthesis of mandelic acid and related structures. The structural versatility of these ligands, where R can be varied from isobutyl to phenyl groups, allows for precise tuning of the catalytic pocket to match specific substrate requirements. 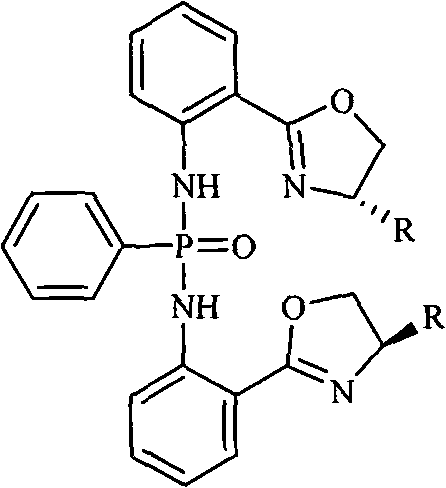 For R&D directors and process chemists, the ability to access such well-defined chiral environments is critical for developing efficient drug synthesis pathways. The patent outlines a clear, reproducible route that avoids the pitfalls of earlier generations of ligands, which often suffered from poor stability or difficult purification profiles. By leveraging a zinc-catalyzed cyclization followed by a controlled phosphonylation step, this technology offers a reliable chiral ligand supplier pathway that balances performance with manufacturability. The detailed characterization data provided, including NMR and HRMS, confirms the structural integrity and purity achievable through this method, setting a high benchmark for quality in fine chemical intermediates.
For R&D directors and process chemists, the ability to access such well-defined chiral environments is critical for developing efficient drug synthesis pathways. The patent outlines a clear, reproducible route that avoids the pitfalls of earlier generations of ligands, which often suffered from poor stability or difficult purification profiles. By leveraging a zinc-catalyzed cyclization followed by a controlled phosphonylation step, this technology offers a reliable chiral ligand supplier pathway that balances performance with manufacturability. The detailed characterization data provided, including NMR and HRMS, confirms the structural integrity and purity achievable through this method, setting a high benchmark for quality in fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phosphorus-nitrogen ligands has been fraught with challenges related to reaction conditions and purification complexity. Traditional methods often rely on harsh acidic conditions or expensive transition metal catalysts to facilitate the formation of the oxazoline ring, which can lead to racemization of the chiral center and a broad impurity profile. Furthermore, the subsequent introduction of the phosphonyl group frequently requires sensitive reagents that demand strictly inert atmospheres and cryogenic temperatures, complicating the scale-up process. These factors contribute to significant cost reduction barriers in pharmaceutical intermediate manufacturing, as the yield losses and extensive purification steps drive up the final price of the ligand. Additionally, the use of stoichiometric amounts of activating agents in older protocols generates substantial chemical waste, posing environmental compliance issues for modern facilities. The lack of modularity in conventional routes also limits the ability to rapidly screen different steric environments, slowing down the optimization of asymmetric catalytic processes.
The Novel Approach
The methodology described in CN101607970A represents a significant technological leap by utilizing a zinc-catalyzed condensation between 2-cyanoaniline and L-amino alcohols. This approach operates under reflux conditions in chlorobenzene, providing sufficient thermal energy to drive the cyclization without compromising the stereochemical integrity of the L-amino alcohol starting material. 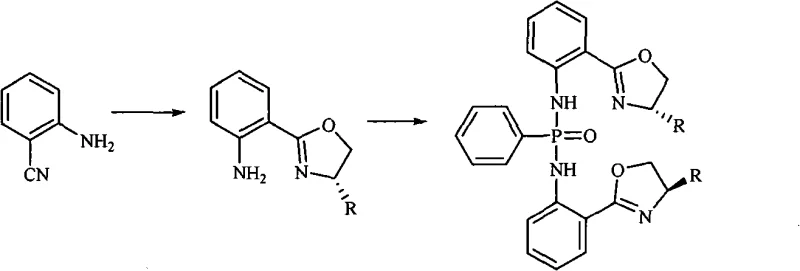 The subsequent phosphonylation step is elegantly simplified by using phenylphosphonyl dichloride in a toluene and triethylamine mixture, where triethylamine acts as an effective acid scavenger to neutralize the generated HCl. This two-step sequence eliminates the need for cryogenic cooling and reduces the sensitivity to moisture compared to previous art, thereby enhancing the operational safety and feasibility for commercial scale-up of complex chiral catalysts. The modular nature of the synthesis allows for the easy interchange of L-amino alcohols, such as L-leucinol or L-phenylalaninol, to generate a library of ligands with diverse steric properties. This flexibility is invaluable for process development teams seeking to optimize enantioselectivity for specific API intermediates without redesigning the entire synthetic backbone.
The subsequent phosphonylation step is elegantly simplified by using phenylphosphonyl dichloride in a toluene and triethylamine mixture, where triethylamine acts as an effective acid scavenger to neutralize the generated HCl. This two-step sequence eliminates the need for cryogenic cooling and reduces the sensitivity to moisture compared to previous art, thereby enhancing the operational safety and feasibility for commercial scale-up of complex chiral catalysts. The modular nature of the synthesis allows for the easy interchange of L-amino alcohols, such as L-leucinol or L-phenylalaninol, to generate a library of ligands with diverse steric properties. This flexibility is invaluable for process development teams seeking to optimize enantioselectivity for specific API intermediates without redesigning the entire synthetic backbone.
Mechanistic Insights into ZnCl2-Catalyzed Oxazoline Formation
The core of this synthetic strategy lies in the activation of the nitrile group in 2-cyanoaniline by the Lewis acid catalyst, zinc chloride (ZnCl2). In the first step, the zinc center coordinates with the nitrogen of the nitrile group, increasing its electrophilicity and facilitating the nucleophilic attack by the hydroxyl group of the L-amino alcohol. This intramolecular cyclization proceeds through a stable intermediate that eventually dehydrates to form the 4,5-dihydro-2-oxazole ring. The choice of chlorobenzene as a solvent is critical, as its high boiling point allows the reaction to proceed at temperatures conducive to overcoming the activation energy barrier while maintaining a homogeneous phase. The reaction time of 24 hours ensures complete conversion of the starting materials, minimizing the presence of unreacted amines that could interfere with the subsequent phosphonylation step. Understanding this mechanism is vital for troubleshooting potential batch failures, as the water content must be rigorously controlled to prevent hydrolysis of the nitrile or the catalyst.
Following the formation of the oxazoline intermediate, the second step involves the nucleophilic substitution at the phosphorus center of phenylphosphonyl dichloride. The amine nitrogen of the oxazoline-aniline intermediate attacks the phosphorus atom, displacing a chloride ion. Triethylamine is essential here not just as a base, but to form a triethylamine hydrochloride salt that precipitates or remains soluble depending on the solvent system, driving the equilibrium forward. The use of toluene as a co-solvent helps in managing the solubility of the growing molecular weight species. Impurity control is achieved through the careful selection of stoichiometry; using a slight excess of the amine intermediate ensures that the dichloride is fully consumed, preventing the formation of polymeric byproducts. The final purification via column chromatography, while labor-intensive on a small scale, can be adapted to crystallization or distillation protocols for larger batches, ensuring the high-purity bisphosphonamide required for sensitive asymmetric transformations.
How to Synthesize Chiral Bisphosphonamide Efficiently
The synthesis of these high-value chiral ligands requires strict adherence to anhydrous and oxygen-free conditions to maintain the integrity of the reactive intermediates and the final phosphonyl product. The process begins with the preparation of the oxazoline-aniline intermediate, which serves as the chiral building block for the entire molecule. Operators must ensure that all glassware is flame-dried and that solvents like chlorobenzene are distilled over drying agents prior to use. The reaction mixture is heated under reflux for a prolonged period to ensure maximum conversion, after which the solvent is removed under reduced pressure. The crude residue is then subjected to a liquid-liquid extraction using chloroform and water to remove inorganic zinc salts, followed by drying over anhydrous sodium sulfate. For the final coupling step, the purified intermediate is dissolved in toluene, and phenylphosphonyl dichloride is added slowly to control the exotherm. Detailed standardized synthetic steps for this process are provided in the guide below to ensure reproducibility across different laboratory settings.
- React 2-cyanoaniline with L-amino alcohol using ZnCl2 catalyst in chlorobenzene under reflux for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via chloroform extraction and column chromatography to ensure high purity before the second step.
- Condense the purified intermediate with phenylphosphonyl dichloride in toluene and triethylamine under reflux for 24 hours to yield the target bisphosphonamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in this patent offers substantial advantages regarding raw material availability and cost structure. The primary starting materials, 2-cyanoaniline and various L-amino alcohols, are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks. Unlike other chiral ligand syntheses that rely on scarce natural products or complex multi-step sequences, this two-step process minimizes the number of unit operations, directly translating to lower manufacturing overheads. The elimination of precious metal catalysts such as palladium or rhodium from the synthetic sequence removes a significant cost driver and simplifies the regulatory documentation required for metal residue analysis in the final product. This streamlined approach supports cost reduction in pharmaceutical intermediate manufacturing by lowering the overall cost of goods sold (COGS) for the ligand, which in turn makes the downstream asymmetric synthesis more economically viable.
- Cost Reduction in Manufacturing: The utilization of zinc chloride as a catalyst represents a strategic shift away from expensive noble metals, significantly lowering the raw material costs associated with the catalytic system. Zinc salts are inexpensive, widely available, and easier to handle than air-sensitive transition metal complexes, which reduces the infrastructure investment needed for storage and handling. Furthermore, the solvents employed, chlorobenzene and toluene, are standard industrial solvents that can be recovered and recycled through distillation, minimizing waste disposal costs and environmental levies. The high conversion rates observed in the patent examples suggest that raw material utilization is efficient, reducing the volume of unreacted starting materials that need to be separated and disposed of. By avoiding cryogenic conditions, the process also reduces energy consumption related to cooling, contributing to a leaner and more cost-effective production profile.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals ensures a stable supply chain that is less susceptible to geopolitical fluctuations or single-source dependencies. L-amino alcohols are produced on a large scale for various industries, ensuring consistent quality and steady availability throughout the year. The robustness of the reaction conditions, which tolerate standard reflux temperatures rather than requiring specialized low-temperature equipment, means that production can be easily transferred between different manufacturing sites without significant re-validation. This flexibility is crucial for supply chain heads who need to mitigate risks associated with plant maintenance or unexpected shutdowns at specific facilities. Additionally, the stability of the intermediate and final products allows for longer storage times, enabling manufacturers to build safety stock and reducing lead time for high-purity ligands when urgent orders arise.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs without fundamental changes to the chemistry. The absence of hazardous reagents like thionyl chloride or phosphorus trichloride in the main synthetic steps improves the safety profile of the process, aligning with modern EHS (Environment, Health, and Safety) standards. Waste generation is minimized through the use of catalytic amounts of zinc and the potential for solvent recycling, supporting sustainability goals and reducing the carbon footprint of the manufacturing process. The purification steps, primarily involving extraction and chromatography, can be optimized for larger scales using continuous processing technologies or preparative HPLC, ensuring that the environmental impact remains low even as production volumes increase to meet commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral bisphosphonamide compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing workflows. The responses cover aspects ranging from reaction optimization to the specific benefits of the zinc-catalyzed mechanism.
Q: What are the key advantages of the ZnCl2 catalyzed route for oxazoline synthesis?
A: The use of ZnCl2 as a catalyst allows for mild reaction conditions compared to traditional strong acid methods, reducing side reactions and improving the enantiomeric purity of the resulting oxazoline intermediate.
Q: How does the choice of R-group affect the catalytic performance?
A: The R-group (isobutyl, isopropyl, phenyl, or benzyl) modulates the steric environment around the phosphorus center, allowing fine-tuning of enantioselectivity for specific asymmetric transformations like mandelic acid synthesis.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like chlorobenzene and toluene and avoids expensive precious metal catalysts, making it highly amenable to cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisphosphonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the enantiomeric excess and chemical purity of every batch, guaranteeing that our products meet the exacting standards required for API synthesis. Our commitment to quality assurance means that clients can rely on us for consistent supply and technical support throughout the lifecycle of their projects.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to accelerate your drug development timeline with reliable, high-quality chiral intermediates.
