Advanced Synthesis of Dopamine Hydrochloride: A Technical Breakthrough for Industrial Scale-up
Advanced Synthesis of Dopamine Hydrochloride: A Technical Breakthrough for Industrial Scale-up
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for critical cardiovascular agents, and the preparation method detailed in patent CN111925293A represents a significant leap forward in the manufacturing of Dopamine Hydrochloride. This innovative protocol utilizes veratraldehyde as a foundational starting material, circumventing the inherent instability and supply chain bottlenecks associated with traditional precursors like vanillin or piperonylethylamine. By leveraging a sophisticated four-step sequence involving Henry condensation, catalytic transfer hydrogenation, and selective ether cleavage, this methodology delivers a product with exceptional purity levels reaching 99.98 percent, making it an ideal candidate for high-grade active pharmaceutical ingredient (API) production. For R&D directors and procurement strategists, understanding the nuances of this veratraldehyde-based route is essential for optimizing supply chains and ensuring the consistent availability of this life-saving medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dopamine hydrochloride has been plagued by significant technical and economic hurdles that compromise industrial feasibility. One prominent legacy route relies on piperonylethylamine as the starting material, which undergoes a ring-opening reaction under harsh acidic conditions. As illustrated in the reaction scheme below, this pathway suffers from the high cost and limited commercial availability of piperonylethylamine, creating a fragile supply chain vulnerable to market fluctuations.
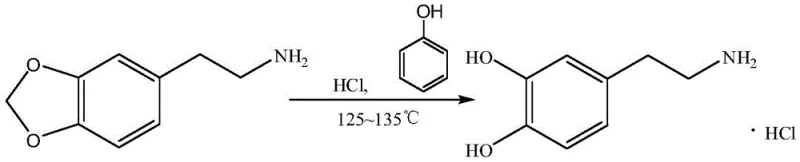
Furthermore, the reaction requires elevated temperatures that promote the formation of numerous byproducts, complicating downstream purification and reducing overall yield. Another common approach utilizes vanillin, which, while cheaper, introduces a highly reactive phenolic hydroxyl group early in the synthesis. This functional group is prone to oxidation, leading to the formation of colored quinone impurities that are difficult to remove. Additionally, the reduction step in the vanillin route traditionally employs zinc powder and hydrochloric acid, a combination that generates substantial amounts of hazardous solid waste and requires extensive wastewater treatment infrastructure, thereby inflating operational expenditures and environmental compliance burdens.
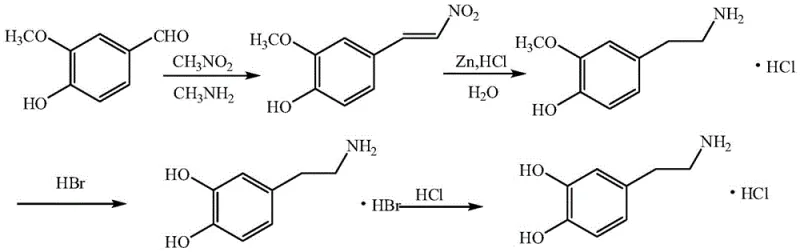
A third enzymatic route utilizing catechol and L-DOPA decarboxylase presents its own set of challenges, primarily due to the exorbitant cost of biocatalysts and the stringent reaction conditions required to maintain enzyme activity. These conventional methods collectively fail to offer a balanced solution that satisfies the trifecta of cost-efficiency, high purity, and environmental sustainability required by modern pharmaceutical manufacturing standards.

The Novel Approach
In stark contrast to these legacy methodologies, the novel approach disclosed in the patent data utilizes veratraldehyde, a stable and abundantly available aromatic aldehyde, to construct the dopamine skeleton with superior control over impurity profiles. By masking the phenolic hydroxyl groups as methoxy ethers during the initial carbon-carbon bond-forming steps, this strategy effectively prevents the oxidative degradation that plagues vanillin-based routes. The process initiates with a Henry condensation between veratraldehyde and nitromethane, followed by a mild catalytic reduction that avoids the use of high-pressure hydrogen gas. This strategic protection-deprotection sequence ensures that the sensitive catechol moiety is only revealed in the final stages of synthesis, minimizing exposure to oxidizing environments. Consequently, this route not only enhances the chemical stability of intermediates but also streamlines the purification process, resulting in a final product that meets rigorous pharmacopeial standards with minimal effort.
Mechanistic Insights into Veratraldehyde-Based Cascade Synthesis
The core of this technological advancement lies in the precise orchestration of organic transformations that maximize atom economy while minimizing side reactions. The initial step involves a base-catalyzed Henry reaction where veratraldehyde condenses with nitromethane in the presence of methylamine hydrochloride and an acid-binding agent such as triethylamine. This reaction proceeds efficiently at room temperature, typically between 10-30°C, to yield 3,4-dimethoxy-beta-nitrostyrene. The choice of mild conditions here is critical, as higher temperatures would accelerate polymerization and the formation of complex oligomeric byproducts. Following isolation, the nitrostyrene intermediate undergoes a transfer hydrogenation reaction using palladium-carbon as the catalyst and ammonium formate as the hydrogen donor. This mechanistic choice is particularly ingenious for industrial applications because it replaces dangerous high-pressure hydrogenation with a safe, atmospheric pressure process that generates nitrogen and carbon dioxide as benign gaseous byproducts.
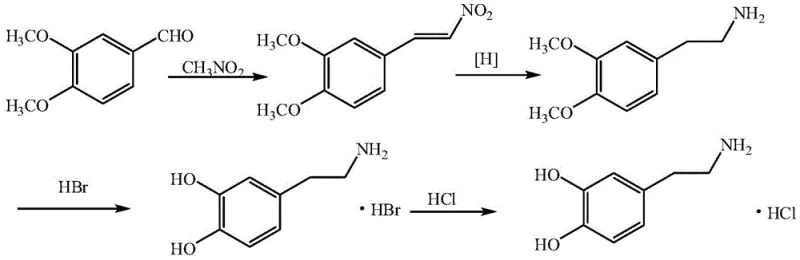
The final transformation involves the cleavage of the methyl ether protecting groups using hydrobromic acid at elevated temperatures of 110-120°C. This step converts the dimethoxy intermediate into the free catechol structure of dopamine hydrobromide, which is subsequently converted to the hydrochloride salt. The mechanism of ether cleavage is driven by the nucleophilic attack of bromide ions on the methyl groups, a reaction that is highly selective and robust. From an impurity control perspective, the absence of phenolic groups during the reduction phase means that there is no risk of forming quinone-imine species, which are notoriously difficult to purge. This mechanistic clarity allows for a much tighter control over the impurity spectrum, ensuring that the final API is suitable for parenteral administration without requiring complex chromatographic separations.
How to Synthesize Dopamine Hydrochloride Efficiently
Implementing this synthesis route in a pilot or commercial plant requires strict adherence to the optimized parameters regarding solvent selection, catalyst loading, and temperature control to ensure reproducibility. The process is designed to be operationally simple, utilizing common solvents like ethanol and methanol which are easily recovered and recycled, further enhancing the economic viability of the method. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and crystallization protocols are critical for maintaining the high yields reported in the patent examples. For technical teams looking to adopt this methodology, the following guide outlines the critical operational milestones necessary to achieve successful scale-up from laboratory to commercial production volumes.
- Condense veratraldehyde with nitromethane in ethanol using methylamine hydrochloride and triethylamine to form 3,4-dimethoxy-beta-nitrostyrene.
- Perform catalytic transfer hydrogenation on the nitrostyrene intermediate using palladium-carbon and ammonium formate in methanol to yield 3,4-dimethoxyphenethylamine.
- Cleave the methoxy groups using hydrobromic acid at elevated temperatures to generate dopamine hydrobromide.
- Convert the hydrobromide salt to the final dopamine hydrochloride product through reaction with hydrochloric acid in isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this veratraldehyde-based synthesis offers profound strategic benefits that extend beyond mere technical superiority. The primary advantage lies in the drastic simplification of the supply chain; veratraldehyde is a commodity chemical with a stable global supply, unlike specialized starting materials like piperonylethylamine which may suffer from availability constraints. Furthermore, the elimination of high-pressure hydrogenation equipment significantly reduces capital expenditure (CapEx) requirements for new production lines, as the transfer hydrogenation step can be performed in standard glass-lined reactors without the need for expensive autoclaves. This reduction in equipment complexity also translates to lower maintenance costs and reduced downtime, ensuring a more reliable delivery schedule for downstream customers.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the replacement of stoichiometric reducing agents like zinc powder with catalytic systems. In traditional vanillin routes, the consumption of large quantities of zinc and acid generates massive amounts of solid waste sludge, the disposal of which incurs significant environmental fees. By shifting to a catalytic hydrogen transfer system using ammonium formate, the process eliminates heavy metal waste streams entirely, leading to substantial savings in waste treatment costs. Additionally, the high yield of the condensation step, often exceeding 90 percent, ensures that raw material utilization is maximized, directly lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The robustness of the intermediates in this synthetic pathway provides a buffer against supply chain disruptions. Because the methoxy-protected intermediates are chemically stable and resistant to oxidation, they can be stored for extended periods without degradation, allowing manufacturers to build strategic inventory buffers. This stability contrasts sharply with phenolic intermediates that require inert atmosphere storage and rapid processing to prevent spoilage. Consequently, manufacturers adopting this route can offer more consistent lead times and guarantee supply continuity even during periods of raw material volatility, a critical factor for pharmaceutical buyers managing just-in-time inventory systems.
- Scalability and Environmental Compliance: From a regulatory and environmental standpoint, this process aligns perfectly with green chemistry principles. The absence of heavy metals and the use of benign solvents simplify the environmental impact assessment (EIA) for new facilities. The reduction in three wastes (waste water, waste gas, and waste residue) lowers the burden on effluent treatment plants, making it easier for manufacturers to comply with increasingly stringent environmental regulations. This environmental friendliness not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a sustainable partner, a quality that is increasingly valued by multinational pharmaceutical corporations in their vendor qualification audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior alternative to existing technologies. Understanding these specifics is vital for technical teams evaluating the feasibility of technology transfer and for procurement officers negotiating supply agreements based on process efficiency.
Q: Why is veratraldehyde preferred over vanillin for dopamine synthesis?
A: Veratraldehyde lacks the reactive phenolic hydroxyl group found in vanillin, preventing oxidative degradation and quinone formation during the process, which significantly improves impurity profiles and yield stability.
Q: What are the safety advantages of the transfer hydrogenation step?
A: The use of ammonium formate as a hydrogen donor allows the reduction to proceed at atmospheric pressure and moderate temperatures, eliminating the need for high-pressure hydrogen gas equipment and reducing explosion risks.
Q: How does this method address environmental concerns compared to zinc reduction?
A: Unlike traditional vanillin routes that utilize zinc powder and hydrochloric acid generating heavy metal waste, this catalytic method produces minimal inorganic waste, simplifying wastewater treatment and lowering environmental compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dopamine Hydrochloride Supplier
As the global demand for cardiovascular therapeutics continues to rise, securing a supply of high-purity dopamine hydrochloride produced via efficient and sustainable methods is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the veratraldehyde route to deliver products that meet the most stringent quality specifications. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. With our rigorous QC labs and commitment to stringent purity specifications, we guarantee that every batch of dopamine hydrochloride is free from critical impurities and ready for formulation.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our optimized grade can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-performance pharmaceutical intermediates.
