Advanced Deamination Reduction Technology for High-Purity 1H-Tetrazole-1-Acetic Acid Manufacturing
The pharmaceutical industry's relentless pursuit of safer and more efficient synthetic routes for critical antibiotic intermediates has led to significant breakthroughs in organic synthesis, specifically regarding the production of 1H-tetrazole-1-acetic acid. As detailed in patent CN102382071B, a novel technology utilizing a deamination reduction method has emerged as a superior alternative to legacy processes. This compound serves as a vital source material for first-generation cephalosporin antibiotics such as Ancef and Kefzol (cephazolin sodium), making its reliable supply chain essential for global healthcare infrastructure. The disclosed innovation fundamentally alters the reaction pathway by employing 5-amino-1H-tetrazoleacetic acid as the starting substrate, subjecting it to a controlled diazotization followed by reduction using sodium nitrite and hypophosphorous acid. This strategic shift not only enhances atom utilization but also mitigates the severe safety risks associated with traditional azide chemistry, offering a robust solution for industrial-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1H-tetrazole-1-acetic acid has been plagued by significant safety hazards and operational inefficiencies inherent to azide-based chemistry. The primary conventional route involves the reaction of ethyl chloroacetate with sodium azide, a process notorious for its extreme toxicity and the explosive instability of sodium azide, which poses catastrophic risks in large-scale reactor environments. Furthermore, this legacy method suffers from excessively long reaction times often exceeding 50 hours, coupled with cumbersome post-reaction processing that generates substantial three-waste pollution, thereby inflating environmental compliance costs. An alternative one-pot method utilizing glycine and sodium azide, while shorter in duration, presents its own set of challenges including difficult solvent recovery of glacial acetic acid and poor crystallization properties of the final product, rendering it unsuitable for consistent commercial production. These structural deficiencies in existing technologies create a bottleneck for reliable pharmaceutical intermediate supplier networks seeking to optimize cost reduction in API manufacturing.

The Novel Approach
In stark contrast to these hazardous legacy pathways, the patented deamination reduction technology introduces a streamlined and inherently safer synthetic strategy that bypasses the direct use of bulk sodium azide. By utilizing 5-amino-1H-tetrazole-1-acetic acid as the precursor, the process leverages a mild diazotization-reduction sequence that operates under significantly gentler conditions, typically ranging from -20°C to 20°C, which drastically lowers energy consumption and equipment stress. The innovation lies in the dual function of hypophosphorous acid, which acts simultaneously as the acidifier and the reducing agent, effectively removing the diazo group to yield the target 1H-tetrazole-1-acetic acid with high efficiency. This approach not only simplifies the operational workflow but also ensures a high degree of process safety and non-toxicity, making it highly conducive to industrialized enforcement and aligning perfectly with modern green chemistry principles for high-purity pharmaceutical intermediate production.
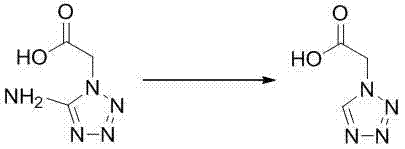
Mechanistic Insights into Deamination Reduction Chemistry
The core mechanistic advantage of this technology rests on the precise control of the diazotization and subsequent reduction steps, which are critical for minimizing impurity formation and maximizing yield. In this system, sodium nitrite serves as the diazotization reagent, reacting with the amino group of the substrate to form a transient diazonium salt intermediate under acidic conditions provided by the in-situ generated hypophosphorous acid. This hypophosphorous acid is economically produced within the reactor by reacting sodium dihydric hypophosphite with sulfuric acid, ensuring a steady supply of protons and reducing equivalents without the need for handling unstable liquid acids. The reduction step proceeds smoothly as the hypophosphorous acid donates hydrogen to the diazonium species, cleaving the nitrogen-nitrogen bond and releasing nitrogen gas while preserving the tetrazole ring integrity, a delicate balance that requires precise stoichiometric control to prevent ring degradation or over-reaction.
Impurity control is rigorously managed through the optimization of reaction parameters and downstream purification techniques, ensuring the final product meets stringent quality specifications required for antibiotic synthesis. The patent data highlights that maintaining the reaction temperature within the optimal window of -5°C to 0°C is crucial for suppressing side reactions that could lead to colored impurities or structural analogs. Following the reaction, the crude product is isolated via extraction with ethyl acetate, a solvent chosen for its favorable partition coefficients, and subsequently purified through recrystallization using isopropanol (Virahol). This purification step is instrumental in achieving the reported purity levels of above 97%, effectively removing residual salts, unreacted starting materials, and minor by-products, thus delivering a high-purity 1H-tetrazole-1-acetic acid suitable for direct use in sensitive cephalosporin coupling reactions.
How to Synthesize 1H-Tetrazole-1-Acetic Acid Efficiently
Implementing this advanced synthetic route requires careful attention to reagent addition rates and temperature profiles to ensure reproducibility and safety at scale. The process begins with the preparation of the reducing environment by dissolving sodium dihydric hypophosphite in water, followed by the sequential addition of sulfuric acid and the amino-tetrazole substrate, establishing the necessary acidic milieu before the introduction of the nitrite source. Operators must strictly adhere to the specified molar ratios, preferably 1:2:2:2 for the substrate, nitrite, hypophosphite, and sulfuric acid respectively, to drive the reaction to completion while minimizing excess reagent waste. The detailed standardized synthesis steps, including specific stirring times and workup procedures, are outlined below to guide technical teams in replicating this high-yield process.
- Prepare the reaction mixture by dissolving sodium dihydric hypophosphite in water, adding sulfuric acid and 5-amino-1H-tetrazole-1-acetic acid sequentially.
- Maintain temperature between -20°C and 20°C while slowly dripping aqueous sodium nitrite solution to effect diazotization.
- Stir for 4 to 6 hours, extract with ethyl acetate, evaporate solvent, and recrystallize the residue using isopropanol to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this deamination reduction technology translates into tangible strategic benefits that extend far beyond simple chemical conversion. By eliminating the reliance on sodium azide, organizations can significantly reduce the regulatory burden and insurance costs associated with storing and transporting explosive hazardous materials, thereby streamlining logistics and enhancing overall facility safety profiles. The simplified operational flow, characterized by shorter reaction times and easier workup procedures compared to the 50-hour legacy cycles, allows for higher equipment utilization rates and faster batch turnover, which is critical for meeting tight production schedules in the competitive antibiotic market. Furthermore, the use of readily available and stable raw materials like sodium dihydric hypophosphite ensures a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages or quality fluctuations common with specialized azide reagents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive hazard mitigation protocols and the efficient use of reagents through in-situ acid generation. By avoiding the complex waste treatment systems required for azide-containing effluents, manufacturers can achieve substantial cost savings in environmental compliance and waste disposal operations. Additionally, the high atom utilization and improved yield range of 70% to 80% mean that less raw material is wasted per kilogram of finished product, directly lowering the cost of goods sold and improving margin potential for high-volume commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The stability of the starting materials and the robustness of the reaction conditions contribute to a more predictable and reliable supply of 1H-tetrazole-1-acetic acid. Unlike processes sensitive to trace moisture or requiring cryogenic conditions that strain utility infrastructure, this method operates under mild temperatures that are easily maintained with standard industrial cooling systems. This operational flexibility reduces the likelihood of batch failures due to equipment malfunction or utility spikes, ensuring consistent delivery timelines and reducing lead time for high-purity pharmaceutical intermediates needed for downstream antibiotic formulation.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates significantly less hazardous waste, aligning with increasingly strict global regulations on chemical manufacturing emissions. The ability to recycle solvents like ethyl acetate with high efficiency further minimizes the environmental footprint, making the technology sustainable for long-term industrial deployment. The simplicity of the reaction setup facilitates easy scale-up from pilot plants to multi-ton production facilities without the need for specialized explosion-proof reactors, allowing for rapid capacity expansion to meet surging market demand for cephalosporin precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deamination reduction technology, based on the specific data and embodiments provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining the high purity and safety standards demanded by the pharmaceutical industry.
Q: Why is the deamination reduction method safer than traditional azide routes?
A: Traditional methods rely on sodium azide, which is highly toxic and explosive. This new method uses stable 5-amino-1H-tetrazole-1-acetic acid and avoids handling bulk azides.
Q: What purity levels can be achieved with this novel process?
A: The patent data indicates that through optimized recrystallization, product purity consistently reaches above 97%, with specific embodiments achieving up to 98.5%.
Q: How does the in-situ generation of hypophosphorous acid benefit the process?
A: Generating hypophosphorous acid from sodium dihydric hypophosphite and sulfuric acid reduces raw material costs and simplifies logistics by using stable solid precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-Tetrazole-1-Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics, and we are committed to delivering excellence through advanced process technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1H-tetrazole-1-acetic acid meets the exacting standards required for GMP-compliant antibiotic synthesis, providing our partners with the confidence they need to plan their long-term production schedules.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this safer and more cost-effective synthetic route for their supply chains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency and secure your supply of this vital pharmaceutical intermediate.
