Advanced Green Synthesis of Ceftriaxone Sodium for Commercial Scale-up
Introduction to Next-Generation Ceftriaxone Sodium Synthesis
The pharmaceutical landscape for third-generation cephalosporins is undergoing a significant transformation driven by the urgent need for greener, safer, and more efficient manufacturing processes. Patent CN111440197A introduces a groundbreaking preparation method for Ceftriaxone Sodium that fundamentally reimagines the condensation strategy used to construct this critical antibiotic. Unlike conventional routes that rely on hazardous active esters, this innovation utilizes a direct condensation between 7-ACT guanidine salt and aminothiazolyloximate anhydride. This strategic shift not only streamlines the synthetic pathway but also addresses critical quality concerns regarding genotoxic impurities. For R&D directors and procurement specialists seeking a reliable antibiotic intermediate supplier, understanding this technological leap is paramount. The method promises to deliver high-purity Ceftriaxone Sodium while drastically simplifying the downstream processing requirements.
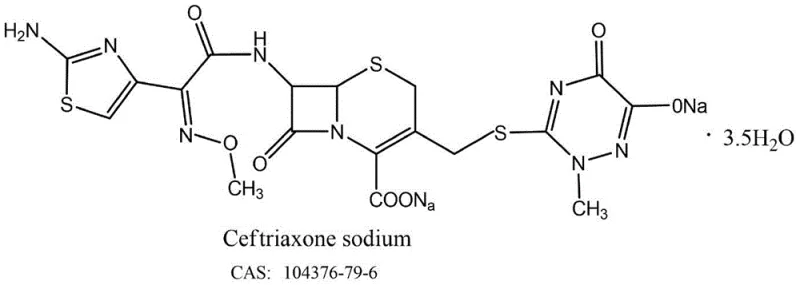
At the heart of this innovation lies the elimination of 2-mercaptobenzothiazole, a notorious byproduct associated with traditional AE-active ester methods. This compound poses significant genotoxicity risks, necessitating rigorous and costly purification steps to ensure patient safety. By bypassing the formation of this toxic species entirely, the new process aligns perfectly with modern regulatory standards for impurity control. Furthermore, the reaction conditions described in the patent, utilizing solvents such as dichloromethane and acetone, are well-suited for existing infrastructure in fine chemical plants. This compatibility reduces the barrier to entry for manufacturers looking to adopt this superior technology, offering a clear path toward cost reduction in cephalosporin manufacturing without compromising on the stringent purity specifications required for parenteral antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ceftriaxone Sodium has relied heavily on the condensation of 7-Aminocephalosporanic Acid (7-ACA) derivatives with AE-active esters, specifically 2-methoxyimino-2-(2-amino-4-thiazolyl)-(Z)-thioacetic acid phenylhydrazine thiazole ester. While effective in forming the necessary amide bond, this legacy approach suffers from a critical flaw: the generation of 2-mercaptobenzothiazole as a stoichiometric byproduct. This sulfur-containing heterocycle is not only environmentally persistent but also carries genotoxic alerts that trigger intense scrutiny from global health authorities. Removing trace amounts of this impurity to acceptable levels often requires multiple recrystallizations or chromatographic separations, which drastically erodes overall yield and increases solvent consumption. Additionally, the handling of phenylhydrazine derivatives introduces safety hazards due to their potential instability and toxicity, complicating the operational safety profile of the manufacturing facility.
The Novel Approach
In stark contrast, the method disclosed in CN111440197A employs a clever activation strategy using aminothiazolyloximate anhydride. This reagent acts as a highly reactive acylating agent that couples efficiently with the 7-ACT guanidine salt without releasing the problematic thiazole thiol. The reaction pathway is depicted clearly in the schematic below, illustrating the direct formation of the ceftriaxone acid nucleus followed by salt formation. This approach effectively decouples the side-chain activation from the generation of toxic leaving groups. By utilizing pivaloyl chloride to generate the anhydride in situ or as a pre-formed reagent, the process achieves a cleaner reaction profile. The absence of the mercaptobenzothiazole byproduct means that the workup procedure is significantly simplified, often requiring only a straightforward extraction and crystallization. This results in a much higher throughput and a substantially reduced environmental footprint, making it an ideal candidate for commercial scale-up of complex beta-lactams.
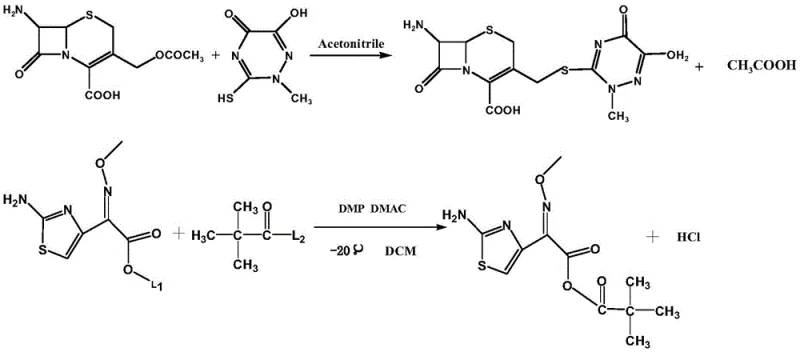
Mechanistic Insights into Anhydride-Mediated Condensation
The core of this synthetic breakthrough lies in the precise generation and utilization of the aminothiazolyloximate anhydride. The mechanism begins with the activation of aminothiazolyloximate (Compound 1) using a chlorinating agent such as pivaloyl chloride in the presence of a base like triethylamine. This reaction, conducted at cryogenic temperatures between -40°C and -50°C, forms a mixed anhydride species that is highly electrophilic at the carbonyl carbon adjacent to the oxime ether. This specific activation mode is crucial because it prevents the racemization of the sensitive beta-lactam ring while ensuring rapid acylation of the amine nucleophile. The use of low temperatures is not merely a preference but a necessity to suppress side reactions such as the hydrolysis of the anhydride or the degradation of the oxime geometry, which is critical for the biological activity of the final antibiotic. The patent specifies a molar ratio of compound 1 to the chlorinating agent of roughly 1:1 to 1:1.4, ensuring complete conversion without excessive reagent waste.
Following the activation, the 7-ACT guanidine salt serves as the nucleophilic partner. The guanidine base plays a dual role: it solubilizes the 7-ACT acid in organic solvents like dichloromethane by forming a soluble salt, and it maintains a basic environment that facilitates the nucleophilic attack on the anhydride. The reaction proceeds smoothly at temperatures below 10°C, typically around -40°C to -50°C, to maintain stereochemical integrity. Once the amide bond is formed, yielding the ceftriaxone acid intermediate, the process transitions to salt formation. The addition of an aqueous sodium acetate solution allows for the extraction of the product into the aqueous phase as the sodium salt, leaving behind organic-soluble impurities. This phase separation is a powerful purification tool that leverages the amphiphilic nature of the molecule. The final crystallization from acetone yields the stable Ceftriaxone Sodium sesquihydrate or anhydrous form, depending on drying conditions, with exceptional purity profiles that meet pharmacopoeial standards.
How to Synthesize Ceftriaxone Sodium Efficiently
Implementing this synthesis requires careful attention to thermal management and reagent stoichiometry to replicate the high yields reported in the patent examples. The process is divided into three critical stages: the preparation of the 7-ACT salt solution, the generation of the anhydride activator, and the final coupling and isolation. Operators must ensure that all solvents are anhydrous during the activation and coupling steps to prevent premature hydrolysis of the reactive anhydride species. The transition from the organic reaction mixture to the aqueous extraction must be controlled to maximize recovery. For a detailed breakdown of the specific operational parameters, reagent grades, and step-by-step instructions required to execute this protocol in a pilot or production setting, please refer to the standardized synthesis guide below.
- Prepare 7-ACT guanidine salt by reacting 7-ACT with an organic base like tetramethylguanidine in dichloromethane at -20 to -30°C.
- Synthesize aminothiazolyloximate anhydride by reacting aminothiazolyloximate with pivaloyl chloride and triethylamine at -40 to -50°C.
- Condense the 7-ACT salt with the anhydride solution, followed by extraction with sodium acetate and crystallization in acetone to obtain the final solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary value driver is the elimination of the toxic 2-mercaptobenzothiazole byproduct, which traditionally necessitates expensive waste treatment protocols and specialized disposal services. By removing this hazard from the process stream, manufacturers can achieve substantial cost savings in environmental compliance and waste management. Furthermore, the simplified workup procedure, which avoids complex chromatographic purifications, reduces the consumption of silica gel and large volumes of elution solvents. This streamlining of the downstream process translates directly into shorter batch cycle times and increased plant capacity, allowing suppliers to respond more agilely to market demand fluctuations for this essential antibiotic.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in raw material complexity and waste disposal costs. Traditional methods require the purchase and handling of AE-active esters, which are more expensive to produce and store due to their stability issues. In contrast, the anhydride method utilizes commodity chemicals like pivaloyl chloride and aminothiazolyloximate, which are readily available and cost-effective. Additionally, the high conversion rates demonstrated in the patent examples imply that less starting material is wasted, improving the overall atom economy of the synthesis. The ability to recycle solvents like dichloromethane and acetone further enhances the cost efficiency, making the final API more competitive in price-sensitive markets without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of raw materials. The reagents used in this method, such as 7-ACT and aminothiazolyloximate, are established intermediates with mature supply chains, reducing the risk of bottlenecks associated with exotic or proprietary catalysts. The process tolerance, indicated by the consistent yields across different examples in the patent, suggests that the method is forgiving of minor variations in scale, which is crucial for maintaining continuity during technology transfer from lab to plant. This reliability ensures that pharmaceutical companies can secure a steady flow of high-quality Ceftriaxone Sodium, mitigating the risk of production stoppages that could impact the availability of life-saving medications.
- Scalability and Environmental Compliance: From a sustainability perspective, this method represents a significant step forward in green chemistry for the pharmaceutical industry. The avoidance of genotoxic impurities simplifies the regulatory filing process, as there is no need to validate complex clearance studies for 2-mercaptobenzothiazole. This accelerates the time-to-market for generic versions or new formulations. Moreover, the reduced solvent usage and simpler isolation steps lower the energy intensity of the manufacturing process. The scalability is evidenced by the use of standard unit operations like liquid-liquid extraction and crystallization, which are easily adaptable from 100 kgs to multi-ton scales. This makes the technology highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their cephalosporin portfolio with a sustainable and compliant offering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in CN111440197A, providing a factual basis for evaluating the technology's potential. Understanding these nuances is essential for technical teams assessing the feasibility of adopting this route for their own production lines or for procurement teams validating the quality credentials of their suppliers.
Q: How does this new method improve safety compared to traditional AE-active ester routes?
A: The traditional method generates 2-mercaptobenzothiazole, a genotoxic byproduct. This novel anhydride route completely eliminates this hazardous substance, significantly enhancing drug safety and reducing environmental waste treatment costs.
Q: What are the critical temperature controls required for this synthesis?
A: Strict low-temperature control is essential. The formation of the 7-ACT salt requires -20 to -30°C, while the anhydride formation and subsequent condensation must be maintained between -40 to -50°C to ensure high conversion rates and prevent degradation.
Q: Can this process be scaled for industrial production?
A: Yes, the process uses common solvents like dichloromethane and acetone and avoids complex purification steps for toxic byproducts. The patent demonstrates molar yields exceeding 95%, indicating strong feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftriaxone Sodium Supplier
As the global demand for high-quality antibiotics continues to rise, partnering with a manufacturer that possesses advanced synthetic capabilities is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge technologies like the anhydride condensation method to deliver superior products. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of Ceftriaxone Sodium meets or exceeds international pharmacopoeial standards. Our commitment to quality assurance means that you receive a product that is not only potent but also free from the genotoxic impurities that plague older manufacturing methods.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced processes can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Whether you are looking for a reliable source of API intermediates or a long-term partner for finished dosage form ingredients, NINGBO INNO PHARMCHEM is dedicated to supporting your success with transparency, expertise, and unwavering reliability.
