Advanced Cefixime Manufacturing: High-Purity API Intermediate Production via Optimized One-Pot Hydrolysis
The global demand for third-generation cephalosporin antibiotics continues to drive innovation in pharmaceutical intermediate manufacturing, specifically for Cefixime, a critical broad-spectrum agent. Patent CN112300198B introduces a transformative synthesis methodology that addresses long-standing inefficiencies in the production of this vital API. Unlike conventional routes that suffer from cumbersome post-processing and toxic reagent usage, this novel approach leverages a streamlined sequence of etherification, catalytic hydrogenation, and a distinctive one-pot acylation-hydrolysis strategy. The core innovation lies in the ability to bypass the isolation of unstable ester intermediates, thereby preserving yield and enhancing overall product quality. 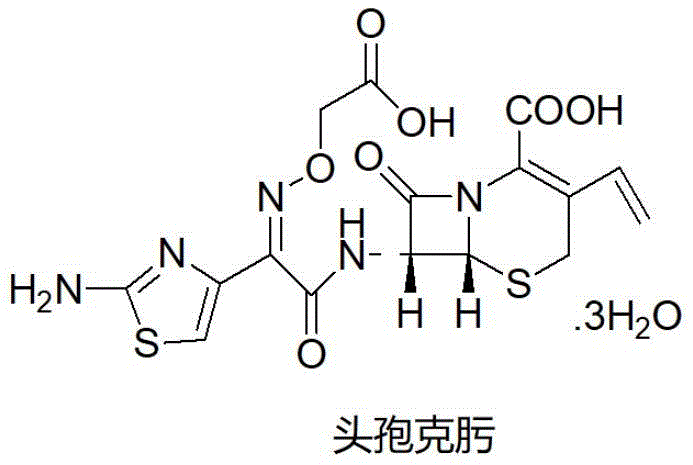 For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is essential for securing a competitive supply chain.
For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefixime has been plagued by significant technical bottlenecks that inflate costs and compromise environmental safety. Traditional methods, such as those described in WO2006103686A1, often rely on the activation of side-chain acids using hazardous reagents like phosphorus pentachloride or complex active esters that require rigorous purification. These legacy processes typically involve multiple isolation steps, including repeated extractions, activated carbon decolorization, and pH adjustments, which lead to substantial product loss. Furthermore, the use of high-boiling solvents like DMF in hydrolysis steps can result in difficult-to-remove residues, negatively impacting the final purity profile. The cumulative effect of these inefficiencies is a lower total yield, often necessitating larger reactor volumes and higher raw material consumption to meet production targets, which directly erodes profit margins for manufacturers.
The Novel Approach
The methodology disclosed in CN112300198B fundamentally restructures the synthesis workflow to eliminate these inefficiencies. A pivotal advancement is the direct hydrolysis performed within the original acylation reaction system, removing the need to isolate the Cefixime ethyl ester intermediate. 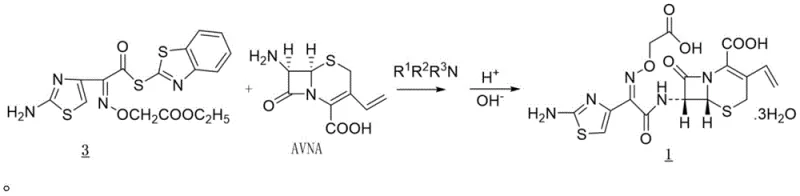 By coupling the acylation of the AVNA nucleus with the active ester Compound 3 and immediately proceeding to alkaline hydrolysis, the process minimizes exposure of the sensitive beta-lactam ring to harsh isolation conditions. This telescoping of steps not only simplifies the operational workflow but also drastically reduces the generation of impurities that typically arise during intermediate handling. The result is a robust, environmentally safer process that delivers high-purity Cefixime with significantly reduced solvent usage and waste generation, aligning perfectly with modern green chemistry principles.
By coupling the acylation of the AVNA nucleus with the active ester Compound 3 and immediately proceeding to alkaline hydrolysis, the process minimizes exposure of the sensitive beta-lactam ring to harsh isolation conditions. This telescoping of steps not only simplifies the operational workflow but also drastically reduces the generation of impurities that typically arise during intermediate handling. The result is a robust, environmentally safer process that delivers high-purity Cefixime with significantly reduced solvent usage and waste generation, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Etherification and Active Ester Formation
The synthesis begins with a precise etherification reaction where Compound 6 reacts with ethyl bromoacetate under basic conditions. 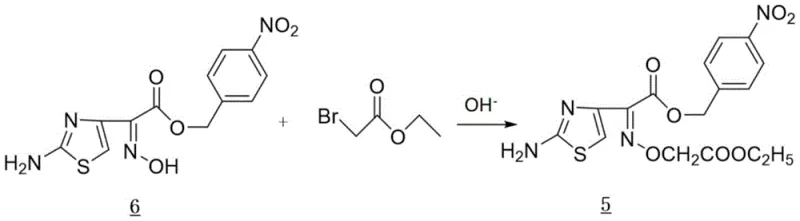 This step utilizes carbonate bases such as potassium carbonate or sodium carbonate in polar aprotic solvents like DMF or DMAc at controlled temperatures between 40°C and 50°C. The choice of base and solvent is critical; it ensures complete conversion while minimizing side reactions that could lead to colored impurities. Following this, the debenzylation step employs catalytic hydrogenation using Pd/C or Raney Nickel in a mixed solvent system containing acid. This specific acidic environment facilitates the cleavage of the benzyl protecting group without degrading the sensitive oxime functionality, yielding Compound 4 with high fidelity. The subsequent activation of Compound 4 involves reacting it with the DM promoter and a trisubstituted phosphine, such as triphenylphosphine. This generates the highly reactive thioester Compound 3 in situ, which is immediately primed for coupling with the AVNA nucleus, ensuring maximum atom economy and reaction efficiency.
This step utilizes carbonate bases such as potassium carbonate or sodium carbonate in polar aprotic solvents like DMF or DMAc at controlled temperatures between 40°C and 50°C. The choice of base and solvent is critical; it ensures complete conversion while minimizing side reactions that could lead to colored impurities. Following this, the debenzylation step employs catalytic hydrogenation using Pd/C or Raney Nickel in a mixed solvent system containing acid. This specific acidic environment facilitates the cleavage of the benzyl protecting group without degrading the sensitive oxime functionality, yielding Compound 4 with high fidelity. The subsequent activation of Compound 4 involves reacting it with the DM promoter and a trisubstituted phosphine, such as triphenylphosphine. This generates the highly reactive thioester Compound 3 in situ, which is immediately primed for coupling with the AVNA nucleus, ensuring maximum atom economy and reaction efficiency.
Impurity control is meticulously managed throughout this sequence, particularly during the final acylation and hydrolysis phases. The patent specifies the use of organic amines like triethylamine or tri-n-butylamine to maintain optimal pH levels during the coupling reaction, preventing the premature hydrolysis of the active ester before it reacts with the AVNA nucleus. Furthermore, the hydrolysis is conducted at low temperatures, typically between 0°C and 15°C, using sodium hydroxide. This thermal control is vital for preserving the stereochemical integrity of the C-6 and C-7 chiral centers in the cephem nucleus. By avoiding the isolation of the ester intermediate, the process prevents the accumulation of degradation products that often occur during drying and storage of unstable beta-lactam esters. The final crystallization is achieved by precise pH adjustment to the isoelectric point, ensuring that the resulting Cefixime trihydrate possesses the correct crystal habit and purity required for downstream pharmaceutical formulation.
How to Synthesize Cefixime Efficiently
The synthesis of Cefixime via this patented route offers a clear pathway for laboratories and pilot plants to achieve superior yields and purity profiles. The process is defined by four distinct chemical transformations that transition smoothly from simple starting materials to the complex antibiotic structure. Operators must pay close attention to temperature controls during the exothermic etherification and the sensitive hydrogenation steps to ensure safety and reproducibility. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Perform etherification of Compound 6 with ethyl bromoacetate using carbonate bases in polar aprotic solvents like DMF or DMAc to yield Compound 5.
- Execute catalytic hydrogenation of Compound 5 using Pd/C or Raney Nickel in an alcohol-acid mixed system to remove the benzyl protecting group, forming Compound 4.
- Activate Compound 4 using DM promoter and trisubstituted phosphine to form the active ester Compound 3, followed by direct acylation with AVNA and in-situ hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology translates into tangible strategic benefits beyond mere technical superiority. The elimination of toxic reagents like phosphorus pentachloride removes the regulatory burden and disposal costs associated with hazardous waste management. Additionally, the simplification of the workflow reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced labor hours per batch. This efficiency gain allows for faster turnaround times and increased production capacity without the need for significant capital investment in new infrastructure. Consequently, partners can expect a more stable supply of high-quality intermediates with reduced risk of production delays caused by complex purification bottlenecks.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the telescoping of the acylation and hydrolysis steps. By eliminating the isolation, drying, and re-dissolution of the Cefixime ethyl ester intermediate, manufacturers save significantly on solvent consumption and processing time. The removal of expensive and hazardous activating agents further lowers the raw material cost bill. Moreover, the high yield achieved in each step means less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final API. These cumulative savings create a more competitive pricing structure for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as ethyl bromoacetate, carbonate bases, and common organic solvents ensures a resilient supply chain. Unlike processes dependent on specialized or volatile reagents that may face market shortages, this route utilizes commodity chemicals that are easily sourced from multiple vendors. The robustness of the catalytic hydrogenation step, which tolerates a range of conditions, further adds to process reliability. This stability minimizes the risk of batch failures and ensures consistent delivery schedules, which is critical for maintaining uninterrupted production lines for finished dosage forms.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex antibiotics, featuring straightforward workup procedures like filtration and crystallization that translate well from pilot to production scale. The absence of toxic phosphorus by-products simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. This eco-friendly profile not only reduces compliance costs but also enhances the corporate sustainability credentials of the manufacturing partner. The ability to scale without encountering significant exothermic hazards or purification difficulties makes this technology a low-risk option for expanding production capacity to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefixime synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on yield expectations, purity standards, and operational safety. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing manufacturing portfolios.
Q: How does the new synthesis method improve upon traditional PCl5 activation routes?
A: Unlike traditional methods utilizing phosphorus pentachloride which generate significant toxic waste and require complex purification, this patented route employs DM promoters and trisubstituted phosphines. This substitution eliminates hazardous phosphorus by-products, simplifies the post-reaction workup to simple filtration and crystallization, and significantly enhances the environmental safety profile of the manufacturing process.
Q: What are the purity specifications achievable with this one-pot hydrolysis technique?
A: The optimized process, particularly the direct hydrolysis step performed in the original reaction system after acylation, minimizes product loss and impurity generation associated with isolation of intermediate esters. Experimental data from the patent indicates that the final Cefixime product achieves an HPLC purity exceeding 99%, with controlled moisture content suitable for direct pharmaceutical formulation.
Q: Is this synthesis route scalable for industrial production of antibiotic intermediates?
A: Yes, the route is designed for scalability. Key steps such as the etherification and hydrogenation utilize standard industrial solvents like DMF, methanol, and ethyl acetate. The elimination of multiple isolation steps between acylation and hydrolysis reduces equipment turnover time and solvent consumption, making it highly suitable for commercial scale-up of complex antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefixime Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain leadership in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112300198B are fully realized in practical manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Cefixime meets the highest international standards. Our commitment to process excellence means we can deliver high-purity pharmaceutical intermediates with the consistency and reliability required by top-tier drug manufacturers.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product quality while reducing overall production costs. Let us be your partner in delivering superior antibiotic solutions to the global market.
