Advanced Synthesis of Prednisone Intermediates: Scalable Routes for Commercial Manufacturing
Advanced Synthesis of Prednisone Intermediates: Scalable Routes for Commercial Manufacturing
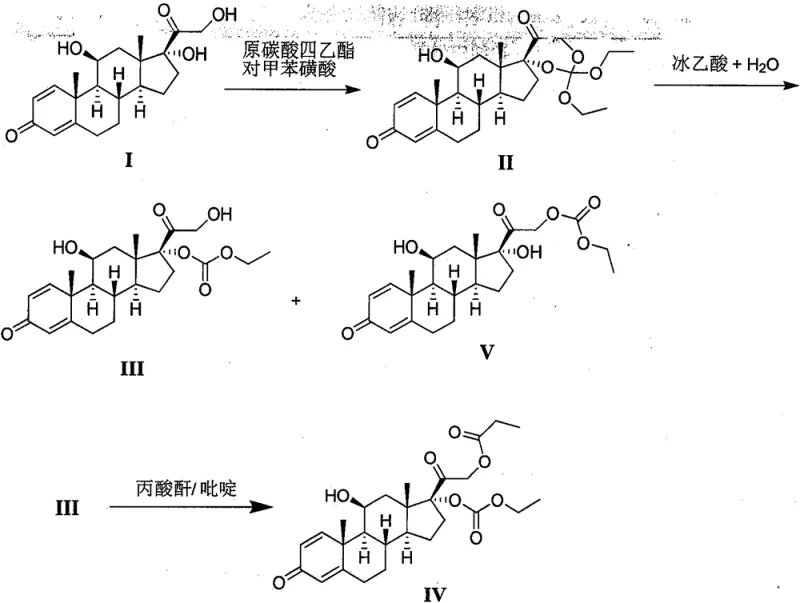
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of potent corticosteroids, and patent CN110684068A presents a significant breakthrough in the synthesis of a key prednisone intermediate, specifically the compound designated as Formula III. This intermediate serves as a critical precursor in the manufacture of prednisone, a halogen-free adrenocortical hormone drug renowned for its anti-inflammatory and anti-allergic properties. The disclosed technology addresses long-standing challenges in the prior art, particularly regarding low conversion rates and the difficulties associated with separating and purifying the target molecule. By optimizing the hydrolysis conditions of the precursor compound (Formula II), this method enables the convenient and rapid acquisition of high-purity intermediates, thereby establishing a more reliable foundation for the downstream production of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the compound of Formula III has been plagued by inefficient processes that hinder large-scale commercial viability. Traditional methods, such as those described in earlier literature, often rely on the use of tetrahydrofuran or 1,4-dioxane as solvents, followed by tedious purification steps involving silica gel column chromatography. These conventional approaches suffer from severe drawbacks, including low reaction conversions due to the formation of unwanted side products like Formula I and Formula V during the hydrolysis stage. Furthermore, the reliance on column chromatography is a major bottleneck for industrial application, as it is time-consuming, costly, and difficult to scale. The subsequent concentration and grinding with diethyl ether to obtain crystals often result in poor yields, typically around 36%, making the process economically unfeasible for high-volume manufacturing requirements.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a streamlined hydrolysis strategy that dramatically enhances both efficiency and product quality. By employing a mixed solvent system comprising water and lower aliphatic alcohols, acetone, or acetonitrile, the new method facilitates a much cleaner conversion of Formula II to Formula III. The process operates under mild acidic conditions, utilizing catalysts such as ammonium chloride or acetic acid, which effectively suppresses side reactions and transesterification issues often seen with methanol solvents. This innovation allows for the direct precipitation of the product upon addition to saline or water, completely bypassing the need for complex chromatographic separation. As a result, the process yields a solid product that is easily filtered and dried, achieving significantly higher purity and yield profiles that are fully compatible with industrial-scale operations.
Mechanistic Insights into Acid-Catalyzed Hydrolysis
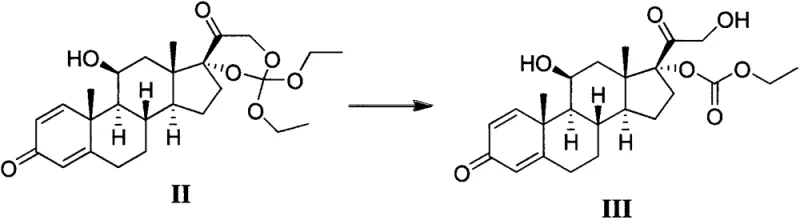
The core of this technological advancement lies in the precise control of the hydrolysis mechanism, which converts the orthoester or protected functionality in Formula II into the free hydroxyl group found in Formula III. The reaction proceeds efficiently in a biphasic or homogeneous mixture of water and organic co-solvents at temperatures ranging from 10°C to 60°C. The choice of acid catalyst is critical; while strong mineral acids can be used, the patent highlights the efficacy of weaker organic acids like acetic acid or salts like ammonium chloride. These milder acidic conditions are sufficient to catalyze the cleavage of the protecting group without degrading the sensitive steroid backbone or promoting the migration of double bonds. This careful balance ensures that the structural integrity of the pregna-1,4-diene-3,20-dione skeleton is maintained throughout the transformation, minimizing the formation of impurities that would otherwise complicate downstream processing.
Furthermore, the mechanistic pathway is designed to mitigate the risk of transesterification, a common pitfall in steroid chemistry when alcoholic solvents are present. In previous methods, the use of methanol often led to the exchange of ester groups, reducing the overall yield and complicating the impurity profile. The new method optimizes the solvent ratio and acid strength to favor hydrolysis over alcoholysis. By controlling the reaction temperature, preferably between 35°C and 45°C, the kinetic energy is sufficient to drive the hydrolysis to completion while thermodynamic conditions disfavor unwanted side reactions. This results in a reaction mixture where the target compound precipitates readily upon quenching, indicating a high degree of supersaturation and purity that facilitates easy isolation without the need for extensive washing or recrystallization steps initially.
How to Synthesize Prednisone Intermediate Efficiently
The synthesis of this critical prednisone intermediate involves a straightforward hydrolysis protocol that transforms the complex precursor Formula II into the desired Formula III with exceptional efficiency. The process begins by dissolving the starting material in a carefully selected solvent system, typically a mixture of ethanol and water, which provides the necessary polarity for the reaction while maintaining solubility. An acid source, such as ammonium chloride or glacial acetic acid, is then introduced to initiate the catalytic cycle at controlled temperatures. Following the reaction period, which can be monitored via TLC to ensure complete conversion, the product is isolated through a simple precipitation technique involving the addition of the reaction mixture to a saline solution. This elegant workflow eliminates the need for hazardous solvents and complex equipment, making it an ideal candidate for technology transfer and scale-up in GMP environments.
- Dissolve the starting material (Formula II) in a mixed solvent system comprising water and a C1-C4 lower aliphatic alcohol, acetone, or acetonitrile.
- Add an acid catalyst such as acetic acid, propionic acid, or ammonium chloride and maintain the reaction temperature between 10°C and 60°C.
- Upon completion, precipitate the product by adding the reaction mixture to saline or water, followed by filtration and optional recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational reliability. The most significant advantage is the complete elimination of silica gel column chromatography, a unit operation that is notoriously expensive, slow, and difficult to validate for large-scale production. By replacing this bottleneck with a simple precipitation and filtration step, manufacturers can drastically reduce processing time and solvent consumption. This simplification not only lowers the direct cost of goods sold but also reduces the environmental footprint of the manufacturing process by minimizing waste generation. Consequently, this leads to substantial cost savings in the manufacturing of pharmaceutical intermediates, allowing for more competitive pricing strategies in the global market.
- Cost Reduction in Manufacturing: The removal of chromatographic purification represents a massive reduction in operational expenditures, as it eliminates the cost of silica gel, the large volumes of elution solvents, and the associated disposal fees. Additionally, the high conversion rates achieved in this process mean that less raw material is wasted on side products, further driving down the cost per kilogram of the final intermediate. The ability to use common, inexpensive solvents like ethanol and water instead of specialized ethers or dioxanes also contributes to a leaner cost structure, ensuring that the production of high-purity pharmaceutical intermediates remains economically viable even at fluctuating raw material prices.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemical process ensures a consistent and reliable flow of materials. The simplified workup procedure reduces the risk of batch failures associated with complex purification steps, thereby enhancing the overall reliability of the supply chain. Since the method does not rely on hard-to-source reagents or specialized equipment, it mitigates the risk of supply disruptions. The high yields reported in the patent examples demonstrate that the process is reproducible, which is essential for maintaining steady inventory levels and meeting the rigorous delivery schedules demanded by downstream API manufacturers.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is seamless with this methodology, as evidenced by the successful execution of multi-kilogram batches described in the patent data. The use of aqueous workups and ethanol-based systems aligns well with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The reduction in hazardous waste and the potential for solvent recovery make this process highly scalable, allowing manufacturers to ramp up production from hundreds of kilograms to multi-ton capacities without encountering the engineering challenges typical of chromatography-dependent processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of prednisone intermediates, derived directly from the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the operational parameters and quality outcomes associated with this advanced manufacturing technique. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this new hydrolysis method over prior art?
A: The new method significantly improves conversion rates and eliminates the need for silica gel column chromatography, which was required in previous methods. This allows for direct crystallization and filtration, drastically simplifying the purification process and making it suitable for large-scale industrial production.
Q: What specific reaction conditions are optimal for synthesizing Formula III?
A: Optimal conditions involve hydrolyzing Formula II in a mixed solvent of water and ethanol or methanol at temperatures between 20°C and 50°C. The use of ammonium chloride or acetic acid as the acid source has been shown to provide high yields and purity without promoting transesterification side reactions.
Q: How does this process impact the purity of the final prednisone intermediate?
A: By avoiding harsh conditions and incompatible solvents like pure methanol which can cause transesterification, this method achieves crude purities exceeding 75% and final purities up to 97.99% after simple recrystallization, ensuring a high-quality feedstock for subsequent esterification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain, and we are uniquely positioned to leverage this advanced synthesis technology for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is smooth and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of prednisone intermediate meets the highest international standards. Our commitment to technical excellence allows us to deliver complex steroid intermediates with the consistency and reliability that top-tier pharmaceutical companies demand.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can add value to your prednisone production program.
