Scaling Biological Dehydrogenation for High-Purity Glucocorticoid Intermediates
Introduction to Advanced Biocatalytic Steroid Synthesis
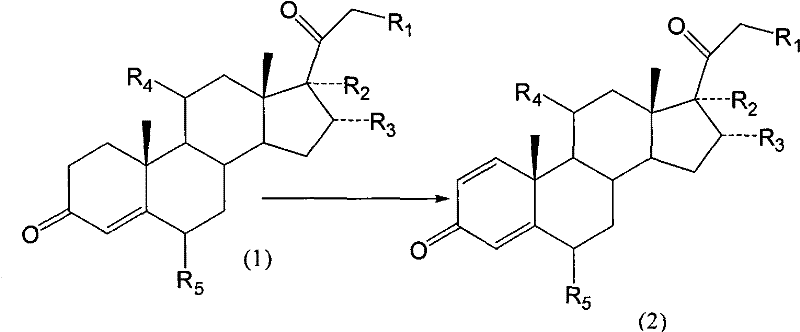
The pharmaceutical industry is currently witnessing a paradigm shift in the manufacturing of critical glucocorticoid intermediates, driven by the urgent need for greener, safer, and more efficient synthetic routes. Patent CN101760496A introduces a groundbreaking biological dehydrogenation preparation method that utilizes Arthrobacter simplex to transform specific steroid substrates into high-value 1,2-dehydrogenated compounds. This technology addresses the longstanding challenges associated with traditional chemical dehydrogenation, particularly the reliance on hazardous reagents and the resulting impurity profiles that complicate downstream purification. For R&D directors and procurement specialists, understanding this biocatalytic pathway is essential for securing a reliable steroid intermediate supplier capable of meeting stringent global regulatory standards while optimizing production costs. The transition from chemical oxidation to enzymatic transformation represents not just a technical upgrade, but a strategic supply chain advantage that ensures continuity and compliance in the manufacturing of anti-inflammatory APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the 1,2-double bond in the A-ring of steroid molecules, a critical structural feature for enhancing anti-inflammatory activity, has relied heavily on chemical dehydrogenation using selenium dioxide (SeO2). This conventional approach is fraught with significant operational and safety liabilities that modern pharmaceutical manufacturers strive to eliminate. Selenium dioxide is a highly toxic substance that poses severe health risks to personnel and creates substantial environmental burdens due to the difficulty in treating selenium-containing waste streams. Furthermore, the chemical process typically suffers from low reaction yields, often hovering around only 55%, which drastically impacts the overall economic efficiency of the synthesis. Perhaps most critically for quality control teams, the residual micro-levels of selenium in the final product are extremely difficult to eliminate completely, leading to potential failures in meeting strict heavy metal specifications required for active pharmaceutical ingredients. These factors combined make the traditional SeO2 route increasingly untenable for sustainable, large-scale commercial production.
The Novel Approach
In stark contrast to the hazardous chemical legacy methods, the novel biological dehydrogenation method disclosed in the patent leverages the specific enzymatic activity of Arthrobacter simplex to achieve the same structural transformation with superior outcomes. This biocatalytic route operates under mild conditions, typically maintaining fermentation temperatures between 30°C and 34°C and a neutral pH range of 7.0 to 7.2, which significantly reduces energy consumption compared to harsh chemical reactions. The process demonstrates a remarkable improvement in transformation efficiency, with conversion rates consistently reaching between 70% and 90%, effectively doubling the productivity of older methods. By utilizing a biological system, the process inherently avoids the introduction of toxic heavy metals, thereby simplifying the purification workflow and ensuring a cleaner impurity profile. This shift not only enhances product quality but also aligns perfectly with the growing global demand for green chemistry practices in the synthesis of complex pharmaceutical intermediates.
Mechanistic Insights into Arthrobacter Simplex Catalyzed Dehydrogenation
The core of this technological advancement lies in the specific regioselectivity and stereoselectivity of the dehydrogenase enzymes produced by the Arthrobacter simplex strain during fermentation. Unlike chemical oxidants which may attack multiple sites on the complex steroid nucleus, the biological catalyst targets specifically the C1 and C2 positions of the A-ring to introduce the double bond without affecting other sensitive functional groups such as the C3 ketone or the side chain at C17. This high degree of specificity is crucial for maintaining the integrity of the molecule, especially when dealing with substrates containing multiple chiral centers and hydroxyl groups that are prone to epimerization or oxidation under chemical conditions. The mechanism involves the abstraction of hydrogen atoms from the C1 and C2 positions, facilitated by the enzyme's active site, leading to the formation of the conjugated enone system characteristic of potent glucocorticoids. Understanding this mechanistic precision allows process chemists to better predict the behavior of various substituted steroid substrates, ensuring that the method is robust across a wide range of analogues including 6-alpha-methyl and 11-beta-hydroxy derivatives.
Furthermore, the control of impurities in this biological system is governed by the metabolic pathways of the microorganism and the specific fermentation conditions employed. The patent highlights the importance of substrate presentation, noting that the method of feeding the steroid into the fermentation tank—whether as a micronized powder with a particle size D90 ≤ 100 μm or dissolved in organic solvents like methanol or ethanol—significantly influences the transformation rate. The addition of chaotropic agents such as Tween-80 is another critical mechanistic factor; these agents likely modify the cell membrane permeability or the solubility of the hydrophobic steroid substrate in the aqueous fermentation broth, thereby enhancing the mass transfer of the substrate to the enzymatic active sites. By optimizing these parameters, manufacturers can minimize the formation of by-products and ensure that the reaction proceeds to completion within a reasonable timeframe of 30 to 72 hours, providing a predictable and controllable manufacturing process that is essential for GMP compliance.
How to Synthesize 1,2-Dehydrogenated Steroids Efficiently
The implementation of this biological dehydrogenation process requires a disciplined approach to fermentation management and downstream processing to maximize yield and purity. The procedure begins with the careful preparation of the Arthrobacter simplex seed culture, progressing through slant culture, primary cultivation, and secondary cultivation stages to ensure a high density of viable cells before the introduction of the steroid substrate. Once the fermentation tank is inoculated, the substrate is introduced either in a dissolved state using solvents such as tetrahydrofuran or dioxane, or as a fine suspension, with the concentration strictly controlled to remain below 4% to prevent substrate inhibition. The reaction is monitored closely, typically lasting between 30 to 72 hours depending on the specific substrate and strain used, after which the reaction is terminated by heating the broth to deactivate the bacteria. For a comprehensive breakdown of the exact operational parameters and step-by-step instructions, please refer to the standardized synthesis guide below.
- Prepare the Arthrobacter simplex strain (e.g., AS 1.754 or AS 1.94*) through slant culture, primary cultivation, and secondary cultivation in a glucose and yeast extract medium at 30-34°C.
- Introduce the steroid substrate (Formula 1) into the fermentation tank either as micronized powder (D90 ≤ 100 μm) or dissolved in solvents like methanol or ethanol, maintaining a substrate concentration of 1-3%.
- Conduct the biotransformation at 30-34°C for 30-72 hours, optionally adding Tween-80 as a chaotropic agent, then terminate by heating to 70-90°C and extract the product with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biological dehydrogenation technology offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of selenium dioxide from the supply chain removes a major bottleneck related to the sourcing, handling, and disposal of hazardous chemicals, which often incur significant regulatory fees and logistical complexities. By switching to a fermentation-based process that utilizes readily available nutrients like glucose and yeast extract, companies can drastically simplify their raw material procurement strategy and reduce dependency on specialized chemical suppliers. This transition also mitigates the risk of supply chain disruptions caused by environmental regulations tightening around heavy metal usage, ensuring a more resilient and continuous production flow for critical steroid intermediates. The ability to produce high-purity materials without the need for extensive heavy metal scavenging steps translates directly into reduced processing time and lower operational expenditures.
- Cost Reduction in Manufacturing: The shift to biological catalysis fundamentally alters the cost structure of steroid intermediate production by eliminating the expensive and complex purification steps required to remove selenium residues. In traditional chemical synthesis, significant resources are allocated to chromatography and specialized filtration to meet heavy metal limits; the bio-process bypasses these needs entirely, leading to substantial cost savings in both consumables and labor. Additionally, the higher conversion rates of 70% to 90% mean that less starting material is wasted, improving the overall atom economy and reducing the cost of goods sold. The use of common fermentation media components further drives down raw material costs compared to proprietary chemical oxidants, creating a more economically sustainable manufacturing model.
- Enhanced Supply Chain Reliability: Relying on biological systems enhances supply chain stability by diversifying the input requirements away from volatile chemical markets. The strains used, such as Arthrobacter simplex AS 1.754, are well-characterized and can be maintained in-house, reducing the risk of external supply shocks. Furthermore, the milder reaction conditions reduce wear and tear on reactor equipment and lower the safety risks associated with storing and transporting toxic reagents, leading to fewer unplanned shutdowns and maintenance delays. This reliability is crucial for meeting the just-in-time delivery expectations of downstream API manufacturers and ensuring that production schedules are met consistently without interruption.
- Scalability and Environmental Compliance: Fermentation processes are inherently scalable, allowing for seamless transition from laboratory benchtop experiments to industrial-scale bioreactors ranging from hundreds of liters to thousands of cubic meters. This scalability ensures that the technology can grow with market demand without requiring fundamental changes to the process chemistry. From an environmental perspective, the bio-process generates significantly less hazardous waste, simplifying compliance with increasingly strict environmental protection laws. The absence of selenium waste streams reduces the burden on wastewater treatment facilities and lowers the carbon footprint of the manufacturing operation, aligning corporate sustainability goals with operational reality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biological dehydrogenation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of biological dehydrogenation over SeO2 methods?
A: Biological dehydrogenation eliminates the use of highly toxic selenium dioxide (SeO2), thereby removing the risk of heavy metal residues in the final API and significantly reducing environmental pollution and waste treatment costs associated with selenium disposal.
Q: What is the typical conversion rate for this biological process?
A: According to the patent data, the dehydrogenation transformation rate using Arthrobacter simplex can reach between 70% to 90%, which is substantially higher than the approximately 55% yield typically observed with traditional chemical dehydrogenation methods.
Q: Which steroid substrates are compatible with this fermentation method?
A: The process is versatile and applicable to various pregna-steroid intermediates, including 6-alpha-methyl-11-beta,17-alpha-dihydroxy-pregna-4-ene-3,20-dione and its derivatives, making it suitable for synthesizing multiple glucocorticoid drugs like prednisone and methylprednisolone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Steroid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced biocatalytic processes requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from pilot scale to full industrialization. Our facilities are equipped with state-of-the-art fermentation tanks and rigorous QC labs capable of handling complex steroid transformations with stringent purity specifications. We understand the critical nature of glucocorticoid intermediates in the global pharmaceutical supply chain and are committed to delivering products that meet the highest international quality standards, free from the impurities associated with legacy chemical methods.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior biological technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target steroid intermediates. Let us help you secure a competitive advantage in the market with a safer, more efficient, and scalable synthesis route.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
