Advanced Synthesis of Moxifloxacin Impurity: Technical Upgrade and Commercial Scalability
Advanced Synthesis of Moxifloxacin Impurity: Technical Upgrade and Commercial Scalability
The pharmaceutical industry continuously demands higher standards for impurity profiling to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). Patent CN112961153B introduces a groundbreaking preparation method for a specific moxifloxacin impurity, designated as MXX014-31, which addresses critical limitations in existing synthetic methodologies. This innovation provides a robust, safe, and high-yielding pathway that is essential for quality control laboratories and process development teams working on fourth-generation quinolone antibiotics. By shifting away from hazardous reagents and optimizing reaction conditions, this technology offers a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in producing high-purity reference standards. The following analysis details the technical mechanisms and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinolone derivatives and their associated impurities often relied on harsh chemical transformations that posed significant safety and environmental challenges. Traditional routes frequently utilized highly toxic substances such as sodium cyanide under high-temperature conditions to effect decarboxylation or structural modifications. As illustrated in the reference reaction below, the use of NaCN in DMSO represents a legacy approach that carries severe risks regarding operator safety and waste disposal compliance.
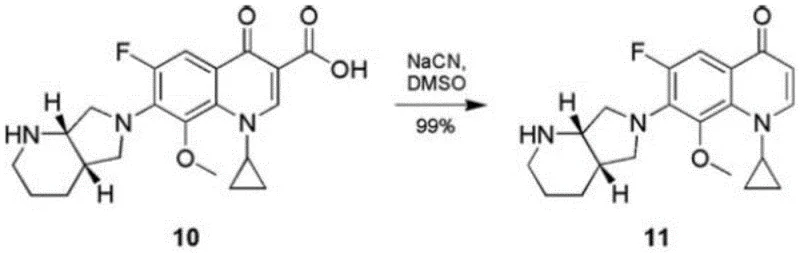
Furthermore, these conventional methods often suffered from poor selectivity, leading to complex impurity profiles that were difficult to purify. The reliance on extreme reaction parameters not only increased energy consumption but also limited the scalability of the process due to safety interlocks and containment requirements. For procurement and supply chain managers, sourcing materials produced via such hazardous routes introduces volatility and potential regulatory bottlenecks, making the search for alternative, greener chemistries a strategic priority for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112961153B employs a sophisticated four-step sequence that prioritizes safety and selectivity without compromising yield. The new route begins with the strategic protection of the piperazine nitrogen, followed by a regioselective iodination, a palladium-catalyzed reduction, and a final acidic deprotection. This logical progression allows for precise control over the molecular architecture, avoiding the chaotic side reactions common in older methods. The overall transformation is depicted in the comprehensive scheme below, highlighting the conversion of the starting API into the target impurity through stable intermediates.
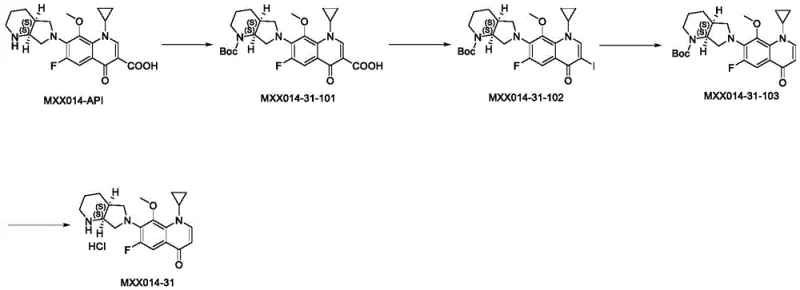
This novel approach effectively circumvents the use of cyanides and high-temperature extremes, replacing them with mild, controllable conditions using reagents like Boc anhydride and Pd/C. The result is a process that is not only safer for the workforce but also generates a product with superior purity profiles. For organizations focused on the commercial scale-up of complex quinolone derivatives, this route represents a significant technological leap, enabling the production of critical impurity standards with greater consistency and lower operational risk.
Mechanistic Insights into Boc-Protection and Catalytic Deiodination
The core of this synthetic success lies in the meticulous management of functional group reactivity, starting with the protection of the secondary amine on the diazabicyclo nonane ring. In the first step, Moxifloxacin (MXX014-API) reacts with di-tert-butyl dicarbonate (Boc anhydride) in the presence of a base like triethanolamine. This step is crucial as it masks the nucleophilic nitrogen, preventing it from interfering with subsequent electrophilic substitutions. The choice of dichloromethane as a solvent ensures excellent solubility and reaction homogeneity, facilitating the formation of the carbamate intermediate MXX014-31-101 with high efficiency. This protection strategy is a hallmark of modern organic synthesis, allowing for orthogonal reactivity in later stages.
Following protection, the process utilizes an iodination-deiodination strategy to achieve the desired structural modification. The iodination step introduces an iodine atom at the C-3 position of the quinolone ring using molecular iodine and sodium bicarbonate. This temporary modification serves as a handle for the subsequent reduction. The critical third step involves catalytic hydrogenation using Pd/C in methanol, which cleanly removes the iodine atom to restore the double bond character or saturate the specific position as required, yielding MXX014-31-103. Finally, the removal of the Boc group using hydrochloric acid in ethanol regenerates the free amine, delivering the final hydrochloride salt. This sequence demonstrates exceptional impurity control mechanisms, as each step produces intermediates that are easily purified, ensuring the final product meets rigorous quality specifications.
How to Synthesize MXX014-31 Efficiently
The synthesis of MXX014-31 is a streamlined process that leverages standard laboratory equipment and widely available reagents to achieve high yields. The procedure is divided into four distinct operational units: protection, iodination, reduction, and deprotection. Each stage has been optimized for molar ratios and solvent volumes to maximize throughput while minimizing waste. For research and development teams looking to implement this protocol, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Protect the secondary amine of Moxifloxacin (MXX014-API) using Boc anhydride and Triethanolamine in dichloromethane to form compound MXX014-31-101.
- Perform iodination on compound MXX014-31-101 using iodine and sodium bicarbonate in a THF/water mixture at elevated temperatures to yield compound MXX014-31-102.
- Execute a catalytic hydrogenation reduction using Pd/C in methanol to remove the iodine atom, converting MXX014-31-102 into the deiodinated intermediate MXX014-31-103.
- Finalize the synthesis by deprotecting the Boc group using hydrochloric acid in ethanol to obtain the target moxifloxacin impurity (MXX014-31) as a hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates into tangible operational benefits that extend beyond mere chemical yield. The elimination of highly toxic cyanides simplifies the regulatory compliance landscape, reducing the administrative burden associated with hazardous material handling and disposal. This shift towards greener chemistry aligns with global sustainability goals and enhances the long-term viability of the supply chain. Furthermore, the use of common solvents and catalysts ensures that raw material sourcing remains stable and cost-effective, mitigating the risk of supply disruptions caused by specialized reagent shortages.
- Cost Reduction in Manufacturing: The new process achieves significant cost optimization by removing the need for expensive containment systems required for cyanide handling. By utilizing standard glassware and common reagents like Boc anhydride and Pd/C, the capital expenditure for setting up production lines is drastically lowered. Additionally, the high purity of the crude product reduces the burden on downstream purification processes, such as chromatography or recrystallization, leading to substantial savings in solvent consumption and labor hours. The overall efficiency of the route ensures that the cost per gram of the high-purity moxifloxacin impurity is competitively low.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method bolsters security by relying on commodity chemicals rather than restricted or scarce reagents. The robustness of the four-step sequence means that production batches are less prone to failure due to sensitive reaction conditions. This stability allows for more accurate forecasting and inventory planning, reducing lead time for high-purity reference standards. Suppliers adopting this route can guarantee consistent delivery schedules, which is critical for pharmaceutical companies managing tight regulatory submission timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently scalable due to its reliance on heterogeneous catalysis and simple workup procedures. The use of Pd/C allows for easy filtration and catalyst recovery, minimizing heavy metal waste. Moreover, the avoidance of cyanide waste streams significantly lowers the environmental impact, simplifying wastewater treatment requirements. This eco-friendly profile facilitates easier permitting for larger production facilities, ensuring that the supply of this critical impurity can grow in tandem with the demand for moxifloxacin APIs without encountering environmental bottlenecks.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the safety, purity, and scalability of the process described in patent CN112961153B. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities.
Q: How does this new synthesis method improve safety compared to traditional routes?
A: The patented method eliminates the use of highly toxic sodium cyanide and high-temperature conditions required in conventional decarboxylation pathways. By utilizing a mild Boc-protection and iodination strategy followed by catalytic reduction, the process significantly reduces occupational health hazards and environmental risks associated with cyanide waste.
Q: What purity levels can be achieved with this manufacturing process?
A: The process is designed to achieve exceptionally high purity, with experimental data demonstrating HPLC purity levels exceeding 99.9%. The specific sequence of protection, selective iodination, and controlled deprotection minimizes the formation of side products, ensuring the material meets stringent requirements for reference standards and impurity profiling.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes commercially available starting materials and standard reagents like Boc anhydride, iodine, and Pd/C. The avoidance of exotic catalysts and the use of common solvents such as dichloromethane, THF, and ethanol facilitate straightforward scale-up from laboratory to industrial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moxifloxacin Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurities play in drug development and quality control. Our team of expert chemists has extensively analyzed the pathway described in CN112961153B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this complex intermediate with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this route, ensuring a seamless transition from pilot scale to full commercial manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and supply security.
