Revolutionizing Beta-Lactam Production: The Two-Enzyme One-Step Route to High-Purity D-7-ACA
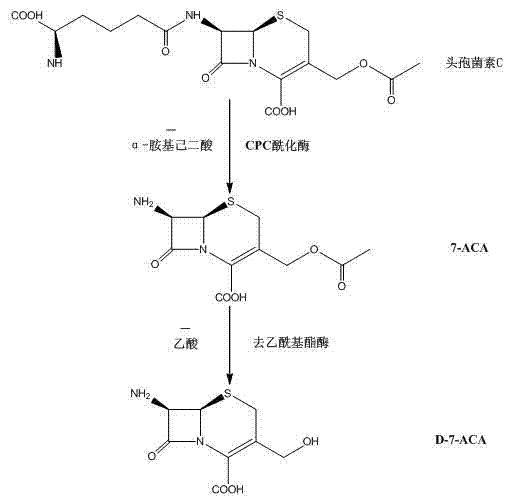
The global demand for advanced cephalosporin antibiotics continues to drive innovation in the synthesis of key pharmaceutical intermediates, specifically D-7-ACA (3-deacetyl-7-aminocephalosporanic acid). As a critical building block for third, fourth, and emerging fifth-generation cephalosporins such as cefuroxime and cefpodoxime axetil, the efficiency of D-7-ACA production directly impacts the cost and availability of life-saving medications. A pivotal advancement in this domain is detailed in patent CN102827912B, which discloses a groundbreaking technology for preparing D-7-ACA via a two-enzyme carrier one-step method. This innovation represents a paradigm shift from traditional multi-step chemical or enzymatic processes, offering a sustainable, high-yield pathway that aligns with the rigorous standards of modern green chemistry. By utilizing Cephalosporin C sodium salt concentrate as a direct substrate and employing a synergistic combination of immobilized CPC acylase and immobilized deacetyl esterase, this technology effectively bypasses the complex isolation of 7-ACA, thereby streamlining the entire manufacturing workflow. For R&D directors and procurement strategists, understanding the mechanistic elegance and commercial viability of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold in antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-7-ACA has been plagued by significant technical and economic hurdles inherent to both chemical and older enzymatic methodologies. The traditional chemical method relies on harsh cracking conditions involving high temperatures, high pressures, and extremely low cooling requirements, which impose a heavy energy burden on manufacturing facilities. Furthermore, this route necessitates the use of toxic and environmentally hazardous organic solvents such as dichloromethane, aniline, and chlorosilanes, alongside large quantities of strong acids and alkalis. These factors not only escalate pollution control costs but also introduce severe safety risks and regulatory compliance challenges. Alternatively, the existing biological enzymatic methods, often referred to as the three-enzyme two-step method, involve immobilized D-amino acid oxidase, GL-7-ACA acylase, and deacetyl esterase. While greener than chemical synthesis, this approach is operationally cumbersome and suffers from a prolonged production cycle. Critically, the oxidative cleavage step mediated by D-amino acid oxidase requires substantial amounts of pure oxygen or liquid oxygen, leading to excessive power consumption and creating potential explosion hazards that complicate plant safety protocols. Additionally, the crystallization of enzymatic intermediates often results in sticky products that are difficult to centrifuge and dry, necessitating additional solvents and separation equipment that further inflate operational expenditures.
The Novel Approach
In stark contrast to these legacy technologies, the two-enzyme one-step method described in the patent offers a streamlined, robust alternative that fundamentally restructures the production logic. By replacing the D-amino acid oxidase and GL-7-ACA acylase system with a specialized immobilized CPC acylase (supported on LKZ118 resin) combined with immobilized deacetyl esterase, the process achieves direct conversion of Cephalosporin C extract to D-7-ACA in a single reaction vessel. This consolidation eliminates the need for intermediate isolation and the dangerous oxygen-dependent oxidation step, thereby significantly shortening the production period and enhancing overall process safety. The reaction conditions are remarkably mild, operating effectively within a temperature range of 18-25°C and a controlled pH environment, which preserves enzyme stability and maximizes catalytic efficiency. This approach not only simplifies the equipment requirements, removing the need for complex cryogenic or high-pressure infrastructure, but also drastically reduces the generation of hazardous by-products. The result is a sustainable technology route that improves the utilization rate of Cephalosporin C fermentation components while delivering a high-purity product with minimal environmental footprint, making it an ideal candidate for modern, eco-conscious pharmaceutical manufacturing.
Mechanistic Insights into Dual-Enzyme Synergistic Catalysis
The core scientific breakthrough of this technology lies in the precise orchestration of two distinct enzymatic activities within a unified reaction matrix. The process initiates with the hydrolysis of the amide side chain of Cephalosporin C, catalyzed by the immobilized CPC acylase. Unlike traditional acylases that might require prior oxidation, this specific enzyme formulation is capable of acting directly on the native Cephalosporin C structure under alkaline conditions. Simultaneously, the immobilized deacetyl esterase targets the acetoxy group at the C-3 position of the cephalosporin nucleus. The synergy between these two enzymes is critical; they operate optimally under nearly identical conditions, allowing them to function concurrently without mutual interference. The reaction mechanism involves the nucleophilic attack on the carbonyl carbons of the respective ester and amide bonds, facilitated by the active sites of the immobilized enzymes. This dual hydrolysis releases alpha-aminoadipic acid and acetic acid as by-products, which are continuously neutralized by the automatic addition of ammonia water to maintain the reaction pH between 8.0 and 8.5. Maintaining this specific pH window is vital, as deviations can lead to rapid enzyme deactivation or incomplete conversion. The use of LKZ118 resin as a carrier provides a stable microenvironment that protects the enzymes from shear stress and denaturation, ensuring prolonged catalytic life and consistent performance throughout the batch cycle.
Furthermore, the control of impurities is intrinsically linked to the mildness of this enzymatic regime. In chemical cracking, the aggressive conditions often lead to ring-opening of the beta-lactam nucleus or epimerization at the chiral centers, generating structurally related impurities that are difficult to separate. In this enzymatic pathway, the specificity of the biocatalysts ensures that only the targeted bonds are cleaved, preserving the integrity of the sensitive beta-lactam ring. The subsequent crystallization step, performed by lowering the temperature to 0-10°C and adjusting the pH to 3.0-5.0 using hydrochloric acid, leverages the solubility differences between the product and residual substrates. This gentle precipitation method avoids the formation of amorphous or sticky solids common in other enzymatic processes, yielding a free-flowing crystalline powder with a purity consistently above 98%. The high selectivity of the enzymes minimizes the formation of polymeric by-products, thereby reducing the load on downstream purification units and ensuring a cleaner final API intermediate profile.
How to Synthesize D-7-ACA Efficiently
The implementation of this two-enzyme one-step method requires precise control over substrate concentration and enzyme loading to maximize economic efficiency. The patent outlines a standardized protocol where Cephalosporin C sodium salt concentrate is diluted to a specific titer range of 28000-32000 u/mL. This concentration balance is crucial; too high a concentration can lead to prolonged reaction times and product degradation, while too low a concentration increases batch frequency and operational costs. The optimized ratio of substrate units to enzyme activity units is maintained between 4000 and 5000 for both the CPC acylase and the deacetyl esterase. This stoichiometric balance ensures that neither enzyme becomes a bottleneck in the reaction kinetics. Following the lysis phase, the process transitions seamlessly to crystallization without the need for intermediate extraction, showcasing the true 'one-step' nature of the technology. For a detailed breakdown of the specific operational parameters, including exact reagent grades and mixing speeds, please refer to the standardized synthesis guide below.
- Dilute Cephalosporin C sodium salt concentrate to 28000-32000 u/mL and adjust initial pH to 6.5-7.5 using ammonia water.
- Add the substrate to a reactor containing immobilized CPC acylase and immobilized deacetyl esterase, maintaining temperature at 18-25°C.
- Automatically regulate reaction pH to 8.0-8.5 during lysis, then cool to 0-10°C and adjust pH to 3.0-5.0 for crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this two-enzyme technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The primary value driver is the substantial reduction in manufacturing complexity, which directly correlates to lower operational expenditures. By eliminating the requirement for liquid oxygen and the associated high-power consumption of the oxidative step found in three-enzyme methods, the facility's utility costs are significantly decreased. Moreover, the removal of toxic organic solvents like dichloromethane eradicates the costly waste treatment and disposal fees associated with hazardous chemical management. This shift not only lowers the variable cost per kilogram but also mitigates the regulatory risks associated with volatile organic compound (VOC) emissions. The simplified equipment list, which no longer requires specialized cryogenic or high-pressure reactors, allows for faster installation and reduced capital expenditure for new production lines, enhancing the agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the high utilization rate of the Cephalosporin C fermentation components. Since the process converts the extract directly to D-7-ACA without isolating 7-ACA, there is a marked reduction in material loss typically incurred during multiple crystallization and filtration steps. The use of immobilized enzymes further contributes to cost efficiency, as these biocatalysts can often be recycled for multiple batches, spreading the initial enzyme cost over a larger production volume. Additionally, the mild reaction conditions reduce the energy load required for heating and cooling, leading to a leaner energy profile. The avoidance of expensive separation equipment needed for sticky enzymatic intermediates further trims the capital and maintenance budget, resulting in a more competitive cost structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the shortened production cycle is a game-changer. The elimination of the cumbersome oxidation step and the reduction in processing time mean that batch turnover is accelerated, allowing for higher throughput within the same facility footprint. This increased velocity enhances the responsiveness of the supply chain, enabling manufacturers to fulfill large orders with shorter lead times. Furthermore, the reliance on commercially available raw materials and the robustness of the immobilized enzyme systems reduce the risk of production stoppages due to reagent shortages or catalyst instability. The improved safety profile, devoid of liquid oxygen hazards, also minimizes the risk of unplanned shutdowns due to safety incidents, ensuring a more consistent and reliable flow of goods to downstream API manufacturers.
- Scalability and Environmental Compliance: The inherent design of this process favors large-scale commercialization. The reaction parameters are easily controllable in standard stainless steel vessels, meaning that scaling from pilot to commercial production does not require exotic engineering solutions. This scalability ensures that supply can be ramped up to meet global demand without compromising quality. Environmentally, the process aligns perfectly with increasingly stringent global regulations on pharmaceutical manufacturing. The absence of heavy metal catalysts and chlorinated solvents simplifies the environmental impact assessment and permits easier compliance with green manufacturing standards. The aqueous nature of the waste stream, primarily containing benign organic acids like alpha-aminoadipic acid and acetic acid, makes wastewater treatment more straightforward and less costly, reinforcing the company's commitment to sustainable development.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel D-7-ACA synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what stakeholders can expect during technology transfer and scale-up. Understanding these nuances is critical for making informed decisions about integrating this biocatalytic platform into existing production portfolios.
Q: What are the primary safety advantages of the two-enzyme method over the traditional three-enzyme process?
A: The traditional three-enzyme method requires D-amino acid oxidase, which necessitates the use of liquid oxygen or pure oxygen for oxidative cleavage, posing significant safety hazards and high power consumption. The novel two-enzyme method eliminates the oxidase step entirely, removing the need for hazardous oxygen handling and drastically improving operational safety.
Q: How does this enzymatic route impact the purity profile of the final D-7-ACA product?
A: By operating under mild conditions (18-25°C) and avoiding harsh chemical solvents like dichloromethane or strong acids used in chemical cracking, the enzymatic route minimizes side reactions and degradation. This results in a superior impurity profile with purity levels consistently exceeding 98%, simplifying downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It utilizes immobilized enzymes which can be reused, and the reaction conditions are mild enough to be managed in standard stainless steel fermentation or lysis tanks without requiring specialized high-pressure or cryogenic equipment, facilitating easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-7-ACA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced biocatalytic processes like the two-enzyme one-step method requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102827912B are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle complex enzymatic reactions with precision, maintaining stringent purity specifications through our rigorous QC labs. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems guarantee that every batch of D-7-ACA meets the highest international standards for potency and impurity profiles.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their antibiotic supply chains. By leveraging our expertise in enzymatic synthesis, we can help you achieve significant cost reduction in antibiotic manufacturing while ensuring a secure and sustainable supply of critical intermediates. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how our advanced production capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
