Advanced Synthesis of 1,4,7,10-Tetraazacyclododecane for Commercial Scale-Up and High-Purity Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for synthesizing complex macrocyclic amines, which serve as critical building blocks for diagnostic agents and therapeutic drugs. Patent CN111808039B introduces a groundbreaking novel method for synthesizing 1,4,7,10-tetraazacyclododecane, a molecule of immense value in biomedicine, molecular recognition, and catalysis. This technology addresses the long-standing challenges associated with traditional synthesis routes by utilizing triethylene tetramine and urea as starting materials, offering a streamlined three-step process that significantly enhances atom utilization. For R&D directors and procurement specialists, this patent represents a pivotal shift towards cleaner, more cost-effective manufacturing protocols that do not compromise on the stringent purity specifications required for medical applications. The ability to achieve a total yield of 74.97 percent with a gas chromatography content of more than 99 percent underscores the robustness of this chemical engineering solution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 1,4,7,10-tetraazacyclododecane has been plagued by inefficient and hazardous chemical processes that hinder scalable commercial production. Traditional disclosures, such as the tosyl group protection-deprotection method, necessitate the use of large quantities of concentrated sulfuric acid, creating significant environmental and safety burdens for manufacturing facilities. Furthermore, methods like the glyoxal or dithiooxamide routes often suffer from poor atom economy and involve high-risk reduction steps that are difficult to control on an industrial scale. These legacy processes frequently result in complex impurity profiles that require extensive and costly purification steps, thereby inflating the overall cost of goods sold. The reliance on such archaic methodologies limits the ability of supply chain heads to guarantee consistent quality and continuity of supply for high-value pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a direct condensation strategy that eliminates the need for hazardous deprotection agents and complex rearrangement steps. By reacting triethylene tetramine with urea to generate ethylene bis-imidazolidinone, the process establishes a robust foundation for subsequent cyclization with high selectivity. This route is characterized by its simplicity and ease of control, allowing for precise management of reaction parameters to minimize by-product formation. The elimination of toxic reagents and the reduction of reaction steps directly translate to substantial cost savings and a reduced environmental footprint, aligning perfectly with modern green chemistry principles. This innovation provides a reliable pharmaceutical intermediate supplier with the capability to offer high-purity products at a more competitive market price point.
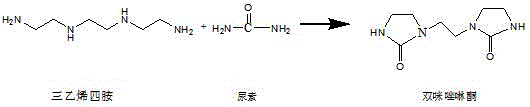
Mechanistic Insights into Urea-Mediated Macrocyclization
The core of this technological breakthrough lies in the precise control of nucleophilic substitution and cyclization mechanisms during the second step of the synthesis. As illustrated in the reaction mechanism, the nitrogen atoms at the 1-position and 10-position of the bis-imidazolidinone exhibit high nucleophilicity, facilitating ring closure with 1,2-dihalogenated ethane under alkaline conditions. The use of non-proton medium polarity solvents like ethyl acetate or toluene, combined with acid-binding agents such as cesium carbonate, is critical for driving the equilibrium towards the target product while suppressing unwanted side reactions. Operating at low temperatures between minus 30°C and 0°C further ensures that the kinetic energy of the system favors the formation of the 12-membered ring over linear polymerization. This meticulous control over the reaction environment is essential for achieving the high molar yields reported in the patent data.
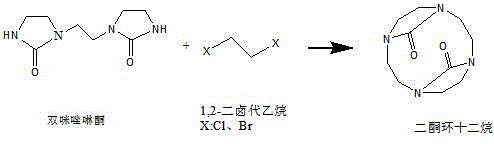
Impurity control is another critical aspect where this novel mechanism excels, particularly in avoiding the formation of polymeric by-products that often plague macrocyclic syntheses. The patent explicitly details how steric hindrance effects can lead to the formation of longer molecular chain polymers if the reaction temperature is too high or if the stoichiometry is incorrect. By maintaining an excess of dihalogenated ethane and strictly controlling the molar ratio of bis-imidazolidinone to dihalogenated ethane, the process effectively mitigates these risks. Additionally, the final hydrolysis step utilizes hydrazine hydrate in an alcohol solvent, a unique choice that prevents the ring-opening decomposition of the target product which is common under strong inorganic alkali conditions. This sophisticated understanding of reaction dynamics ensures that the final product meets the rigorous purity standards demanded by the diagnostic and therapeutic sectors.
How to Synthesize 1,4,7,10-Tetraazacyclododecane Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters, specifically regarding temperature gradients and reagent stoichiometry to ensure reproducibility. The detailed standardized synthesis steps involve a initial solvent-free condensation followed by a low-temperature cyclization and a final hydrazine-mediated hydrolysis, each requiring specific equipment configurations for optimal results. For R&D teams looking to replicate or scale this process, adherence to the specified molar ratios and solvent choices is paramount to avoiding the carbonization and polymerization issues seen in less optimized attempts. The following guide outlines the critical operational phases derived directly from the patent examples to facilitate a smooth technology transfer.
- React triethylene tetramine with urea at 130-200°C to generate ethylene bis-imidazolidinone with high atom utilization.
- Perform condensation with 1,2-dihalogenated ethane at -30°C to 0°C using non-proton solvents to form diketone cyclododecane.
- Hydrolyze the diketone intermediate with hydrazine hydrate in alcohol solvent at 40-70°C to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers transformative advantages for procurement managers and supply chain heads focused on cost reduction in fine chemical manufacturing. The elimination of expensive transition metal catalysts and hazardous deprotection reagents significantly simplifies the raw material procurement process and reduces the complexity of waste treatment protocols. By streamlining the production workflow into just three high-yield steps, manufacturers can drastically reduce the operational time and energy consumption required per kilogram of finished product. This efficiency gain allows for a more agile response to market demand fluctuations, ensuring that supply chain continuity is maintained even during periods of high volatility in the global chemical market. The robust nature of the process also minimizes the risk of batch failures, providing a more predictable and reliable supply of critical intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and readily available starting materials like urea and triethylene tetramine, avoiding the need for costly protecting groups. The high atom utilization rate means that less raw material is wasted as by-products, directly lowering the material cost per unit of production. Furthermore, the simplified purification steps reduce the consumption of solvents and energy associated with extensive chromatography or recrystallization processes. These factors combine to create a highly competitive cost structure that enhances the margin potential for downstream pharmaceutical applications.
- Enhanced Supply Chain Reliability: The use of stable and common reagents ensures that the supply chain is not vulnerable to the shortages often associated with specialized or hazardous chemicals. The robustness of the reaction conditions allows for flexible manufacturing scheduling, reducing the lead time for high-purity chelating agents needed for urgent medical projects. By avoiding complex multi-step protection strategies, the overall production cycle time is shortened, enabling faster turnaround from order to delivery. This reliability is crucial for maintaining the production schedules of partner pharmaceutical companies developing time-sensitive diagnostic or therapeutic agents.
- Scalability and Environmental Compliance: The solvent-free nature of the first step and the use of recoverable organic solvents in subsequent steps align with strict environmental regulations, reducing the burden of hazardous waste disposal. The process is designed for commercial scale-up of complex macrocyclic amines, with reaction conditions that are easily manageable in standard industrial reactors without requiring exotic high-pressure or high-temperature equipment. This scalability ensures that production volumes can be increased from 100 kgs to 100 MT annual commercial production without significant re-engineering of the process. The clean production profile also facilitates easier regulatory approval and compliance with international green chemistry standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived from the specific technical disclosures and experimental data provided in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new route for their supply chain.
Q: What are the limitations of conventional tosyl protection methods for Cyclen synthesis?
A: Conventional methods often require large amounts of concentrated sulfuric acid for deprotection, suffer from poor atom economy, and involve high-risk reduction processes that are difficult to apply to industrial production.
Q: How does the novel urea-based route improve impurity control?
A: By utilizing a specific molar ratio of triethylene tetramine to urea and controlling reaction temperatures, the process minimizes carbonization and polymerization side reactions, achieving >99% GC content.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method features high atom utilization, few reaction steps, and simplicity in control, representing a clean and efficient production route suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,7,10-Tetraazacyclododecane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation medical technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical manufacturing setting. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 1,4,7,10-tetraazacyclododecane meets the exacting standards required for pharmaceutical applications. Our expertise in process optimization allows us to deliver consistent quality while maximizing the efficiency gains offered by this novel synthesis route.
We invite you to collaborate with us to explore how this advanced chemistry can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning efforts, ensuring a seamless integration of this high-value intermediate into your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
