Scalable Industrial Synthesis of 1,4,7,10-Tetraazacyclododecane for High-Purity MRI Contrast Agents
Scalable Industrial Synthesis of 1,4,7,10-Tetraazacyclododecane for High-Purity MRI Contrast Agents
The pharmaceutical and diagnostic imaging sectors rely heavily on the availability of high-purity macrocyclic chelators, specifically 1,4,7,10-tetraazacyclododecane, often referred to as Cyclen. This critical building block serves as the foundational scaffold for gadolinium-based contrast agents like Dotarem® and Prohance®, which are essential for enhancing magnetic resonance imaging (MRI) diagnostics. However, traditional manufacturing routes have long been plagued by complex protection-deprotection sequences and low atom economy. The groundbreaking methodology detailed in patent CN1343203A introduces a transformative approach that bypasses these historical bottlenecks. By leveraging a novel salt isolation strategy and a selective hydrolysis mechanism, this process offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the technical nuances of this patent is vital for securing a sustainable supply chain for next-generation contrast media.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
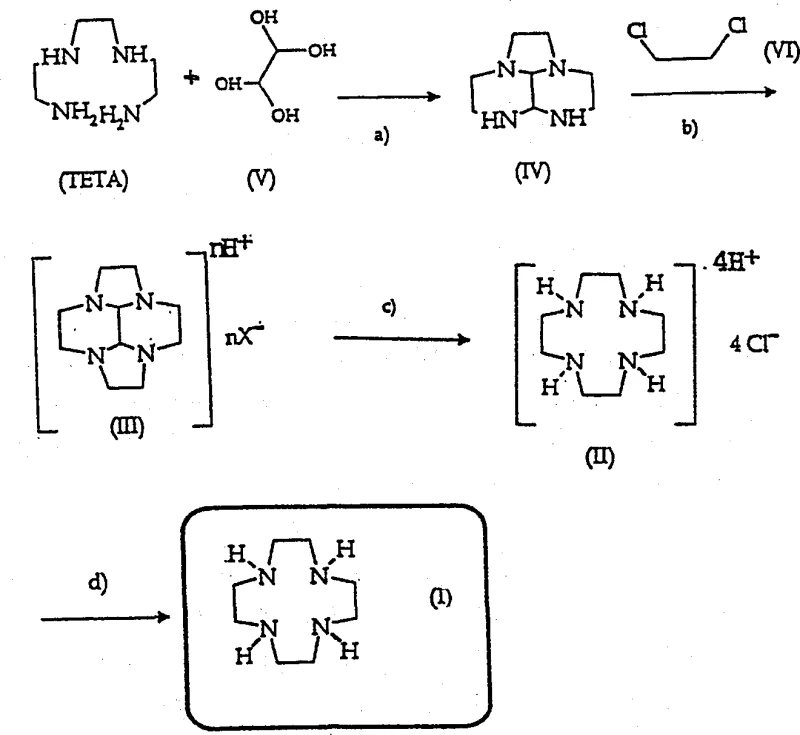
Historically, the industrial production of 1,4,7,10-tetraazacyclododecane has depended on the Richman-Atkins synthesis, a method that, while chemically sound, presents significant logistical and economic challenges for large-scale operations. This classical route necessitates the preparation of amine tosyl derivatives, introducing multiple synthetic steps that increase both material costs and environmental waste. Furthermore, prior art attempts to streamline this process, such as those disclosed in WO97/49691, encountered severe difficulties during the isolation of key intermediates. Specifically, the extraction of the bicyclic intermediate (III) using hexane often resulted in substantial product loss during the concentration of reaction mixtures. These losses were attributed not only to physical transport phenomena but also to chemical degradation caused by parasitic alkylating agents formed as byproducts. Such instability renders the free base form of the intermediate unsuitable for consistent factory-scale application, creating volatility in supply and yield that procurement managers cannot afford.
The Novel Approach
The methodology outlined in CN1343203A fundamentally reengineers the synthesis pathway to overcome these stability and yield issues through a clever manipulation of solubility and salt formation. Instead of struggling with the unstable free base, the process isolates the critical intermediate (III) as a stable inorganic acid salt, specifically using hydrochloric or phosphoric acid. This modification allows the intermediate to precipitate directly from the dipolar aprotic solvent (DMAC) reaction mixture, effectively separating it from parasitic alkylating impurities before they can cause degradation. The subsequent conversion to the final macrocycle utilizes a mild hydrolysis step with a primary diamine, avoiding the harsh oxidative cleavage methods of the past. This streamlined reaction sequence, depicted in the comprehensive process flow below, ensures that cost reduction in MRI contrast agent manufacturing is achieved through both higher yields and simplified downstream processing.
Mechanistic Insights into Salt-Mediated Cyclization and Hydrolysis
The core innovation of this technology lies in the precise control of the reaction environment during the cyclization and deprotection phases. In the cyclization step (step b), the reaction between intermediate (IV) and 1,2-dichloroethane is conducted in dimethylacetamide (DMAC) with sodium carbonate and a catalytic amount of sodium bromide. The addition of acid post-reaction is the critical pivot point; by introducing HCl or H3PO4, the basic nitrogen atoms of the bicyclic structure are protonated. This protonation drastically reduces the solubility of the intermediate in DMAC, causing it to crash out as a solid salt while leaving soluble impurities in the mother liquor. This purification-by-precipitation technique eliminates the need for energy-intensive distillation or chromatography, which are common sources of yield loss in fine chemical synthesis. The use of phosphoric acid is particularly advantageous as it requires less water than hydrochloric acid, facilitating easier solvent recovery and reducing the thermal load on the system.
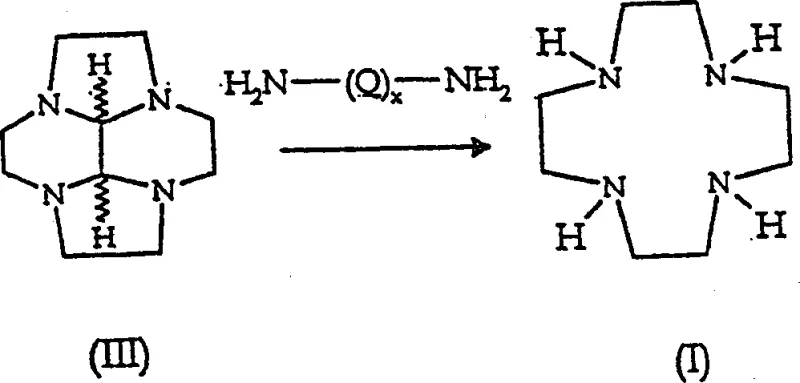
Following isolation, the conversion of the protected intermediate (III) to the target macrocycle (I) proceeds via a transamination mechanism rather than oxidative cleavage. As illustrated in the mechanistic diagram below, the process employs diethylenetriamine (DETA) in an aqueous medium at a controlled pH of 5.5 to 9 and temperatures between 60 to 100°C. The primary amine groups of DETA attack the imine carbons of the glyoxal bridge, irreversibly displacing the protecting group and releasing the free macrocycle. Crucially, the final product is isolated as a tetrahydrochloride salt. This final salt formation step provides a second layer of purification, as the tetrahydrochloride of 1,4,7,10-tetraazacyclododecane has distinct solubility properties compared to the excess DETA and other reaction byproducts. This dual-salt strategy (intermediate salt and final product salt) ensures that high-purity 1,4,7,10-tetraazacyclododecane is obtained with minimal contamination, meeting the stringent purity specifications required for parenteral applications.
How to Synthesize 1,4,7,10-Tetraazacyclododecane Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to maximize the benefits of the salt isolation strategy. The process begins with the condensation of triethylenetetramine (TETA) and glyoxal hydrate at low temperatures (0-5°C) to form the linear precursor, followed by cyclization in DMAC. The critical operational window involves the precise addition of acid to precipitate the intermediate salt, followed by the aqueous hydrolysis with DETA. Operators must maintain the pH between 5 and 9 during hydrolysis to ensure selective deprotection without degrading the macrocyclic ring. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below to assist process engineers in replicating these results.
- Condense Triethylenetetramine (TETA) with glyoxal hydrate at 0-5°C using calcium hydroxide to form intermediate (IV).
- React intermediate (IV) with 1,2-dichloroethane in DMAC with Na2CO3/NaBr to form compound (III), isolating it as a stable phosphate or hydrochloride salt.
- Hydrolyze compound (III) salt with diethylenetriamine (DETA) in water at pH 5-9 and 90-120°C to yield the final tetrahydrochloride salt of 1,4,7,10-tetraazacyclododecane.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this patented synthesis route offers profound strategic advantages beyond mere technical elegance. The elimination of tosyl protection groups removes a significant cost center associated with purchasing specialized sulfonating reagents and managing the resulting sulfonate waste streams. Furthermore, the ability to isolate intermediates as stable salts mitigates the risk of batch-to-batch variability, a common pain point in the supply of complex heterocycles. By stabilizing the intermediate, the process allows for flexible scheduling; the intermediate salt can potentially be stored or transported more safely than the reactive free base, decoupling the cyclization and deprotection steps if necessary. This flexibility enhances supply chain reliability, ensuring that production timelines for downstream API synthesis are not compromised by upstream instability.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the purification workflow. By utilizing precipitation as the primary purification method for the intermediate, the need for extensive solvent exchanges or chromatographic separations is significantly reduced. The recovery of DMAC, a high-boiling solvent, is also optimized because the use of phosphoric acid introduces less water into the system compared to hydrochloric acid, lowering the energy demand for fractional distillation. Additionally, the high selectivity of the DETA hydrolysis step minimizes the formation of difficult-to-remove impurities, reducing the burden on final crystallization steps and increasing the overall mass efficiency of the plant.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as TETA, glyoxal, and 1,2-dichloroethane ensures that raw material sourcing remains robust and unaffected by the supply volatility often seen with specialized reagents. The process operates under relatively mild conditions (temperatures below 150°C and atmospheric pressure), which reduces the stress on reactor equipment and lowers maintenance downtime. This operational simplicity translates to higher plant availability and more consistent lead times for customers. The ability to produce the intermediate as a stable salt also means that inventory can be managed more effectively, buffering against sudden spikes in demand for the final contrast agent precursors.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a significant improvement over legacy methods. Avoiding the use of tosyl chlorides eliminates the generation of sulfonic acid waste, which can be corrosive and difficult to treat. The aqueous nature of the final hydrolysis step aligns with green chemistry principles by reducing the volume of organic solvents required in the later stages of synthesis. The process is inherently scalable, as demonstrated by the successful isolation of multi-kilogram batches in the patent examples, making it suitable for commercial scale-up of complex pharmaceutical intermediates without requiring exotic high-pressure equipment or cryogenic facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on critical process parameters. Understanding these details is essential for technology transfer teams evaluating the feasibility of adopting this route for GMP manufacturing.
Q: Why is salt isolation critical for the industrial scale-up of compound (III)?
A: Isolating compound (III) as a hydrochloride or phosphate salt prevents product loss and chemical degradation associated with parasitic alkylation during solvent concentration, significantly improving yield stability compared to free base extraction.
Q: How does this process improve upon the classical Richman-Atkins synthesis?
A: This method eliminates the need for expensive and environmentally burdensome tosyl protection and deprotection steps, utilizing direct cyclization and aqueous hydrolysis to achieve high purity with reduced waste generation.
Q: What is the role of Diethylenetriamine (DETA) in the final hydrolysis step?
A: DETA acts as a transamination agent that irreversibly displaces the glyoxal protecting group under mild aqueous conditions (pH 5-9), allowing for the selective release of the macrocycle without harsh oxidative cleavage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,7,10-Tetraazacyclododecane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN1343203A process are fully realized in a GMP-compliant environment. We understand that for MRI contrast agents, impurity profiles are critical; therefore, our rigorous QC labs and stringent purity specifications guarantee that every batch of 1,4,7,10-tetraazacyclododecane meets the highest global standards. Our commitment to quality extends beyond simple compliance, as we actively optimize each unit operation to maximize yield and minimize environmental impact.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this salt-isolation route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a stable, cost-effective, and high-quality supply of this critical diagnostic imaging intermediate.
