Revolutionizing Rebamipide Production: A Direct Minisci Approach for Commercial Scale-up
The pharmaceutical landscape for gastrointestinal therapeutics continues to evolve, with Rebamipide standing out as a critical mucosal protective agent. However, the manufacturing of its key intermediates has historically been burdened by inefficient, multi-step synthetic routes. A groundbreaking patent, CN108341775B, discloses a transformative synthetic method for producing the Rebamipide intermediate, 2-amino-3-[2(1H)-quinolone-4]propionic acid. This innovation leverages a direct Minisci-type radical addition reaction, fundamentally shifting the paradigm from laborious construction of the carbon skeleton to a streamlined coupling of readily available building blocks. By reacting aspartic acid directly with 2-hydroxyquinoline under silver-catalyzed oxidative conditions, this method bypasses the need for complex protecting group strategies and lengthy precursor synthesis. For global supply chain leaders and R&D directors, this represents a pivotal opportunity to optimize the production of high-purity pharmaceutical intermediates, ensuring both economic efficiency and robust supply continuity for anti-ulcer medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Rebamipide and its intermediates has relied on classical routes, such as the one reported by Uchida et al., which are characterized by excessive operational complexity and extended production cycles. As illustrated in the traditional pathway below, the synthesis of the critical intermediate (R,S)-2-amino-3-[2(1H)-quinolone-4]propionic acid involves a convoluted sequence starting from diethyl malonate. This legacy process requires oximation, reduction, and acetylation to generate diethyl acetamidomalonate, alongside a separate branch involving the condensation of aniline and acetoacetyl chloride followed by bromination to create 4-bromomethyl quinolone. These two complex fragments must then be coupled, resulting in a racemate that subsequently requires tedious resolution steps—esterification, chiral separation, and hydrolysis—to obtain the active single isomers. This multiplicity of unit operations not only inflates capital expenditure and operational costs but also introduces significant points of failure in the supply chain, leading to longer lead times and potential yield losses at every stage.
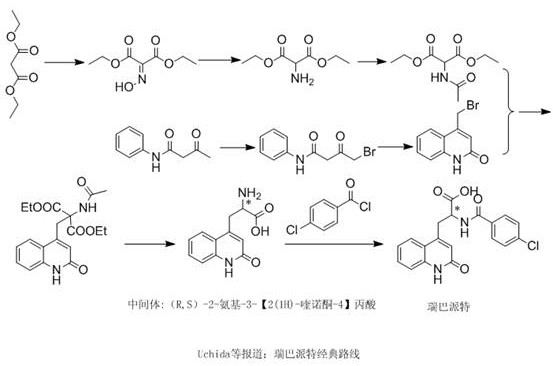
The Novel Approach
In stark contrast to the fragmented legacy workflows, the novel approach detailed in patent CN108341775B introduces a convergent, one-pot strategy that dramatically simplifies the molecular assembly. This method utilizes a Minisci reaction to directly functionalize the electron-deficient C4 position of 2-hydroxyquinoline with a nucleophilic radical derived from aspartic acid. By employing inexpensive and commercially abundant raw materials—aspartic acid and 2-hydroxyquinoline—the process eliminates the need for synthesizing specialized halogenated quinolones or malonate derivatives. The reaction proceeds efficiently in a mixed solvent system of acetonitrile and water, utilizing silver nitrate as a catalyst and persulfate as a terminal oxidant. This direct coupling not only reduces the total number of reaction steps from over ten to essentially a single transformation but also inherently preserves the stereochemical integrity of the starting amino acid, allowing for the direct production of chiral intermediates without the need for downstream resolution.
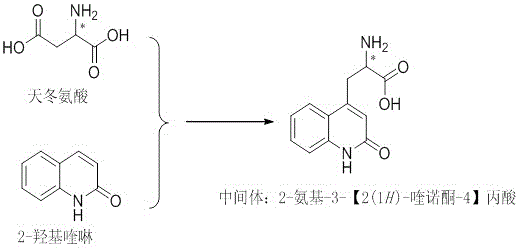
Mechanistic Insights into Silver-Catalyzed Minisci Alkylation
The core of this technological breakthrough lies in the sophisticated application of free-radical chemistry to achieve site-selective C-H functionalization. The mechanism operates through a silver-catalyzed oxidative decarboxylation pathway. In the acidic reaction medium, the amino group of aspartic acid is protonated to form an ammonium salt, which effectively protects it from oxidation by the persulfate oxidant. Simultaneously, the silver catalyst facilitates the generation of sulfate radicals or higher-valent silver species that induce the decarboxylation of the aspartic acid side chain. This generates a nucleophilic carbon-centered radical at the beta-position relative to the amino group. This radical species then selectively attacks the electron-deficient C4 position of the protonated 2-hydroxyquinoline ring, which acts as the heteroaromatic acceptor. The subsequent rearomatization and oxidation steps yield the desired 2-amino-3-[2(1H)-quinolone-4]propionic acid scaffold. This mechanistic elegance allows for the direct use of unprotected amino acids, a feat that is typically challenging due to competing oxidation pathways.
Furthermore, the process demonstrates exceptional chemoselectivity and stereochemical fidelity, which are paramount for R&D teams focused on impurity control. The difference in pKa values between the two carboxyl groups in aspartic acid, combined with the specific reaction conditions, ensures that decarboxylation occurs selectively at the desired position, minimizing the formation of regioisomers or over-oxidized byproducts. When chiral aspartic acid (L- or D-enantiomer) is employed as the starting material, the chirality at the alpha-carbon is retained throughout the radical process, as the radical center is generated at the beta-carbon, distant from the stereocenter. This results in the direct formation of the corresponding (S)- or (R)-intermediate with high optical purity, as evidenced by HPLC content exceeding 99.5% in the patent examples. This inherent ability to control stereochemistry at the source rather than through purification significantly simplifies the impurity profile and reduces the burden on analytical quality control.
How to Synthesize Rebamipide Intermediate Efficiently
The implementation of this Minisci-based synthesis offers a straightforward protocol suitable for immediate scale-up in pilot and commercial plants. The process begins by charging a reactor with aspartic acid, 2-hydroxyquinoline, and a catalytic amount of silver nitrate in a mixture of acetonitrile and water. The mixture is heated to a moderate temperature range of 50-80°C, and an aqueous solution of potassium or sodium persulfate is added dropwise to control the exotherm and radical flux. Following a reaction period of 2 to 5 hours, the workup involves a standard liquid-liquid extraction with ethyl acetate, washing to remove inorganic salts, and crystallization from acetone to isolate the high-purity product. For a detailed, step-by-step standard operating procedure including exact molar ratios and safety protocols, please refer to the technical guide below.
- Prepare a mixed solvent system of acetonitrile and water, adding aspartic acid (L, D, or racemic), 2-hydroxyquinoline, and silver nitrate catalyst.
- Heat the reaction mixture to 50-80°C and slowly add an aqueous solution of alkali metal persulfate (e.g., potassium persulfate) dropwise.
- Maintain reaction temperature for 2-5 hours, then cool, extract with ethyl acetate, wash, dry, and crystallize to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route translates into tangible improvements in cost structure and operational resilience. By collapsing a multi-step linear synthesis into a single convergent step, manufacturers can drastically reduce the consumption of solvents, reagents, and energy associated with intermediate isolations and purifications. The reliance on commodity chemicals like aspartic acid, which is produced on a massive scale for the food and feed industries, ensures a stable and low-cost raw material supply, insulating the production process from the volatility often seen with specialized fine chemical intermediates. Furthermore, the elimination of bromination steps removes the handling hazards and waste disposal costs associated with corrosive brominating agents and hydrogen bromide byproducts, aligning the process with modern green chemistry principles and environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this route is driven primarily by the simplification of the bill of materials and the reduction of processing time. By eliminating the synthesis of diethyl acetamidomalonate and 4-bromomethyl quinolone, the process removes several high-cost unit operations, including low-temperature reactions and vacuum distillations. Additionally, the ability to produce single isomers directly from chiral starting materials obviates the need for expensive chiral resolution agents and the associated yield losses (typically 50% theoretical maximum in racemic resolutions). This structural efficiency leads to substantial cost savings in both raw material procurement and utility consumption, enhancing the overall margin profile of the final API.
- Enhanced Supply Chain Reliability: From a logistics perspective, shortening the synthetic chain significantly de-risks the supply of this critical intermediate. Fewer reaction steps mean fewer opportunities for batch failures, equipment downtime, or quality deviations, resulting in more predictable lead times for downstream API manufacturers. The use of robust, non-cryogenic reaction conditions (50-80°C) allows the process to be run in standard glass-lined or stainless steel reactors without the need for specialized low-temperature infrastructure, increasing the number of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This flexibility ensures continuity of supply even in the face of regional manufacturing disruptions or capacity constraints.
- Scalability and Environmental Compliance: The scalability of the Minisci reaction is supported by its simple workup procedure, which relies on standard extraction and crystallization techniques familiar to plant operators. The aqueous/organic solvent system is manageable and allows for potential solvent recovery loops to further minimize waste. Moreover, the avoidance of heavy metal catalysts (using only catalytic silver which can potentially be recovered) and halogenated reagents simplifies the wastewater treatment profile. This makes the process highly attractive for facilities operating under strict environmental permits, facilitating easier regulatory approval and long-term sustainability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within patent CN108341775B, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: How does this new method improve optical purity compared to traditional resolution?
A: Unlike traditional methods that require synthesizing a racemate and performing complex esterification and resolution steps, this novel Minisci reaction allows for the direct use of chiral starting materials (L- or D-aspartic acid). The chirality is transferred directly to the product, yielding single isomers (S or R) with high optical purity (>99% HPLC content) without the need for downstream resolution.
Q: What are the primary cost drivers eliminated in this synthetic route?
A: This route eliminates the need for expensive and multi-step precursors like diethyl acetamidomalonate and 4-bromomethyl quinolone. By utilizing commodity chemicals such as aspartic acid and 2-hydroxyquinoline, and removing the bromination and malonate synthesis steps, the overall material cost and processing time are significantly reduced.
Q: Is the silver catalyst recoverable in this process?
A: The patent describes the use of silver nitrate as a catalyst in conjunction with persulfate. While the specific recovery protocol is detailed in the full experimental section, the use of heterogeneous or recoverable silver species is a common optimization target in industrial Minisci reactions to further lower heavy metal costs and meet environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rebamipide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Minisci-based synthetic route for the global Rebamipide market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry and silver-catalyzed processes, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international pharmacopeial standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development and commercialization timelines.
We invite you to leverage our technical expertise to optimize your supply chain for Rebamipide and related gastrointestinal therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how this innovative synthesis can drive value for your organization.
