Optimizing Rebamipide Production Advanced Synthesis and Commercial Scalability
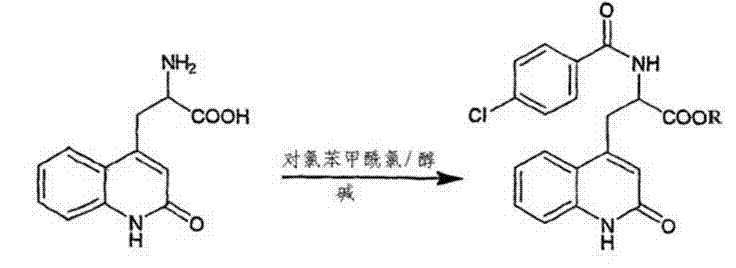
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for gastroprotective agents like Rebamipide. The patent CN103113294A introduces a transformative approach to synthesizing Rebamipide from 2-amino-3-(1,2-dihydro-2-oxo-4-quinolyl) propionic acid hydrochloride, commonly referred to as the amino acid salt. This innovation addresses critical bottlenecks in traditional manufacturing by streamlining the acylation process into a highly efficient one-step reaction. By integrating activated carbon adsorption decoloration under alkaline conditions prior to the main reaction, the method effectively removes color bodies and trace impurities that often plague bulk pharmaceutical production. The result is a finished product with a verified purity of greater than 99.5% and yields ranging consistently from 80% to 95%. For global procurement teams and R&D directors, this represents a significant leap forward in process reliability, ensuring that the reliable pharmaceutical intermediates supplier partners can meet stringent regulatory standards without compromising on throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rebamipide has been fraught with challenges that escalate production costs and complicate supply chain logistics. Conventional methods often involve multi-step sequences that require harsh reaction conditions, leading to the formation of complex impurity profiles that are difficult to separate. These traditional routes frequently suffer from low overall yields, sometimes dropping below acceptable commercial thresholds, which necessitates extensive recycling of materials and increases solvent consumption. Furthermore, the presence of stubborn impurities often requires additional purification steps such as column chromatography or repeated recrystallization, which are not only time-consuming but also environmentally burdensome. The acylation reaction in older processes is particularly sensitive to environmental conditions, demanding precise control that is hard to maintain at scale, resulting in batch-to-batch variability that undermines cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a direct acylation strategy that simplifies the workflow while enhancing product quality. By starting with the amino acid salt and reacting it directly in an alkaline alcohol liquid, the process eliminates unnecessary intermediate isolation steps that typically contribute to yield loss. The strategic use of activated carbon at elevated temperatures (55°C to 70°C) serves as a powerful purification tool early in the process, preventing the carryover of colored impurities into the final crystal lattice. This proactive purification strategy ensures that the subsequent crystallization yields a white crystalline powder of exceptional quality. The method's robustness is evidenced by its ability to maintain high yields even when scaled up, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. This shift from a reactive purification model to a proactive one fundamentally changes the economics of Rebamipide production.
Mechanistic Insights into Acylation and Purification Dynamics
The core of this synthetic breakthrough lies in the precise control of the acylation mechanism between the amino group of the quinolyl propionic acid derivative and p-chlorobenzoyl chloride. Under alkaline conditions, the amino group is deprotonated, increasing its nucleophilicity and facilitating a rapid attack on the carbonyl carbon of the acid chloride. However, the true innovation is the management of the reaction environment to suppress side reactions. By maintaining the reaction temperature between 0°C and 15°C during the addition of the acylating agent, the process minimizes hydrolysis of the acid chloride and prevents over-acylation or polymerization side products. This thermal control is critical for maintaining the integrity of the quinolone ring system, which is susceptible to degradation under harsher conditions. The subsequent standing period allows for the complete conversion of the transition body, ensuring that the reaction equilibrium favors the desired amide bond formation.
Impurity control is further enhanced through the specific pH adjustments and washing protocols described in the later stages of the process. After the formation of the alkali salt transition body, acidification to a pH of 2 to 4 triggers the precipitation of the free acid form of Rebamipide. This pH window is narrow and critical; deviating from it could result in the co-precipitation of unreacted starting materials or soluble impurities. The washing step using hot water (65°C to 70°C) and ethanol is designed to exploit solubility differences, removing residual salts and organic byproducts while keeping the Rebamipide in the solid phase. This rigorous washing protocol is essential for achieving the reported purity levels of over 99.5%, satisfying the rigorous demands of high-purity pharmaceutical intermediates required for final drug formulation.
How to Synthesize Rebamipide Efficiently
Implementing this synthesis route requires strict adherence to the specified parameters to replicate the high yields and purity reported in the patent data. The process begins with the dissolution of the raw amino acid salt in purified water, followed by the addition of an alkali such as sodium hydroxide to create the necessary alkaline environment. The mixture is heated to facilitate the adsorption decoloration step, which is a prerequisite for obtaining a high-quality final product. Once the solution is filtered and cooled, the acylating agent is introduced under controlled stirring to ensure homogeneity and heat dissipation. The detailed standardized synthesis steps see the guide below for exact operational parameters regarding stoichiometry and timing.
- Dissolve 2-amino-3-(1,2-dihydro-2-oxo-4-quinolyl) propionic acid hydrochloride in purified water with alkali at 55-70°C and perform adsorption decoloration using activated carbon.
- Cool the filtrate to 0-15°C and react with a mixed solution of p-chlorobenzoyl chloride and alcohol for 1.5 to 2.5 hours to form the transition body.
- Acidify the transition body solution to pH 2-4 at 18-22°C, crystallize, filter, and wash with hot water and ethanol to obtain pure Rebamipide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic method translates directly into tangible operational benefits and risk mitigation. The simplification of the process flow reduces the number of unit operations required, which in turn lowers the capital expenditure needed for equipment and the operational expenditure related to labor and energy. By eliminating the need for complex purification technologies like preparative HPLC or extensive chromatography, the manufacturing footprint is reduced, allowing for higher throughput within existing facilities. This efficiency gain is crucial for maintaining competitive pricing in the global market for gastroprotective drugs. Furthermore, the high yield and purity reduce the volume of waste generated per kilogram of product, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of solvent usage significantly lower the raw material costs associated with production. By achieving high conversion rates in a single step, the process minimizes the loss of expensive starting materials, thereby optimizing the overall cost of goods sold. The simplified workup procedure also reduces the consumption of utilities such as steam for distillation and electricity for extended stirring, contributing to substantial cost savings. Additionally, the high purity of the crude product reduces the need for reprocessing, ensuring that resources are utilized efficiently throughout the manufacturing cycle.
- Enhanced Supply Chain Reliability: The use of readily available raw materials such as p-chlorobenzoyl chloride and common alkalis ensures that the supply chain is not vulnerable to shortages of exotic reagents. The robustness of the reaction conditions means that production can be maintained consistently without frequent stoppages for equipment cleaning or process troubleshooting. This reliability is essential for meeting tight delivery schedules and maintaining inventory levels for downstream formulators. The ability to scale from laboratory to industrial reactors without significant process redesign further secures the supply continuity for long-term contracts.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from small laboratory batches to 300-liter reactors, indicating strong potential for multi-ton annual production. The reduced generation of hazardous waste and the use of less toxic solvents simplify the wastewater treatment process, lowering the environmental compliance burden. This eco-friendly profile not only reduces disposal costs but also enhances the corporate social responsibility standing of the manufacturer. The streamlined nature of the process facilitates easier validation and regulatory approval, accelerating the time to market for new generic formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Rebamipide using this advanced methodology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this supply source into your existing procurement strategy.
Q: What is the primary advantage of this Rebamipide synthesis method?
A: The method utilizes a one-step reaction from amino acid salts with activated carbon decoloration, achieving yields of 80-95% and purity exceeding 99.5%, significantly reducing impurity removal costs.
Q: How does the process control environmental pollution?
A: By optimizing reaction conditions and utilizing efficient filtration and washing steps, the process minimizes waste generation and avoids the use of harsh solvents typically found in conventional multi-step syntheses.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the patent examples demonstrate successful scaling from 500mL laboratory beakers to 300L glass-lined reactors, confirming the robustness and reproducibility required for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rebamipide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and scalable production in the pharmaceutical sector. Our expertise as a CDMO partner allows us to adapt complex synthetic pathways like the one described in CN103113294A to meet the specific needs of our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with precision and timeliness. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Rebamipide meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and profitability.
