Advanced Synthesis of Edaravone Glucuronide for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Edaravone Glucuronide for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for active metabolites to support pharmacokinetic profiling and drug development. Patent CN101747390B introduces a transformative methodology for the synthesis of Edaravone metabolites, specifically the glucuronide conjugate, which addresses critical bottlenecks in previous manufacturing protocols. This technical disclosure outlines a refined chemical sequence that not only shortens the overall synthetic route but also dramatically enhances the yield and purity of the target compound. For R&D directors and process chemists, this represents a pivotal shift from labor-intensive, low-efficiency methods to a streamlined, scalable protocol suitable for commercial production. The innovation lies in the strategic selection of intermediates and the optimization of reaction conditions, particularly in the final deprotection stage, ensuring that the final product meets stringent quality specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of Edaravone glucuronides was plagued by significant inefficiencies that hindered large-scale production. As noted in the background technology of the patent, existing methods, particularly those derived from early Japanese research, suffered from notoriously low productivity and a proliferation of unwanted by-products. The conventional routes often resulted in a final step yield of merely 32.5%, with the resulting crude product exhibiting a purity of only 77.4%. Such poor metrics necessitate extensive and costly purification processes, such as repeated chromatography or recrystallization, which are impractical for industrial scale-up. Furthermore, the accumulation of impurities complicates the regulatory approval process for drug substances, as impurity profiles must be tightly controlled. The reliance on harsh conditions or non-selective reagents in older methodologies often led to the degradation of the sensitive pyrazolone ring or the sugar moiety, further diminishing the overall mass balance and economic viability of the process.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient four-step sequence starting from glucuronic acid methyl ester. This methodology systematically constructs the target molecule through well-defined intermediates: the peracetylated ester (I), the glycosyl bromide (II), and the protected conjugate (III), before arriving at the final metabolite (IV). By optimizing the glycosylation promoter and the hydrolysis conditions, this route achieves a remarkable improvement in performance. The visual representation of this streamlined pathway highlights the logical progression from simple starting materials to the complex conjugate.
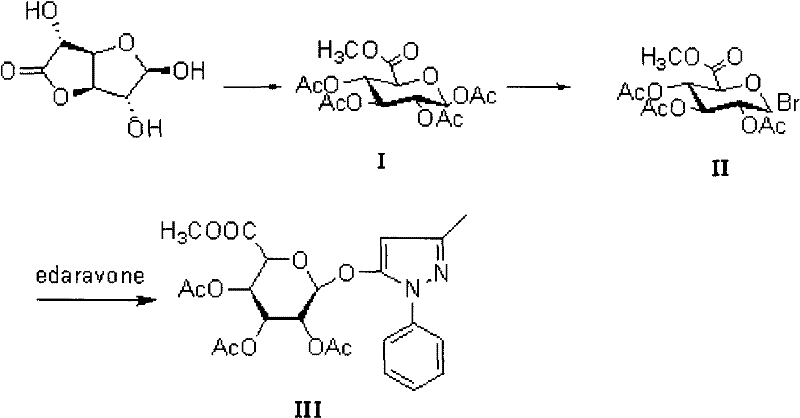
This comprehensive route allows for better control over stereochemistry and functional group compatibility. The use of specific solvents like acetone in the final step, combined with precise temperature control, ensures that the delicate ester and glycosidic bonds are managed effectively, preventing side reactions that typically plague carbohydrate chemistry. This structural clarity and operational simplicity make the novel approach a superior choice for reliable pharmaceutical intermediate supplier operations aiming for high throughput.
Mechanistic Insights into AgOTf-Promoted Glycosylation and Alkaline Hydrolysis
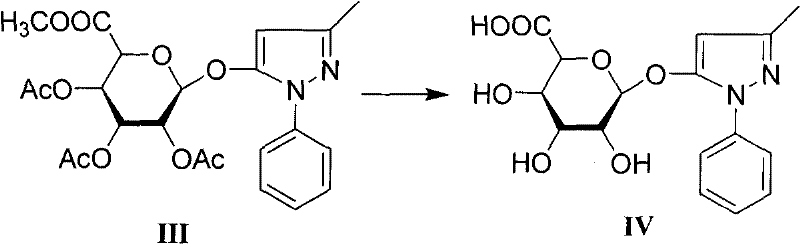
The core of this synthetic success lies in the mechanistic precision of the glycosylation and subsequent deprotection steps. The formation of Intermediate III involves the coupling of the glycosyl bromide (II) with Edaravone. This reaction is facilitated by silver trifluoromethanesulfonate (AgOTf), a potent Lewis acid promoter that activates the anomeric bromide for nucleophilic attack by the hydroxyl group of Edaravone. The presence of molecular sieves in the reaction mixture is crucial, as they sequester trace water that could otherwise hydrolyze the reactive bromide intermediate, thereby driving the equilibrium towards the desired glycoside formation. This step is conducted in dichloromethane at room temperature, providing a mild environment that preserves the integrity of the 3-methyl-1-phenyl-2-pyrazolin-5-one scaffold while ensuring high conversion rates.
Following the coupling, the transformation of Intermediate III to the final product IV involves a carefully controlled alkaline hydrolysis. This step removes the acetyl protecting groups and hydrolyzes the methyl ester to the free carboxylic acid without cleaving the glycosidic bond. The patent specifies the use of sodium hydroxide in a mixed solvent system of organic solvent S (preferably acetone) and water at low temperatures ranging from -5 to 0°C. This低温 condition is vital; it suppresses the potential for base-catalyzed degradation of the pyrazolone ring or epimerization at the anomeric center. The reaction proceeds rapidly, typically completing within 30 to 40 minutes, after which the pH is carefully adjusted to 4-6.5 using hydrochloric acid to precipitate or isolate the zwitterionic product. This precise control over pH and temperature is what enables the achievement of purities exceeding 98.7%, as confirmed by HPLC analysis.
How to Synthesize Edaravone Glucuronide Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of the peracetylated sugar donor, followed by bromination to create the reactive glycosyl halide. The coupling with Edaravone must be performed under anhydrous conditions to prevent premature hydrolysis. Finally, the global deprotection is executed under mild alkaline conditions. The detailed standardized synthetic steps for this high-efficiency route are outlined below, providing a clear roadmap for laboratory and pilot plant execution.
- Prepare peracetylated glucuronic acid methyl ester (Intermediate I) via acetylation of glucuronic acid methyl ester using acetic anhydride and perchloric acid.
- Convert Intermediate I to the glycosyl bromide (Intermediate II) using red phosphorus and liquid bromine in glacial acetic acid under inert atmosphere.
- Couple Intermediate II with Edaravone using silver trifluoromethanesulfonate (AgOTf) promoter to form the protected metabolite (Intermediate III).
- Perform final deprotection by hydrolyzing Intermediate III with sodium hydroxide in aqueous acetone at low temperature (-5 to 0°C) to yield the target metabolite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers substantial strategic benefits beyond mere technical superiority. The drastic improvement in yield and purity directly translates to a more resilient and cost-effective supply chain. By eliminating the need for complex, multi-stage purification procedures associated with lower-yielding legacy methods, manufacturers can significantly reduce processing time and resource consumption. This efficiency gain is critical in the competitive landscape of API intermediate manufacturing, where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The most immediate impact is seen in the reduction of raw material costs per kilogram of finished product. With the final step yield jumping from roughly 32% to over 88%, the effective consumption of expensive starting materials like Edaravone and specialized sugar derivatives is cut by more than half. Furthermore, the use of common, inexpensive solvents such as acetone and methanol, rather than exotic or highly regulated solvents, lowers the overall operational expenditure. The simplified workup procedure, which involves basic filtration and pH adjustment rather than column chromatography, reduces labor costs and equipment downtime, leading to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent supply of high-quality intermediates. The starting materials, including glucuronic acid methyl ester and Edaravone, are commercially available from multiple global sources, mitigating the risk of single-source dependency. The mild reaction conditions (-5 to 25°C) mean that the process does not require specialized high-pressure or cryogenic equipment, making it easier to replicate across different manufacturing sites. This flexibility enhances supply continuity, allowing partners to scale production up or down in response to market demand without facing significant technical barriers or long lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is designed for scalability. The avoidance of heavy metal catalysts in the final steps and the use of recoverable solvents align with modern green chemistry principles. The waste stream is primarily composed of aqueous salts and organic solvents that can be treated using standard effluent management systems. The high purity of the crude product reduces the volume of solvent waste generated during purification, lowering the environmental footprint. This compliance with environmental standards facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste disposal, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the reaction mechanism, scalability, and quality control measures inherent to this patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation.
Q: What is the primary advantage of the new synthesis route for Edaravone metabolites compared to prior art?
A: The new route described in patent CN101747390B significantly improves the yield of the final step from 32.5% to over 88%, while increasing purity from 77.4% to nearly 99%, drastically reducing downstream purification costs.
Q: Which catalyst is critical for the glycosylation step in this process?
A: Silver trifluoromethanesulfonate (AgOTf) is used as the promoter in dichloromethane to facilitate the coupling between the glycosyl bromide intermediate and Edaravone, ensuring high stereoselectivity and conversion.
Q: How does the process ensure environmental compliance and safety during scale-up?
A: The process utilizes mild reaction temperatures ranging from -5°C to 25°C and employs common organic solvents like acetone and methanol, which simplifies solvent recovery and waste treatment compared to more hazardous alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Metabolite Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality metabolites play in the drug development lifecycle. Our team of expert chemists has extensively analyzed the pathway described in CN101747390B and is fully equipped to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Edaravone metabolite delivered meets the highest international standards for pharmaceutical research and development.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and cost-efficiency in the competitive global market.
