Advanced Synthesis of Edaravone Glucuronide: A Breakthrough in Metabolic Intermediate Manufacturing
Advanced Synthesis of Edaravone Glucuronide: A Breakthrough in Metabolic Intermediate Manufacturing
The pharmaceutical industry's relentless pursuit of understanding drug metabolism has placed a premium on the availability of high-quality metabolite standards. Patent CN101747390A introduces a transformative synthesis process for the Edaravone metabolite, specifically the glucuronide conjugate, which is critical for pharmacokinetic and toxicological profiling of the potent free-radical scavenger Edaravone. This technical disclosure addresses a long-standing bottleneck in the field: the inability to produce sufficient quantities of the metabolite with the requisite purity for rigorous analytical validation. By re-engineering the deprotection strategy and optimizing the glycosylation sequence, this methodology offers a robust pathway that significantly outperforms historical precedents. For R&D directors and procurement specialists alike, this represents a pivotal shift from a fragile, low-yield academic curiosity to a viable, industrial-grade manufacturing route. The implications for supply chain stability and cost efficiency in the production of complex pharmaceutical intermediates are profound, ensuring that critical metabolic standards are no longer a limiting factor in drug development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Edaravone glucuronide has been plagued by inefficiencies that render it commercially unviable for large-scale applications. Prior art, including methods explored by Japanese researchers, relied on synthetic routes that were not only cumbersome but also chemically brutal on the sensitive pyrazolone scaffold. The most glaring deficiency in these conventional approaches was the catastrophic loss of material during the final deprotection stage. Literature data indicates that traditional hydrolysis methods resulted in a dismal yield of merely 32.5% for the final step, accompanied by a purity profile of only 77.4%. Such poor performance is unacceptable for a reliable pharmaceutical intermediate supplier, as it necessitates extensive and costly purification protocols to remove a myriad of by-products generated under harsh reaction conditions. Furthermore, the accumulation of impurities complicates the downstream analytical characterization, potentially leading to false positives in metabolic assays. The reliance on aggressive reagents and non-selective cleavage conditions in older methods often led to the degradation of the aglycone, further depressing the overall mass balance and inflating the cost of goods sold.
The Novel Approach
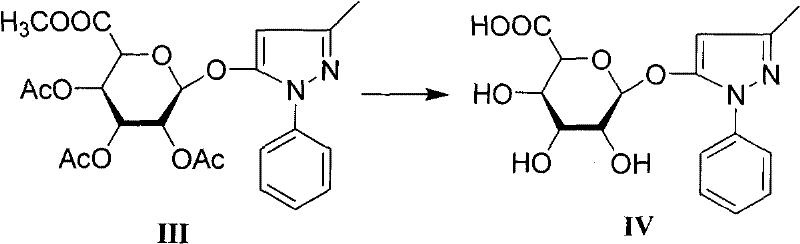
In stark contrast, the methodology disclosed in CN101747390A employs a sophisticated, mild alkaline hydrolysis strategy that preserves the integrity of the target molecule while efficiently stripping away protecting groups. The core innovation lies in the specific solvent system and temperature control utilized during the conversion of Intermediate III to the final Product IV. By dissolving the protected precursor in a mixture of organic solvent (preferably acetone) and water, and maintaining the reaction temperature between -5°C and 10°C, the process achieves a near-quantitative transformation. This gentle approach prevents the base-catalyzed degradation of the pyrazolone ring, a common failure mode in previous attempts. The result is a dramatic escalation in process efficiency, with the final step yield soaring to between 88.7% and 93.3%, and HPLC purity consistently exceeding 98.7%. This leap in performance transforms the economic model of producing this metabolite, turning a loss-leader synthesis into a high-value, efficient operation.
Mechanistic Insights into Silver-Promoted Glycosylation and Selective Deprotection
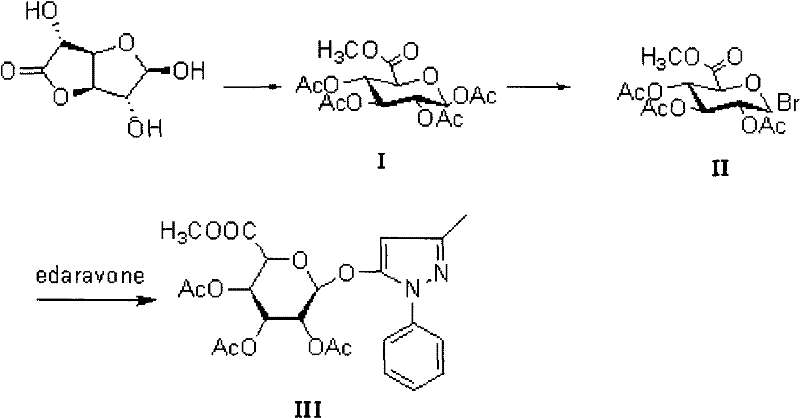
The success of this synthetic route is anchored in the precise orchestration of the glycosylation event and the subsequent selective deprotection. The formation of the glycosidic bond between the sugar moiety and the Edaravone aglycone is facilitated by the use of silver trifluoromethanesulfonate (AgOTf) as a promoter. Unlike traditional heavy metal promoters that might coordinate indiscriminately, AgOTf acts as a soft Lewis acid, effectively activating the glycosyl bromide (Intermediate II) towards nucleophilic attack by the hydroxyl group of Edaravone. This activation occurs under mild conditions in dichloromethane, avoiding the thermal stress that could trigger tautomerization or decomposition of the 3-methyl-1-phenyl-2-pyrazolin-5-one core. The inclusion of molecular sieves in the reaction matrix is a critical mechanistic detail, as it sequesters trace moisture that could otherwise hydrolyze the reactive glycosyl bromide prematurely, leading to the formation of hemiacetal by-products. This attention to anhydrous conditions ensures that the coupling proceeds with high stereocontrol and minimal waste.
Following the coupling, the removal of the acetyl protecting groups is executed with surgical precision. The mechanism involves a nucleophilic acyl substitution where the hydroxide ion attacks the carbonyl carbon of the acetate esters. By conducting this in a biphasic or homogeneous aqueous-acetone system at sub-zero temperatures, the reaction kinetics are tuned to favor ester hydrolysis over amide hydrolysis or ring opening. The choice of acetone as the co-solvent is particularly astute; it ensures the solubility of the lipophilic intermediate while remaining miscible with the aqueous base, creating a uniform reaction environment that prevents localized hot-spots of high pH. This uniformity is key to suppressing side reactions. Once the deprotection is complete, the workup involves a careful pH adjustment to the isoelectric point range (pH 4-6.5), allowing for the crystallization or precipitation of the target glucuronide while leaving ionic impurities in the aqueous phase. This elegant simplicity in the workup procedure underscores the industrial viability of the process.
How to Synthesize Edaravone Glucuronide Efficiently
The execution of this synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and solvent quality to replicate the high yields reported in the patent. The process begins with the preparation of the peracetylated sugar donor, followed by its conversion to the reactive bromide, and culminates in the coupling and deprotection sequence. Operators must ensure that the silver promoter is fresh and that the molecular sieves are activated to guarantee the success of the glycosylation step. The final hydrolysis step demands precise temperature monitoring to maintain the reaction window between -5°C and 0°C, as deviations can impact the purity profile. For a detailed, step-by-step breakdown of the reagent quantities, reaction times, and isolation procedures, please refer to the standardized protocol below.
- Preparation of peracetylated glucuronic acid methyl ester (Intermediate I) via acetylation of glucuronic acid methyl ester using acetic anhydride and perchloric acid catalyst.
- Conversion of Intermediate I to glycosyl bromide (Intermediate II) using red phosphorus and liquid bromine in glacial acetic acid under inert atmosphere.
- Coupling of Intermediate II with Edaravone using silver trifluoromethanesulfonate (AgOTf) promoter in dichloromethane to form the protected glycoside (Intermediate III).
- Final deprotection of Intermediate III using sodium hydroxide in aqueous acetone at low temperature (-5 to 0°C) to yield high-purity Edaravone Glucuronide (Compound IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible operational benefits and risk mitigation. The primary advantage lies in the drastic improvement in mass efficiency. By elevating the yield of the final step from a historical low of roughly one-third to over ninety percent, the amount of starting material required to produce a kilogram of the final metabolite is reduced by a factor of three. This fundamental shift in atom economy drastically lowers the raw material costs and reduces the volume of chemical waste generated, aligning with modern green chemistry principles and reducing disposal costs. Furthermore, the high purity achieved (>98%) minimizes the need for resource-intensive recrystallization or chromatographic purification, shortening the production cycle time and freeing up reactor capacity for other campaigns. This efficiency makes the compound a much more attractive candidate for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents in favor of common solvents like acetone and methanol significantly lowers the variable cost of production. The high yield means that the fixed costs associated with labor, energy, and equipment depreciation are amortized over a much larger output of saleable product. Additionally, the avoidance of complex purification steps reduces the consumption of silica gel and eluents, which are often significant hidden costs in fine chemical manufacturing. The overall process intensity is lower, allowing for a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is paramount for regulatory compliance in the pharmaceutical sector. The use of stable, commercially available starting materials like glucuronic acid methyl ester and Edaravone mitigates the risk of supply disruptions associated with exotic or custom-synthesized precursors. The mild reaction conditions also reduce the safety risks associated with exothermic runaways, facilitating smoother operations in multipurpose plants and ensuring uninterrupted delivery schedules for clients relying on these intermediates for critical toxicology studies.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with parameters that translate well from gram-scale laboratory experiments to multi-kilogram pilot runs. The solvent system is amenable to recovery and recycling, further enhancing the sustainability profile of the manufacture. The reduction in by-product formation simplifies the effluent treatment process, lowering the environmental burden and ensuring compliance with increasingly stringent waste discharge regulations. This makes the technology future-proof against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Edaravone metabolites. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their own research or supply chains.
Q: How does this new synthesis route improve upon previous methods for Edaravone metabolites?
A: Previous methods, particularly those reported in earlier literature, suffered from critically low yields in the final deprotection step (around 32.5%) and poor purity (77.4%). This novel process utilizes a mild alkaline hydrolysis strategy in aqueous acetone at controlled low temperatures, boosting the final step yield to over 90% and achieving purity levels exceeding 99%.
Q: What are the critical reaction conditions for the glycosylation step?
A: The glycosylation coupling between the glycosyl bromide and Edaravone requires a silver promoter, specifically silver trifluoromethanesulfonate (AgOTf), in anhydrous dichloromethane. The presence of molecular sieves is crucial to scavenge trace moisture, and the reaction proceeds efficiently at room temperature, minimizing thermal degradation of the sensitive pyrazolone ring.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It avoids cryogenic conditions (using only mild cooling to -5°C) and utilizes common, recoverable solvents like acetone and dichloromethane. The high yield and robustness of the reaction steps make it suitable for multi-kilogram to ton-scale manufacturing required for clinical supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Metabolite Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-fidelity metabolites is the cornerstone of successful drug development. Our technical team has thoroughly analyzed the synthetic route described in CN101747390A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of Edaravone Glucuronide we deliver meets the exacting standards required for global regulatory submissions. Our commitment to quality assurance means that we do not just supply chemicals; we provide the data integrity necessary to support your IND filings.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for this critical intermediate. Whether you require a Customized Cost-Saving Analysis for your specific volume requirements or need to review specific COA data and route feasibility assessments, we are ready to assist. Let us handle the complexities of synthesis so you can focus on advancing your therapeutic candidates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
