Advanced Acridone Derivatives: Scalable Synthesis for Next-Generation OLED Materials
Advanced Acridone Derivatives: Scalable Synthesis for Next-Generation OLED Materials
The rapid evolution of the optoelectronic industry demands materials that offer superior luminous efficiency, stability, and color purity. In this context, patent CN103694173A introduces a significant breakthrough with a novel class of acridone derivatives designed specifically for high-performance organic luminescent applications. These compounds feature a unique structural architecture where the central acridone core is functionalized with bulky triphenylamine-biphenyl substituents, creating an extended pi-conjugated system that drastically enhances fluorescence properties. The patent details a robust synthetic methodology that overcomes traditional limitations in yield and purity, making these materials highly attractive for commercial adoption in the display and lighting sectors. By integrating rigid planar structures with electron-donating and electron-withdrawing groups, these derivatives exhibit strong intramolecular charge transfer characteristics, resulting in intense fluorescent emission suitable for next-generation OLED devices.
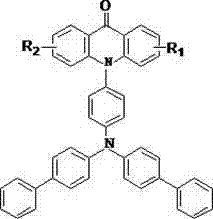
The strategic design of these molecules addresses the critical need for materials with high quantum yields and thermal stability. Unlike conventional acridone dyes which may suffer from aggregation-caused quenching or limited solubility, the incorporation of the biphenyl-triphenylamine moiety not only extends the conjugation length but also improves the morphological stability of the resulting films. This structural modification ensures that the materials can be processed into uniform layers, a prerequisite for high-efficiency light-emitting diodes. Furthermore, the versatility of the synthesis allows for the modulation of emission wavelengths through simple variations in the R1 and R2 substituents, offering a tunable platform for developers seeking specific color coordinates in their device architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex acridone derivatives has been plagued by inefficient coupling reactions and harsh reaction conditions that limit scalability. Traditional methods often rely on prolonged heating under reflux in high-boiling solvents, leading to significant energy consumption and the formation of unwanted by-products that complicate downstream purification. The construction of the C-N bond between the acridone nitrogen and bulky aryl groups typically requires stoichiometric amounts of copper salts or expensive palladium catalysts under inert atmospheres for extended periods, resulting in suboptimal yields often below 60%. Additionally, the removal of residual metal catalysts from the final product is a persistent challenge, particularly for electronic grade materials where trace metal impurities can severely degrade device performance and lifespan. These inefficiencies create bottlenecks in the supply chain, driving up costs and extending lead times for manufacturers.
The Novel Approach
The methodology outlined in patent CN103694173A presents a streamlined three-step process that significantly enhances operational efficiency and product quality. A key innovation is the use of a magnetic CuFe2O4 catalyst in the initial step, which facilitates the formation of the diphenylamine intermediate under milder conditions while allowing for easy magnetic separation, thereby reducing waste and purification complexity. The subsequent cyclization step utilizes p-toluenesulfonic acid in toluene to efficiently close the acridone ring with high regioselectivity. Most notably, the final coupling reaction employs microwave irradiation to accelerate the palladium-catalyzed amination, reducing reaction times from hours to minutes while maintaining excellent yields exceeding 70%. This modern approach not only improves the economic viability of the process but also ensures the production of high-purity intermediates essential for advanced electronic applications.
Mechanistic Insights into Microwave-Assisted Pd-Catalyzed Coupling
The core of this synthetic strategy lies in the efficient construction of the C-N bond via a Buchwald-Hartwig amination mechanism, facilitated by the Pd(dppf)Cl2 catalyst system. In this transformation, the palladium catalyst undergoes oxidative addition with the aryl bromide species, forming a reactive Pd(II) complex that is crucial for the subsequent transmetallation step. The presence of the bulky dppf ligand stabilizes the palladium center and promotes the reductive elimination step, which releases the coupled product and regenerates the active Pd(0) species. The application of microwave irradiation provides rapid and uniform heating, which increases the kinetic energy of the molecules and accelerates the rate-determining steps of the catalytic cycle. This results in a dramatic reduction in reaction time compared to conventional thermal heating, while simultaneously suppressing side reactions such as homocoupling of the aryl halide, thus preserving the integrity of the sensitive acridone core.
Furthermore, the structural rigidity imparted by the acridone scaffold plays a pivotal role in the photophysical properties of the final compound. The planar nature of the acridone ring system allows for effective overlap of p-orbitals, extending the delocalization of pi-electrons across the entire molecular framework. When excited, these electrons transition from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO), and the extended conjugation lowers the energy gap, leading to emission in the visible region with high intensity. The electron-withdrawing carbonyl group on the acridone ring works in concert with the electron-donating triphenylamine units to create an internal push-pull system, which enhances the dipole moment and improves the fluorescence quantum yield. This precise control over electronic structure ensures that the materials exhibit the desired luminescent characteristics required for high-performance optoelectronic devices.
How to Synthesize High-Fluorescence Acridone Derivatives Efficiently
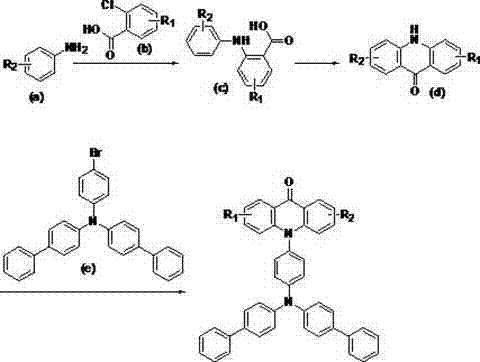
The synthesis of these advanced materials follows a logical progression from simple aniline precursors to the final functionalized acridone derivative. The process begins with the condensation of substituted anilines and chlorobenzoic acids to form the diphenylamine intermediate, followed by an acid-catalyzed cyclization to generate the acridone core. The final step involves the coupling of this core with a brominated triphenylamine derivative under microwave conditions. This standardized protocol ensures reproducibility and high purity, making it suitable for both laboratory research and pilot-scale production. For detailed operational parameters, safety precautions, and specific workup procedures, please refer to the comprehensive guide below which outlines the exact stoichiometry and conditions required for optimal results.
- Synthesize the acridone core intermediate using a magnetic CuFe2O4 catalyst in DMSO at elevated temperatures.
- Perform acid-catalyzed cyclization in toluene with p-toluenesulfonic acid to form the rigid acridone scaffold.
- Execute a palladium-catalyzed Buchwald-Hartwig amination under microwave irradiation to attach the triphenylamine-biphenyl moiety.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost efficiency and supply reliability. The elimination of difficult-to-remove heavy metal contaminants through the use of magnetic catalysts and efficient purification steps reduces the burden on quality control laboratories and minimizes the risk of batch rejection. This streamlined process translates directly into lower manufacturing costs and faster turnaround times, enabling suppliers to respond more agilely to market demands. Moreover, the use of commercially available starting materials such as substituted anilines and bromo-triphenylamines ensures a stable supply chain that is not dependent on exotic or scarce reagents, mitigating the risk of supply disruptions.
- Cost Reduction in Manufacturing: The implementation of microwave-assisted synthesis significantly reduces energy consumption by shortening reaction times from several hours to mere minutes. Additionally, the high yields reported in the patent, consistently exceeding 70%, mean that less raw material is wasted per unit of product produced. The ability to recover and reuse the magnetic catalyst in the first step further contributes to cost savings by reducing the consumption of expensive catalytic materials. These factors combined result in a substantially lower cost of goods sold, allowing for more competitive pricing strategies in the global market for electronic chemicals.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route, characterized by its tolerance to various functional groups and mild reaction conditions, ensures consistent production output. The reliance on commodity chemicals for the starting materials reduces dependency on single-source suppliers, thereby diversifying the supply base and enhancing resilience against geopolitical or logistical shocks. Furthermore, the simplified purification process reduces the complexity of the manufacturing workflow, decreasing the likelihood of production delays caused by equipment bottlenecks or lengthy purification cycles.
- Scalability and Environmental Compliance: The process is inherently scalable, as the microwave technology and magnetic separation techniques can be adapted for larger reactor volumes without compromising reaction efficiency. The reduction in solvent usage and the minimization of hazardous waste generation align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. This green chemistry approach not only safeguards the environment but also enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious partners and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these acridone derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these materials into your specific product development pipelines.
Q: What are the primary applications of these novel acridone derivatives?
A: These compounds are specifically designed for use as organic luminescent materials in optoelectronic devices, such as OLED displays and fluorescent markers, due to their extended pi-conjugated systems and high quantum yields.
Q: How does the synthesis method ensure high product purity?
A: The process utilizes specific purification steps including silica gel column chromatography and precise pH regulation during intermediate isolation, consistently achieving purity levels above 99% as verified by HPLC and elemental analysis.
Q: Is the synthetic route suitable for large-scale industrial production?
A: Yes, the method employs robust reagents and microwave acceleration which simplifies scale-up. The use of a magnetic catalyst in the first step also facilitates easier separation, enhancing operational efficiency for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acridone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of advanced electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the optoelectronic industry. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex synthetic routes, such as the microwave-assisted coupling described in patent CN103694173A, positions us as a strategic partner for companies seeking to innovate in the OLED and display sectors.
We invite you to collaborate with us to explore the potential of these novel acridone derivatives in your applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your time-to-market while optimizing your supply chain costs.
