Scalable Production of High-Purity Near-Infrared Squaraine Dyes with Tunable Thioether Chains
Scalable Production of High-Purity Near-Infrared Squaraine Dyes with Tunable Thioether Chains
The landscape of functional dye manufacturing is undergoing a significant transformation driven by the demand for highly stable, near-infrared (NIR) active materials capable of precise molecular recognition. Patent CN103613948A introduces a groundbreaking class of squaraine dyes featuring flexible thioether chains, representing a major leap forward in the design of chemical sensors and optical probes. Unlike traditional rigid scaffolds, these novel structures leverage the unique electronic and steric properties of sulfur linkages to achieve exceptional stability and tunable optical responses. The core innovation lies in the strategic placement of thioether bridges between aniline donor units, creating a robust donor-acceptor-donor (D-A-D) system that exhibits narrow, intense absorption bands in the 630-650 nm range. This technological advancement not only solves longstanding issues regarding solvent sensitivity and aggregation but also opens new avenues for the rapid, sensitive detection of low-concentration analytes such as calcium and mercury ions.
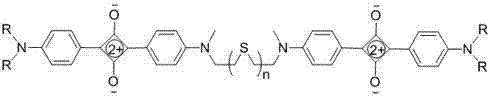
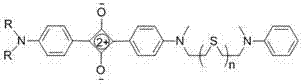
For procurement specialists and supply chain managers seeking a reliable near-infrared squaraine dye supplier, this patent outlines a synthesis pathway that is remarkably amenable to commercial scale-up. The process eliminates the need for complex transition metal catalysts often required in cross-coupling reactions, relying instead on straightforward condensation chemistry between readily available dianiline derivatives and semisquaric acid. By utilizing n-heptanol as a solvent under reduced pressure reflux, the reaction efficiently removes water byproduct, driving the equilibrium towards high yields without the need for expensive dehydrating agents. This simplicity translates directly into cost reduction in functional dye manufacturing, as it minimizes raw material costs and simplifies downstream processing. Furthermore, the ability to tune the chain length (n=1 to 5) allows manufacturers to produce a diverse portfolio of dyes from a common set of intermediates, enhancing supply chain flexibility and reducing lead time for high-purity specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-chromophoric squaraine dyes has been plagued by challenges related to rigidity and poor solubility, which severely limit their practical application in biological and environmental sensing. Traditional approaches often utilize polyether chains or rigid aromatic linkers to connect the squaraine units, resulting in molecules that are prone to unpredictable aggregation in aqueous media. This aggregation leads to broad, ill-defined absorption spectra and significant fluorescence quenching, rendering the dyes ineffective for quantitative analysis. Moreover, conventional ether-linked dyes frequently suffer from poor selectivity towards target metal ions due to the hard oxygen donor atoms, which interact weakly with soft metal ions like mercury or silver. The synthetic routes for these older generations of dyes often involve multi-step protections and deprotections, requiring harsh reaction conditions that degrade the sensitive squaraine core, ultimately resulting in low overall yields and difficult purification processes that hinder commercial viability.
The Novel Approach
The methodology described in CN103613948A fundamentally addresses these deficiencies by introducing a flexible thioether linkage that acts as both a structural spacer and a functional binding site. This novel approach utilizes the soft nucleophilicity of sulfur to facilitate the formation of stable complexes with specific metal ions, inducing distinct exciton coupling effects that serve as a clear optical signal. By replacing rigid ether chains with flexible thioether chains, the new dyes exhibit improved solubility in organic solvents and controlled aggregation behavior in water-miscible mixtures. The synthesis strategy is elegantly simple, involving a direct condensation of thioether-linked dianiline derivatives with semisquaric acid. This one-pot condensation under reduced pressure avoids the formation of complex byproducts and allows for the selective isolation of either symmetrical or unsymmetrical dyes simply by adjusting the molar ratios of the starting materials. This level of control is unprecedented in squaraine chemistry and provides a robust platform for the commercial scale-up of complex functional dyes.
Mechanistic Insights into Thioether-Mediated Exciton Coupling
From a mechanistic perspective, the superior performance of these dyes stems from the intricate interplay between the squaraine zwitterionic core and the flexible thioether tether. The squaraine ring acts as a strong electron acceptor, while the aniline nitrogen atoms serve as electron donors, establishing a potent intramolecular charge transfer (ICT) pathway. When metal ions such as Ca2+ or Hg2+ are introduced, the sulfur atoms in the thioether chain coordinate with the metal, forcing the two squaraine chromophores into a specific spatial arrangement. This conformational restriction triggers exciton coupling, a phenomenon where the excited states of the two chromophores interact, leading to a splitting of energy levels. In the case of calcium detection, this results in a characteristic blue shift and the appearance of new absorption peaks around 562 nm, providing a ratiometric signal that is independent of probe concentration. For mercury detection, the coordination disrupts pre-formed aggregates, leading to a dramatic "turn-on" fluorescence response. This dual-mode sensing capability—colorimetric and fluorometric—is a direct result of the thioether chain's ability to modulate inter-chromophore distance and orientation dynamically.
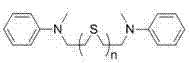
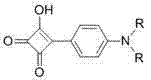
Impurity control in this synthesis is critical for maintaining the sharp optical properties required for high-end sensor applications. The primary impurities typically arise from incomplete condensation or the formation of asymmetric intermediates when stoichiometry is not strictly controlled. However, the patent details a highly effective purification protocol that leverages the polarity differences between the symmetrical product, the asymmetric intermediate, and the unreacted starting materials. By employing silica gel column chromatography with specific eluent systems—such as dichloromethane:ethyl acetate (1:1) for asymmetric dyes and chloroform:methanol (40:1) for symmetrical dyes—manufacturers can achieve high-purity isolates. The stability of the thioether bond under these chromatographic conditions ensures that the final product retains its structural integrity. Furthermore, the use of reduced pressure reflux during the reaction minimizes thermal degradation of the squaraine core, a common issue in high-temperature condensations, thereby ensuring a cleaner crude product profile that simplifies the final purification steps.
How to Synthesize Near-Infrared Squaraine Dyes Efficiently
The synthesis of these advanced functional materials follows a logical, two-stage progression that begins with the construction of the thioether-linked diamine backbone followed by the final condensation with the squaric acid derivative. The initial step involves the nucleophilic substitution of chloroethyl-aniline derivatives with sodium sulfide or the reaction of ethanedithiol with aniline halides, creating the flexible bridge essential for ion recognition. This intermediate is then subjected to a dehydration condensation with semisquaric acid in n-heptanol. The detailed standardized synthesis steps below outline the precise conditions, including temperature controls and stoichiometric ratios, required to maximize yield and selectivity for both symmetrical and unsymmetrical variants. Adhering to these protocols ensures reproducible results suitable for pilot plant operations.
- Preparation of the key intermediate: Synthesize the thioether-linked dianiline derivative by reacting chloroethyl-aniline derivatives with sodium sulfide or ethanedithiol under basic conditions.
- Condensation Reaction: Mix the dianiline derivative with semisquaric acid in n-heptanol and reflux under reduced pressure (76 mmHg) at 132°C for water separation.
- Purification: Remove solvent under reduced pressure, wash the crude product with petroleum ether, and purify via silica gel column chromatography using specific eluent ratios.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in electronic chemical manufacturing and sensor production, the adoption of this thioether-based squaraine technology offers substantial economic benefits derived from process simplification. The elimination of transition metal catalysts, which are often expensive and require rigorous removal to meet purity standards for biological applications, significantly lowers the cost of goods sold (COGS). Additionally, the use of common solvents like n-heptanol and petroleum ether, rather than exotic or highly regulated solvents, streamlines procurement and reduces hazardous waste disposal costs. The robustness of the thioether linkage also implies a longer shelf life for the final product, reducing inventory write-offs and ensuring consistent quality over time. From a supply chain reliability standpoint, the starting materials—aniline derivatives and semisquaric acid—are commodity chemicals with established global supply networks, mitigating the risk of raw material shortages that often plague specialty dye production.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive palladium or copper catalysts typically used in C-N or C-S bond formation, replacing them with cost-effective base-mediated substitutions. This shift drastically reduces the catalyst cost per kilogram of product and removes the necessity for specialized metal scavenging resins during purification. Furthermore, the high atom economy of the condensation reaction, driven by efficient water removal under reduced pressure, maximizes the conversion of raw materials into the final dye, minimizing waste generation and improving overall process efficiency.
- Enhanced Supply Chain Reliability: The reliance on bulk chemical feedstocks such as chloroethyl-aniline and sodium sulfide ensures a stable and continuous supply of raw materials, unlike proprietary intermediates that may be sourced from single suppliers. The simplicity of the reaction conditions—reflux in standard solvents without the need for inert atmosphere gloveboxes or cryogenic cooling—allows for production in multipurpose chemical reactors, increasing manufacturing capacity and flexibility. This adaptability ensures that production schedules can be met consistently, reducing lead times for customers requiring large volumes of high-purity dyes for sensor fabrication.
- Scalability and Environmental Compliance: The process generates minimal hazardous byproducts, primarily consisting of inorganic salts and spent solvents that can be easily recycled or treated using standard wastewater protocols. The absence of heavy metals in the final product simplifies regulatory compliance for applications in food safety or environmental monitoring, where strict limits on metal residues are enforced. The scalability of the reduced pressure reflux technique is well-established in the fine chemical industry, allowing for seamless transition from gram-scale laboratory synthesis to ton-scale commercial production without significant re-engineering of the process equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thioether-linked squaraine dyes. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, providing clarity on performance metrics and handling requirements. Understanding these details is crucial for R&D teams integrating these dyes into new sensor platforms and for procurement officers evaluating supplier capabilities.
Q: What distinguishes thioether-linked squaraine dyes from traditional ether-linked variants?
A: Thioether-linked squaraine dyes offer superior flexibility and specific metal-ion binding capabilities compared to rigid ether chains. The sulfur atom provides a soft donor site that enhances selectivity for heavy metal ions like Hg2+ and alkaline earth metals like Ca2+, enabling precise exciton coupling for dual-channel sensing applications.
Q: How does the synthesis method ensure high purity for commercial scale-up?
A: The patented process utilizes a simplified condensation route in n-heptanol with reduced pressure reflux, which effectively drives the equilibrium forward by removing water. Subsequent purification via silica gel chromatography with tailored eluent systems (e.g., chloroform:methanol 40:1) ensures the removal of asymmetric byproducts, yielding high-purity symmetrical dyes suitable for sensitive optical applications.
Q: Are these dyes stable enough for long-term storage and industrial use?
A: Yes, the incorporation of the flexible thioether chain significantly enhances the photostability and thermal stability of the squaraine core. Experimental data indicates decomposition temperatures exceeding 300°C for symmetrical variants, ensuring robustness during processing and extended shelf life in various solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Near-Infrared Squaraine Dye Supplier
As the demand for advanced optical materials grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and rigorous quality assurance. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including HPLC, NMR, and HR-MS analysis, to guarantee that every batch of near-infrared squaraine dye meets the highest international standards. We understand the critical nature of optical purity in sensor applications and employ advanced chromatographic techniques to remove trace impurities that could interfere with signal accuracy.
We invite you to collaborate with our technical procurement team to explore how this innovative thioether-based technology can enhance your product portfolio. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your development timeline. Let us help you accelerate your time-to-market with reliable, high-performance chemical solutions designed for the next generation of intelligent sensing systems.
