Advanced Palladium-Catalyzed Carbonylation for High-Purity Diaryl Ester Manufacturing
Advanced Palladium-Catalyzed Carbonylation for High-Purity Diaryl Ester Manufacturing
The synthesis of diaryl ester compounds represents a critical challenge in the production of high-value pharmaceutical intermediates and fine chemicals. Traditional methodologies often struggle with harsh reaction conditions, poor atom economy, and the generation of significant hazardous waste. However, recent advancements detailed in patent CN112110817B have introduced a transformative approach utilizing a specialized pyridine-palladium catalyst system. This technology enables the efficient carbonylation of aryl iodides with phenolic compounds under remarkably mild conditions, achieving high yields with minimal catalyst loading. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation offers a pathway to drastically simplify supply chains while enhancing product purity profiles.
The core breakthrough lies in the unique stability and activity of the pyridine-palladium complex, which effectively mitigates the common issue of catalyst deactivation via palladium black formation. By operating at moderate temperatures of 60~100°C and carbon monoxide pressures of 3~6 atm, this method circumvents the need for aggressive activating agents like acid chlorides or anhydrides. The result is a streamlined synthetic route that aligns perfectly with modern green chemistry principles, offering substantial potential for cost reduction in fine chemical manufacturing. As we delve deeper into the technical specifics, it becomes evident that this protocol is not merely an academic curiosity but a robust industrial solution ready for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aromatic esters has relied heavily on the reaction between acyl halides or acid anhydrides and nucleophilic phenols. While chemically straightforward, these classical approaches suffer from severe drawbacks that hinder their viability in modern, regulated industries. The use of acyl halides necessitates the handling of corrosive reagents and generates stoichiometric amounts of acidic byproducts, requiring extensive neutralization and waste treatment procedures. Furthermore, the atom economy of such reactions is inherently poor, as a significant portion of the molecular mass of the starting materials ends up as waste rather than incorporated into the final product. In the context of producing high-purity diaryl esters for sensitive applications, the residual halide impurities from acid chlorides can be particularly problematic, often demanding rigorous and costly purification steps to meet stringent regulatory specifications.
Additionally, alternative palladium-catalyzed carbonylation methods using carbon monoxide have faced their own set of challenges. A persistent issue in these systems is the tendency of the palladium catalyst to reduce to metallic palladium (palladium black) under the reducing atmosphere of CO gas. This deactivation phenomenon leads to a rapid loss of catalytic activity, forcing operators to use excessive amounts of expensive precious metal catalysts to drive the reaction to completion. Such inefficiencies not only inflate raw material costs but also complicate downstream processing due to the presence of colloidal metal particles. Consequently, there has been a long-standing industry demand for a catalytic system that combines high turnover numbers with exceptional stability against deactivation.
The Novel Approach
The methodology disclosed in patent CN112110817B addresses these historical pain points through the deployment of a specifically engineered pyridine-palladium catalyst. This novel system exhibits remarkable resistance to deactivation, maintaining high catalytic activity even under the reducing environment of carbon monoxide. The reaction proceeds smoothly by coupling aryl iodides with phenols in the presence of triethylamine and the palladium complex, utilizing carbon monoxide as the carbonyl source. This approach fundamentally shifts the paradigm from substitution chemistry to addition chemistry, thereby maximizing atom economy. The general reaction scheme is illustrated below, showcasing the direct conversion of readily available starting materials into valuable ester linkages.
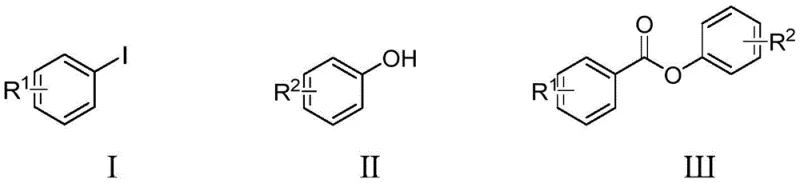
By eliminating the need for pre-activated acid derivatives, this method significantly reduces the E-factor (environmental factor) of the process. The operational simplicity is another key advantage; the reaction tolerates a wide range of functional groups, including halogens, alkoxy groups, and trifluoromethyl substituents, without the need for protecting group strategies. This broad substrate scope ensures that the process is versatile enough to handle diverse molecular architectures required in drug discovery and agrochemical development. For supply chain managers, this translates to a more resilient sourcing strategy, as the reliance on unstable or hazardous intermediates is minimized.
Mechanistic Insights into Pyridine-Palladium Catalyzed Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The catalytic cycle likely initiates with the oxidative addition of the aryl iodide to the active palladium(0) species generated in situ from the pyridine-palladium precursor. The unique ligand environment provided by the pyridine moiety stabilizes the metal center, preventing the aggregation that typically leads to palladium black precipitation. Following oxidative addition, the coordination and subsequent migratory insertion of carbon monoxide into the palladium-carbon bond occur, forming an acyl-palladium intermediate. This step is critical and is facilitated by the moderate CO pressure (3~6 atm) specified in the patent, which ensures sufficient concentration of the gaseous reagent without overwhelming the catalyst.
The final stage of the cycle involves the nucleophilic attack of the phenol (activated by the base, triethylamine) on the acyl-palladium species, followed by reductive elimination to release the diaryl ester product and regenerate the active catalyst. The structural integrity of the catalyst, as depicted in the patent, plays a pivotal role in this regeneration step. The specific coordination geometry prevents the formation of inactive dimers or clusters. This mechanistic robustness allows the catalyst loading to be kept exceptionally low, ranging from 0.1% to 0.2% molar relative to the substrate. Such efficiency is rare in carbonylation chemistry and underscores the commercial viability of the technology. The structure of this high-performance catalyst is shown below, highlighting the chelating nature of the ligand system.
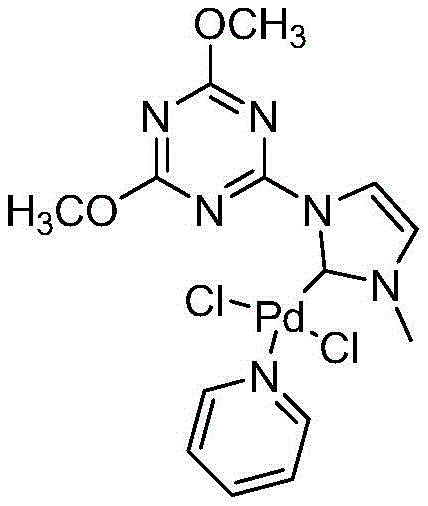
Furthermore, the choice of triethylamine as the base is not arbitrary; it serves to neutralize the hydrogen iodide byproduct formed during the reaction, driving the equilibrium forward and preventing acid-catalyzed side reactions. The use of toluene as the preferred solvent provides an optimal balance of solubility for the organic substrates and thermal stability at the reaction temperatures of 80~100°C. This careful optimization of every reaction parameter—from ligand design to solvent selection—ensures that the process delivers consistent, high-quality results. For technical teams, this level of mechanistic clarity provides a solid foundation for troubleshooting and scaling the reaction to multi-kilogram batches without compromising yield or purity.
How to Synthesize Diaryl Esters Efficiently
Implementing this advanced carbonylation protocol requires adherence to specific operational parameters to maximize the benefits of the pyridine-palladium system. The process is designed to be operationally simple, avoiding the need for exotic equipment beyond standard high-pressure reactors capable of handling carbon monoxide safely. The following section outlines the generalized workflow derived from the patent examples, serving as a starting point for process development. It is important to note that while the patent provides robust baseline conditions, fine-tuning may be required for sterically hindered substrates or highly sensitive functional groups. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining aryl iodide, phenol, pyridine-palladium catalyst (0.1-0.2 mol%), and triethylamine in toluene solvent.
- Introduce carbon monoxide gas into the reactor and maintain a pressure between 3 to 6 atm while heating the mixture to 60-100°C.
- Stir the reaction for 6 to 12 hours, then cool to room temperature, remove solvent, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver for cost optimization lies in the drastic reduction of catalyst consumption. Traditional palladium-catalyzed reactions often require loadings of 1-5 mol% or higher to compensate for deactivation, whereas this method operates efficiently at 0.1-0.2 mol%. Given the high market price of palladium, this order-of-magnitude reduction translates directly into significant raw material cost savings. Moreover, the lower metal content in the crude reaction mixture simplifies the purification process, potentially reducing the number of chromatography steps or the amount of scavenger resin required to meet heavy metal specifications.
- Cost Reduction in Manufacturing: The elimination of acid chlorides and anhydrides removes the need for purchasing these expensive and hazardous activating agents. Additionally, the high atom economy of using carbon monoxide means that nearly all atoms from the starting materials are incorporated into the final product, minimizing waste disposal costs. The mild reaction conditions (60-100°C) also reduce energy consumption compared to high-temperature processes, contributing to a lower overall carbon footprint and utility expenditure. These factors combined create a leaner, more cost-effective manufacturing profile that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis—aryl iodides, phenols, and carbon monoxide—are commodity chemicals with stable and widespread supply chains. Unlike specialized reagents that may face shortages or long lead times, these inputs are readily available from multiple vendors. The robustness of the catalyst, which is stable to air, further simplifies logistics by removing the need for strict inert atmosphere storage and handling during transportation. This reliability ensures consistent production schedules and reduces the risk of delays caused by raw material scarcity or degradation.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of triethylamine hydroiodide salts which are easier to manage than the mixed waste streams from acid chloride routes. The high selectivity of the reaction reduces the formation of byproducts, leading to cleaner crude products and higher overall throughput. From a regulatory perspective, the ability to produce high-purity intermediates with controlled impurity profiles facilitates faster regulatory approval for downstream pharmaceutical applications. The scalability is proven by the successful synthesis of various substituted derivatives with consistently high yields, demonstrating the method's versatility for commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine-palladium catalyzed carbonylation technology. These answers are derived directly from the experimental data and technical specifications outlined in patent CN112110817B. They are intended to provide clarity for decision-makers evaluating the feasibility of integrating this method into their existing manufacturing workflows. Understanding these nuances is essential for accurate risk assessment and project planning.
Q: What are the advantages of using pyridine-palladium catalyst over traditional methods?
A: The pyridine-palladium catalyst offers superior air stability and prevents the formation of inactive palladium black, allowing for significantly lower catalyst loading (0.1-0.2 mol%) compared to conventional systems, which reduces heavy metal residues and purification costs.
Q: What is the substrate scope for this carbonylation reaction?
A: The method demonstrates excellent tolerance for various functional groups, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (Cl, Br) and trifluoromethyl, yielding products with 93-98% efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (60-100°C, 3-6 atm CO) and inexpensive raw materials (aryl iodides, phenols, CO), making it highly scalable with improved atom economy and reduced environmental impact compared to acid chloride routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthetic routes in the modern chemical industry. Our technical team has thoroughly analyzed the potential of the pyridine-palladium catalyzed carbonylation method and is fully equipped to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. Our state-of-the-art facilities are designed to handle high-pressure carbonylation reactions safely, adhering to the highest international safety and quality standards.
We invite you to collaborate with us to explore how this advanced chemistry can optimize your supply chain. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how switching to this catalytic system can improve your margins. Please contact our technical procurement team today to request specific COA data for similar diaryl ester intermediates and to discuss route feasibility assessments. Let us partner with you to deliver high-purity pharmaceutical intermediates that drive your innovation forward.
