Advanced Aqueous Synthesis of Dichloroaryl Ethers for Scalable Pharmaceutical Production
Advanced Aqueous Synthesis of Dichloroaryl Ethers for Scalable Pharmaceutical Production
The chemical industry constantly seeks methods to optimize the synthesis of critical intermediates, balancing efficiency with economic viability. Patent CN1276905C introduces a transformative approach for synthesizing dichloroaryl ethers, a class of compounds vital for pharmaceutical and agrochemical applications. This technology leverages a synergistic catalytic system comprising potassium iodide and phase transfer catalysts within a purely aqueous medium. By shifting away from traditional organic solvents and expensive alkylating agents, this method addresses long-standing challenges in nucleophilic substitution reactions. The process enables the use of low-cost chloroalkanes, which are typically sluggish reactants, by activating them in situ to achieve yields comparable to more expensive bromoalkane routes. This innovation represents a significant leap forward for manufacturers aiming to enhance the sustainability and cost-effectiveness of their intermediate production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aryl ethers via nucleophilic substitution has been hindered by the reactivity hierarchy of haloalkanes, where chloroalkanes are significantly less reactive than their bromo or iodo counterparts. To achieve acceptable reaction rates and yields, conventional processes often rely on bromoalkanes, which impose a heavy financial burden due to their high market price. Alternatively, when chloroalkanes are used to save on raw material costs, the reactions suffer from excessively long durations, often extending beyond 15 to 35 hours, and result in suboptimal yields around 80%. Furthermore, the low boiling points of many chloroalkanes restrict the operating temperature of the reaction, preventing the thermal acceleration necessary for efficient conversion. These limitations frequently lead to side reactions, such as the hydrolysis of the chlorophenol substrate or the alkylating agent, which compromises the purity of the final product and complicates downstream purification efforts.
The Novel Approach
The methodology described in CN1276905C overcomes these kinetic and economic barriers through a clever dual-catalyst strategy in a water-only solvent system. By introducing potassium iodide alongside a quaternary ammonium or phosphonium phase transfer catalyst, the process effectively converts the inert chloroalkane into a highly reactive iodoalkane species within the organic phase. This in situ generation of the active alkylating agent allows the reaction to proceed smoothly at elevated temperatures, exceeding 90°C, which would be hazardous or impossible with volatile organic solvents. The result is a dramatic reduction in reaction time to approximately 8-10 hours and an increase in yield to roughly 85%, all while utilizing the cheapest available alkyl halide feedstock. Additionally, the elimination of organic solvents simplifies the workup procedure, removing the need for energy-intensive solvent recovery and distillation steps.
Mechanistic Insights into KI-Assisted Phase Transfer Catalysis
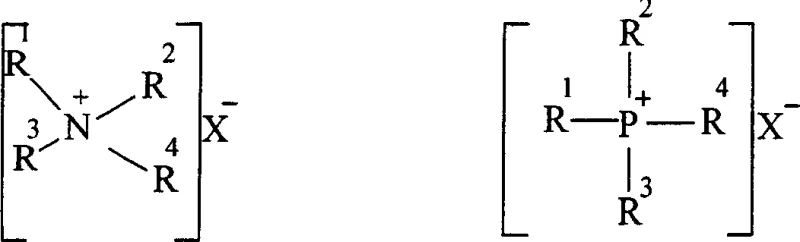
The core of this technological advancement lies in the intricate interplay between the phase transfer catalyst and the iodide anion. The phase transfer catalyst, typically a quaternary ammonium salt like cetyltrimethylammonium bromide or a phosphonium salt, functions to shuttle anions across the interface between the aqueous and organic phases. In this specific system, the catalyst transports the iodide anion from the aqueous sodium hydroxide solution into the organic phase containing the chloroalkane. Once in the organic phase, the iodide ion acts as a potent nucleophile, displacing the chloride atom on the alkane to form an iodoalkane. This transformation is crucial because the carbon-iodine bond is weaker and the iodide is a better leaving group than chloride, making the resulting iodoalkane far more susceptible to attack by the dichlorophenoxy anion.
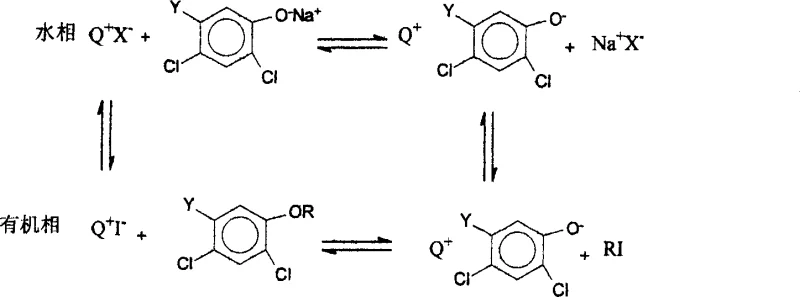
Following the activation of the alkylating agent, the dichlorophenoxy anion, also transported into the organic phase by the catalyst, attacks the newly formed iodoalkane to generate the desired dichloroaryl ether. A key feature of this cycle is the regeneration of the iodide anion; as the etherification occurs, the iodide is released back into the system to activate another molecule of chloroalkane. This means that potassium iodide acts in a catalytic capacity rather than being consumed stoichiometrically, further enhancing the economic efficiency of the process. The phase transfer catalyst plays a secondary but equally vital role in transporting the phenoxide nucleophile, ensuring that both reactive species meet in the organic phase where the substitution takes place. This dual-transport mechanism ensures high collision frequency between reactants, driving the reaction to completion with high selectivity and minimal formation of hydrolysis byproducts.
How to Synthesize Dichloroaryl Ether Efficiently
Implementing this synthesis route requires careful control of reaction parameters to maximize the benefits of the KI/PTC system. The process begins with the preparation of an aqueous slurry containing the dichlorophenol substrate, solid sodium hydroxide, the selected phase transfer catalyst, and a catalytic amount of potassium iodide. The mixture is heated to initiate the formation of the phenoxide salt and dissolve the components. Once the temperature stabilizes between 70°C and 100°C, the chloroalkane is added dropwise to maintain thermal control and prevent runaway exotherms. Detailed standardized synthetic steps see the guide below.
- Prepare the aqueous reaction mixture by dissolving dichlorophenol and sodium hydroxide in water, then adding the phase transfer catalyst and potassium iodide.
- Heat the mixture to 70-100°C and slowly dropwise add the chloroalkane while maintaining the temperature between 65-100°C for 8-10 hours.
- Cool the reaction, wash with NaOH solution to remove impurities, separate the organic layer, distill off excess chloroalkane, and purify the final ether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this aqueous, chloroalkane-based process offers profound strategic advantages beyond simple yield improvements. The most immediate impact is seen in raw material procurement, where the substitution of expensive bromoalkanes with commodity-grade chloroalkanes drastically lowers the bill of materials. This cost reduction is compounded by the elimination of organic solvents, which removes the entire logistical and financial burden associated with purchasing, storing, handling, and recovering volatile organic compounds. The simplified process flow also translates to reduced utility consumption, as there is no need for complex solvent distillation trains, thereby lowering the overall operational expenditure per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of low-cost chloroalkanes and the catalytic nature of the iodide additive. By avoiding the use of stoichiometric amounts of expensive reagents and eliminating the capital and operational costs associated with organic solvent recovery systems, manufacturers can achieve substantial cost savings. The ability to run the reaction at higher temperatures in water also improves energy efficiency by reducing the total reaction time, allowing for higher throughput in existing reactor vessels without the need for significant capital investment in new equipment.
- Enhanced Supply Chain Reliability: Relying on chloroalkanes rather than specialized bromoalkanes mitigates supply risk, as chloroalkanes are produced on a massive global scale for various industries and are less prone to market shortages. The robustness of the aqueous system also reduces the likelihood of batch failures caused by solvent quality issues or moisture sensitivity, which are common pain points in organic synthesis. This stability ensures a more predictable production schedule, allowing supply chain planners to commit to delivery timelines with greater confidence and reduce the need for safety stock inventory.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the water-based nature of this reaction significantly simplifies waste management and compliance reporting. The absence of halogenated or aromatic organic solvents reduces the toxicity of the waste stream and lowers the costs associated with hazardous waste disposal. Furthermore, the process is inherently safer to scale up, as water acts as a heat sink to manage exothermic events, reducing the risk of thermal runaway. This makes the technology ideal for large-scale commercial production, facilitating the transition from pilot plant to multi-ton manufacturing with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this dichloroaryl ether synthesis technology. These insights are derived directly from the experimental data and mechanistic principles outlined in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific intermediate requirements.
Q: Why is potassium iodide added to the reaction involving chloroalkanes?
A: Potassium iodide acts as a co-catalyst that converts the less reactive chloroalkane into a highly reactive iodoalkane in situ via a halogen exchange reaction. This activation significantly accelerates the nucleophilic substitution rate, overcoming the inherent low reactivity of chloroalkanes compared to bromo- or iodoalkanes.
Q: What are the advantages of using water as the sole solvent in this synthesis?
A: Using water eliminates the need for expensive and hazardous organic solvents, drastically reducing raw material costs and environmental waste. Furthermore, the absence of volatile organic solvents allows the reaction to be heated to higher temperatures (above 90°C) safely, which further enhances reaction kinetics and overall yield.
Q: How does this method improve supply chain reliability for ether intermediates?
A: By enabling the use of cheap and readily available chloroalkanes instead of costly bromoalkanes, the process reduces dependency on scarce reagents. The simplified workup procedure, which avoids complex solvent recovery distillation, also shortens production cycles and ensures more consistent batch-to-batch availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dichloroaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value intermediates like dichloroaryl ethers. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of innovative patents like CN1276905C are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to process excellence allows us to deliver products that are not only chemically pure but also economically optimized for our partners' downstream applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and reduce your overall time to market.
