Advanced Synthesis of Dichloroaryl Ethers: A Cost-Effective Phase Transfer Catalytic Route for Industrial Scale-Up
Introduction to Patent CN1276905C Technology
The chemical industry is constantly seeking more efficient and cost-effective pathways for synthesizing critical organic intermediates, and the technology disclosed in patent CN1276905C represents a significant breakthrough in the production of dichloroaryl ethers. This specific patent outlines a novel method for synthesizing dichloroaryl ethers by utilizing dichlorophenols and chloroalkanes as primary raw materials within a unique phase transfer catalytic system. Unlike traditional methods that rely on expensive bromoalkanes or suffer from low reactivity when using chloroalkanes, this invention introduces a synergistic catalytic approach involving potassium iodide and quaternary ammonium or phosphonium salts. The process is conducted exclusively in an aqueous medium, eliminating the need for hazardous organic solvents and thereby simplifying the downstream purification steps significantly. By overcoming the inherent kinetic barriers associated with chloroalkane etherification, this technology enables the industrial realization of high-yield synthesis using the most economically viable alkylating agents available in the global chemical market today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dichloroaryl ethers such as 2,4-dichlorophenyl isopropyl ether has been plagued by significant economic and technical inefficiencies when relying on standard nucleophilic substitution protocols. Conventional wisdom dictates that for efficient etherification, one must utilize highly reactive haloalkanes like bromoalkanes or iodoalkanes because the reactivity order follows the trend of iodide greater than bromide greater than chloride. However, iodoalkanes are prohibitively expensive for large-scale industrial applications, while bromoalkanes, though more affordable, still impose a substantial cost burden on the final product pricing. When manufacturers attempt to use the cheapest option, chloroalkanes, they typically encounter sluggish reaction kinetics that require excessively long reaction times, often exceeding thirty hours, which drastically reduces throughput. Furthermore, the low boiling points of many chloroalkanes limit the operating temperature of the reaction, preventing the thermal acceleration necessary to drive the equilibrium towards the desired ether product, ultimately resulting in poor yields and significant formation of hydrolysis byproducts.
The Novel Approach
The methodology presented in this patent fundamentally disrupts the traditional cost-performance trade-off by enabling the use of low-cost chloroalkanes without sacrificing reaction speed or yield. By introducing a dual-catalyst system comprising a phase transfer catalyst and potassium iodide, the process effectively activates the inert chloroalkane in situ through a halogen exchange mechanism. This innovative approach generates a small amount of highly reactive iodoalkane within the reaction mixture, which then rapidly undergoes nucleophilic attack by the phenoxide anion to form the target ether. Crucially, the reaction is performed in water as the sole solvent, which not only reduces raw material costs but also enhances safety by removing flammable organic solvents from the process equation. The ability to operate at elevated temperatures above ninety degrees Celsius, facilitated by the aqueous environment, further accelerates the reaction rate, allowing the process to achieve yields of approximately eighty-five percent in a fraction of the time required by conventional chloroalkane methods.
Mechanistic Insights into KI-Assisted Phase Transfer Catalysis
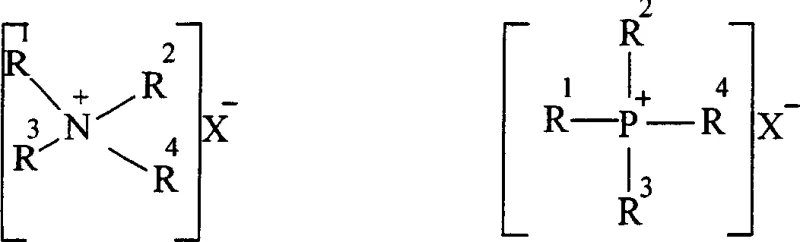
The core scientific innovation of this synthesis route lies in the intricate interplay between the phase transfer catalyst and the iodide anion, which creates a highly efficient catalytic cycle for nucleophilic substitution. In this system, the phase transfer catalyst, typically a quaternary ammonium salt as shown in the structural diagrams, functions to transport the hydrophilic phenoxide anion from the aqueous phase into the organic phase where the alkylating agent resides. Simultaneously, the added potassium iodide participates in a Finkelstein-type reaction where the iodide anion displaces the chloride atom on the chloroalkane to generate an iodoalkane intermediate. This in situ generated iodoalkane possesses a much lower activation energy for nucleophilic attack, allowing the phenoxide anion to displace the iodine atom rapidly and regenerate the iodide anion. The regenerated iodide is then free to react with another molecule of chloroalkane, establishing a continuous catalytic loop that maintains a low but effective concentration of the highly reactive iodo-species throughout the reaction duration.
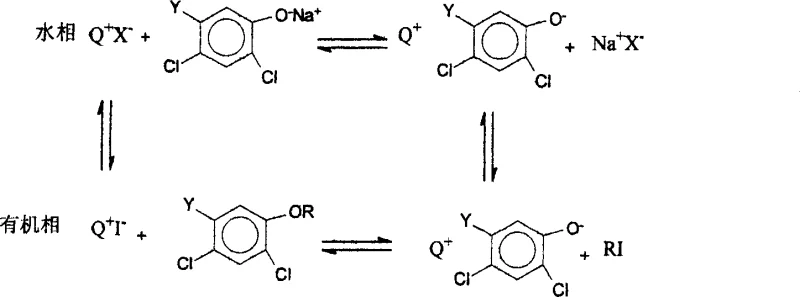
From an impurity control perspective, this mechanism offers distinct advantages over direct alkylation methods by minimizing the residence time of reactive intermediates that could lead to side reactions. The rapid conversion of the activated iodoalkane prevents its accumulation, thereby reducing the likelihood of competing elimination reactions or poly-alkylation events that often degrade product purity. Additionally, the use of water as the solvent medium facilitates the easy removal of inorganic salts and unreacted phenolic starting materials through simple aqueous washing steps post-reaction. The phase transfer catalyst ensures that the organic product remains in the organic phase while ionic byproducts are sequestered in the aqueous layer, leading to a crude product profile that is significantly cleaner than those obtained from traditional organic solvent-based processes. This inherent selectivity contributes to the final product purity exceeding ninety-five percent after standard purification techniques such as distillation or recrystallization are applied.
How to Synthesize Dichloroaryl Ether Efficiently
The practical implementation of this synthesis route involves a straightforward procedure that begins with the preparation of an aqueous alkaline solution containing the dichlorophenol substrate and the catalytic system. Operators must carefully control the addition rate of the chloroalkane to manage the exothermic nature of the etherification while maintaining the optimal temperature window for maximum conversion efficiency. The standardized protocol ensures consistent quality and yield by strictly adhering to the molar ratios and thermal profiles defined in the patent specifications. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the aqueous reaction mixture by dissolving dichlorophenol and sodium hydroxide in water, then add the phase transfer catalyst and potassium iodide.
- Heat the mixture to 70-100°C and slowly dropwise add the chloroalkane while maintaining the temperature between 65-100°C for 8-10 hours.
- Cool the reaction, wash with NaOH solution to remove impurities, separate the organic layer, and purify via distillation or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into immediate and tangible improvements in the cost structure and reliability of the supply base for dichloroaryl ether intermediates. The primary driver of value creation is the substitution of expensive bromoalkanes or iodoalkanes with widely available and inexpensive chloroalkanes, which constitute a major portion of the raw material bill of materials. This switch does not merely offer marginal savings but represents a fundamental restructuring of the input costs, allowing manufacturers to offer highly competitive pricing without compromising on the quality or purity specifications required by downstream pharmaceutical and agrochemical clients. Furthermore, the elimination of organic solvents from the reaction matrix removes the logistical and financial burden associated with solvent procurement, storage, and the complex regulatory compliance required for volatile organic compound emissions.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, primarily driven by the drastic reduction in raw material expenses achieved by utilizing chloroalkanes instead of their heavier halogenated counterparts. By avoiding the need for costly solvent recovery and distillation units, the capital expenditure and operational energy consumption of the manufacturing facility are significantly lowered. The simplified workup procedure, which relies on aqueous washing rather than complex extraction sequences, further reduces labor costs and processing time per batch. These cumulative efficiencies result in a substantially lower cost of goods sold, providing a robust buffer against market volatility in raw material pricing and enhancing overall profit margins for the production of high-purity dichloroaryl ethers.
- Enhanced Supply Chain Reliability: From a supply chain resilience standpoint, relying on chloroalkanes offers a distinct advantage due to their status as commodity chemicals with stable and abundant global availability. Unlike specialized bromoalkanes which may be subject to supply constraints or long lead times from niche suppliers, chloroalkanes can be sourced from multiple vendors, ensuring continuity of supply even during market disruptions. The robustness of the aqueous-based process also mitigates risks associated with solvent shortages or transportation restrictions on hazardous flammable liquids. This stability allows for more accurate production planning and inventory management, ensuring that delivery commitments to key accounts in the pharmaceutical and agrochemical sectors are consistently met without unexpected delays.
- Scalability and Environmental Compliance: The environmental profile of this synthesis method aligns perfectly with modern green chemistry principles, making it easier to scale up while maintaining strict regulatory compliance. The absence of organic solvents eliminates the generation of hazardous waste streams associated with solvent disposal, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing site. The high atom economy and improved yield of the process mean that less raw material is wasted, contributing to a more sustainable production lifecycle. These factors facilitate smoother regulatory approvals for capacity expansions and enable the seamless transition from pilot scale to multi-ton commercial production, ensuring that the supply chain can grow in tandem with market demand for these critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phase transfer catalytic method for dichloroaryl ether synthesis. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this technology into existing manufacturing workflows or for qualifying new suppliers who utilize this advanced synthetic route.
Q: Why is potassium iodide added to the reaction system?
A: Potassium iodide acts as a co-catalyst that reacts with the cheap chloroalkane to generate a highly reactive iodoalkane in situ, significantly accelerating the nucleophilic substitution rate.
Q: What are the advantages of using water as the sole solvent?
A: Using water eliminates the need for expensive organic solvents, simplifies the workup process by avoiding solvent recovery distillation, and allows for higher reaction temperatures due to the absence of volatile organics.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes inexpensive raw materials like chloroalkanes and avoids complex solvent handling, making it highly suitable for large-scale commercial manufacturing with yields reaching approximately 85%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dichloroaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern chemical supply chain, and we have integrated advanced technologies like the one described in CN1276905C into our manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global multinational corporations. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of dichloroaryl ether meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to process optimization allows us to deliver high-quality intermediates with consistent impurity profiles, supporting our partners in their own regulatory filings and product development timelines.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project needs and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our production efficiencies can translate into value for your organization. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a transparent and productive partnership from the initial inquiry through to commercial supply.
