Advanced Synthesis of Dabigatran Etexilate Intermediate Tosylate via Direct Alcoholysis
Introduction to Next-Generation Anticoagulant Intermediate Manufacturing
The pharmaceutical landscape for anticoagulant therapy has been revolutionized by direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule since its approval. However, the commercial viability of such complex small molecules relies heavily on the efficiency of their synthetic routes, particularly the formation of stable, high-purity intermediates. Patent CN102977077A discloses a groundbreaking methodology for preparing the p-toluenesulfonate salt of the key Dabigatran intermediate, addressing critical bottlenecks in prior art. This technical insight report analyzes the shift from multi-step isolation processes to a streamlined, one-pot alcoholysis-ammonolysis strategy. By leveraging the dual functionality of p-toluenesulfonic acid as both a reactant and a promoter, this process eliminates the need for concentrating unstable amidine intermediates, thereby enhancing overall process robustness. For R&D and supply chain leaders, understanding this transition is vital for securing a reliable supply of high-quality API precursors that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Dabigatran intermediate amidine salt has been plagued by significant operational challenges that hinder scalability and cost-efficiency. Traditional routes, such as those disclosed in WO2008/095928, typically employ a Pinner reaction to generate the amidine base, followed by a separate salification step. A major drawback of this approach is the inherent instability of the free amidine compound (Formula IV), which is susceptible to decomposition and difficult to purify. Furthermore, alternative pathways involving the reduction of oxadiazole precursors using palladium on carbon (Pd/C), as shown in the reaction scheme below, introduce additional complexities including the requirement for high-pressure hydrogenation and the removal of heavy metal catalysts.
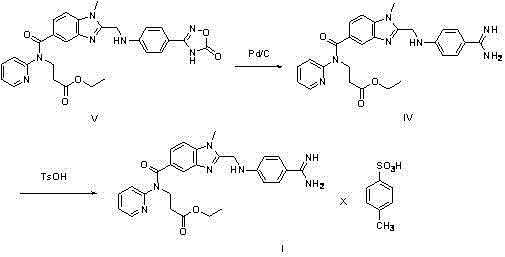
These conventional methods necessitate multiple isolation, concentration, and drying steps, each representing a potential point of yield loss and impurity generation. The hydrochloride and acetate salts of the intermediate are notably hygroscopic, making them prone to absorbing moisture from the environment, which complicates handling, storage, and downstream processing. Consequently, these factors contribute to a loaded-down production technique that expends excessive reagents and energy, ultimately driving up the cost of goods sold (COGS) and extending lead times for pharmaceutical manufacturers.
The Novel Approach
In stark contrast to the fragmented traditional workflows, the novel method described in CN102977077A introduces a cohesive, telescoped synthesis that transforms the nitrile precursor (Formula II) directly into the stable tosylate salt (Formula I). This innovative route capitalizes on the unique properties of p-toluenesulfonic acid, which serves not merely as a counter-ion for salt formation but actively promotes the initial alcoholysis of the nitrile group. By dissolving the starting material in an organic solvent like ethanol and introducing mineral acid alongside the tosic acid, the reaction proceeds through an imidate intermediate (Formula III) without the need for isolation.
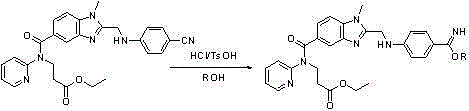
The strategic advantage of this approach lies in its ability to bypass the isolation of the unstable amidine entirely. Instead of filtering and drying a sensitive intermediate, the process flows seamlessly into an ammonolysis step where an ammonia source is added to the same reaction vessel. This direct conversion results in the precipitation of the final tosylate product, which possesses superior crystallinity and stability compared to its hydrochloride or acetate counterparts. The elimination of concentration and drying steps between the alcoholysis and ammonolysis phases drastically simplifies the operational workflow, reducing the risk of thermal degradation and ensuring a consistently high-purity output suitable for GMP manufacturing environments.
Mechanistic Insights into Acid-Promoted Alcoholysis and Ammonolysis
The core chemical innovation of this process revolves around the synergistic catalytic effect of mineral acids and p-toluenesulfonic acid during the alcoholysis phase. When the nitrile substrate is exposed to dry hydrogen chloride gas in an alcoholic solvent, the nitrile nitrogen is protonated, increasing the electrophilicity of the carbon atom and facilitating nucleophilic attack by the alcohol. The presence of p-toluenesulfonic acid further enhances this transformation, likely by stabilizing the transition state or the resulting imidate intermediate through hydrogen bonding or ion-pairing interactions. This dual-acid system ensures that the conversion to the imidate (Formula III) proceeds rapidly and completely even at moderate temperatures ranging from -10°C to 30°C, preventing the accumulation of unreacted starting material.
Following the formation of the imidate, the introduction of an ammonia source triggers the ammonolysis reaction, converting the imidate ester into the desired amidine functionality while simultaneously locking the molecule into its stable tosylate salt form. This in-situ salt formation is critical for impurity control; by immediately generating the crystalline tosylate, the process prevents the amidine from undergoing hydrolysis back to the amide or nitrile, which are common side reactions in aqueous or moist environments. The resulting crystal lattice of the tosylate salt is robust and non-hygroscopic, effectively excluding solvent inclusions and organic impurities that might otherwise co-precipitate. This mechanistic elegance ensures that the final product achieves purity levels exceeding 99% as confirmed by HPLC analysis, meeting the rigorous specifications required for downstream coupling reactions in the synthesis of Dabigatran Etexilate.
How to Synthesize Dabigatran Etexilate Intermediate Tosylate Efficiently
Implementing this novel synthesis route requires precise control over stoichiometry and temperature to maximize yield and minimize byproduct formation. The process begins with the dissolution of the nitrile precursor in a suitable alcohol, followed by the controlled addition of p-toluenesulfonic acid and the saturation of the solution with dry hydrogen chloride gas. Maintaining the reaction temperature within the optimal window of 0-10°C during the alcoholysis phase is essential to prevent exothermic runaways that could degrade the sensitive intermediates. Once the imidate formation is complete, the reaction mixture is warmed to ambient temperature, and the ammonia source is introduced to drive the final conversion. The detailed standardized operating procedures, including specific molar ratios and stirring times derived from the patent embodiments, are outlined in the technical guide below.
- Dissolve the nitrile precursor (Formula II) in an organic solvent such as ethanol and add p-toluenesulfonic acid.
- Introduce dry hydrogen chloride gas at low temperature (-10 to 30°C) to catalyze the alcoholysis reaction, forming the imidate intermediate in situ.
- Add an ammonia source (such as liquefied ammonia or ammonium carbonate) to effect ammonolysis, precipitating the final tosylate salt (Formula I) directly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound advantages in terms of cost structure and logistical reliability. By fundamentally altering the process architecture to eliminate intermediate isolation steps, the method significantly reduces the consumption of solvents and energy associated with evaporation and drying operations. The removal of the Pd/C reduction step also eradicates the need for expensive noble metal catalysts and the subsequent costly purification stages required to remove trace heavy metals from the API, leading to substantial cost reductions in pharmaceutical intermediate manufacturing. Furthermore, the use of commodity chemicals like ethanol and tosic acid ensures that raw material sourcing remains stable and unaffected by the volatility of specialized reagent markets.
- Cost Reduction in Manufacturing: The telescoped nature of this reaction sequence drastically cuts down on unit operations, directly translating to lower labor and utility costs per kilogram of product. By avoiding the concentration and drying of the unstable amidine intermediate, the process minimizes material losses that typically occur during transfer and handling, thereby improving the overall mass balance. The high yield profile, consistently demonstrated above 90% in patent examples, ensures that less starting material is required to produce the same amount of final product, optimizing the cost of goods. Additionally, the simplified work-up procedure reduces the volume of waste solvent generated, lowering disposal costs and environmental compliance burdens.
- Enhanced Supply Chain Reliability: The physical properties of the tosylate salt play a pivotal role in securing the supply chain against disruptions caused by product degradation. Unlike the hygroscopic hydrochloride and acetate salts which require strict humidity control and specialized packaging, the tosylate form is chemically stable and easy to handle under standard warehouse conditions. This robustness extends the shelf life of the intermediate, allowing for larger batch production and strategic stockpiling without the risk of quality drift. The reliability of the synthesis also means that production schedules are less prone to delays caused by failed batches or extended purification cycles, ensuring a steady flow of materials to downstream API manufacturers.
- Scalability and Environmental Compliance: From a scale-up perspective, the absence of high-pressure hydrogenation equipment removes a significant barrier to increasing production capacity, allowing the reaction to be performed in standard glass-lined or stainless steel reactors. The mild reaction conditions, operating near ambient temperatures and pressures, reduce the safety risks associated with exothermic events, making the process inherently safer for large-scale operations. Moreover, the streamlined workflow generates less chemical waste and utilizes greener solvents like ethanol, aligning with modern sustainability goals and facilitating easier regulatory approval for the manufacturing site. This combination of safety, scalability, and environmental friendliness makes the technology highly attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this critical anticoagulant intermediate. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their existing supply chains and ensures alignment with quality expectations.
Q: Why is the tosylate salt preferred over hydrochloride or acetate salts for this intermediate?
A: The tosylate salt exhibits significantly lower hygroscopicity compared to the hydrochloride and acetate forms, which are prone to absorbing water and difficult to crystallize. This superior chemical and thermal stability facilitates easier purification and storage, making it ideal for large-scale pharmaceutical manufacturing.
Q: How does this novel method improve yield compared to traditional Pinner reactions?
A: Traditional methods often require isolating the unstable amidine intermediate, leading to decomposition and loss of material during concentration and drying. This novel approach performs the reaction in a one-pot sequence where the tosic acid promotes alcoholysis and immediate salt formation, bypassing the isolation of the unstable free base and achieving yields exceeding 90% with purity above 99%.
Q: What are the critical reaction conditions for the alcoholysis step?
A: The alcoholysis reaction is optimally conducted in ethanol or methanol at temperatures between -10°C and 30°C, with a preferred range of 0-10°C. The presence of p-toluenesulfonic acid acts as a crucial promoter, while dry hydrogen chloride gas serves as the catalyst to drive the conversion of the nitrile to the imidate efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of anticoagulant synthesis demands a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Dabigatran intermediate meets the highest international standards. Our facility is designed to handle sensitive chemistries safely, utilizing the latest process intensification techniques to deliver cost-effective solutions without compromising on product integrity.
We invite you to collaborate with us to optimize your supply chain for Dabigatran Etexilate production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized synthetic route can reduce your overall manufacturing expenses. Please contact us today to request specific COA data for our current inventory and to discuss route feasibility assessments for your upcoming projects. Let us be your strategic partner in delivering life-saving medications to patients worldwide through superior chemical manufacturing excellence.
