Advanced Catalytic Hydrogenation for High-Purity Dabigatran Etexilate Intermediates
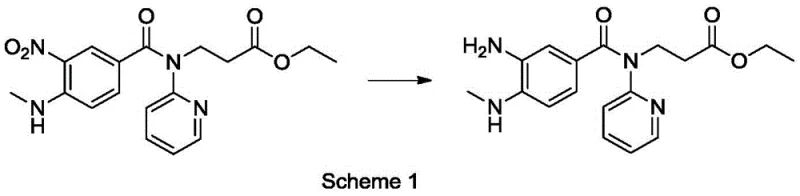
The pharmaceutical industry continuously seeks robust manufacturing routes for critical anticoagulant therapies, particularly for direct thrombin inhibitors like dabigatran etexilate. A pivotal advancement in this domain is detailed in patent CN108273523B, which discloses a highly efficient production method for a key dabigatran intermediate. This technology addresses long-standing challenges in catalytic hydrogenation by introducing a tungsten disulfide-modified Pt/Ni bimetallic high-dispersion nano catalyst. Unlike conventional noble metal systems that suffer from rapid deactivation or require hazardous high-pressure conditions, this novel catalytic architecture enables the reduction of nitro groups to amino groups with exceptional precision. The process operates under mild hydrogen pressures ranging from 0.3 to 0.5 MPa, achieving nearly 100% substrate conversion while maintaining product selectivity above 97%. For global supply chain leaders and R&D directors, this represents a significant leap forward in process safety and purity control, ensuring a reliable supply of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-[(3-amino-4-methylamino benzoyl)(pyridin-2-yl)amino]ethyl propionate has relied on methods fraught with operational inefficiencies and environmental burdens. Traditional routes often employ palladium on carbon (Pd/C) for catalytic hydrogenation, a process that typically necessitates high-pressure equipment and is prone to catalyst poisoning by sulfur or other impurities present in the reaction matrix. Alternative chemical reduction methods utilizing zinc powder generate massive quantities of heavy metal wastewater, creating severe disposal challenges and escalating environmental compliance costs. Furthermore, many existing protocols result in the target product forming as an viscous oil rather than a free-flowing solid. This physical state complicates isolation, necessitates complex chromatographic purification, and hinders the ability to achieve the stringent purity specifications required for active pharmaceutical ingredient (API) manufacturing. These factors collectively drive up the cost of goods sold and introduce significant variability into the supply chain.
The Novel Approach
The methodology outlined in the patent introduces a paradigm shift by utilizing a specialized bimetallic nanocatalyst composed of platinum and nickel, promoted by tungsten disulfide. This unique composition facilitates the reduction reaction under remarkably mild conditions, specifically at temperatures between 40°C and 45°C and low hydrogen pressures. The inclusion of tungsten disulfide acts as a structural promoter, preventing the aggregation of metal particles and maintaining high dispersion throughout the reaction lifecycle. Crucially, this system drives the reaction to completion with almost 100% conversion while suppressing the formation of common by-products such as hydroxylamines and azoxy dimers. Perhaps most importantly for downstream processing, the product precipitates as a high-purity solid upon crystallization, eliminating the need for difficult oil-to-solid conversions and enabling straightforward filtration and drying operations.
Mechanistic Insights into Pt-Ni-WS2 Catalyzed Hydrogenation
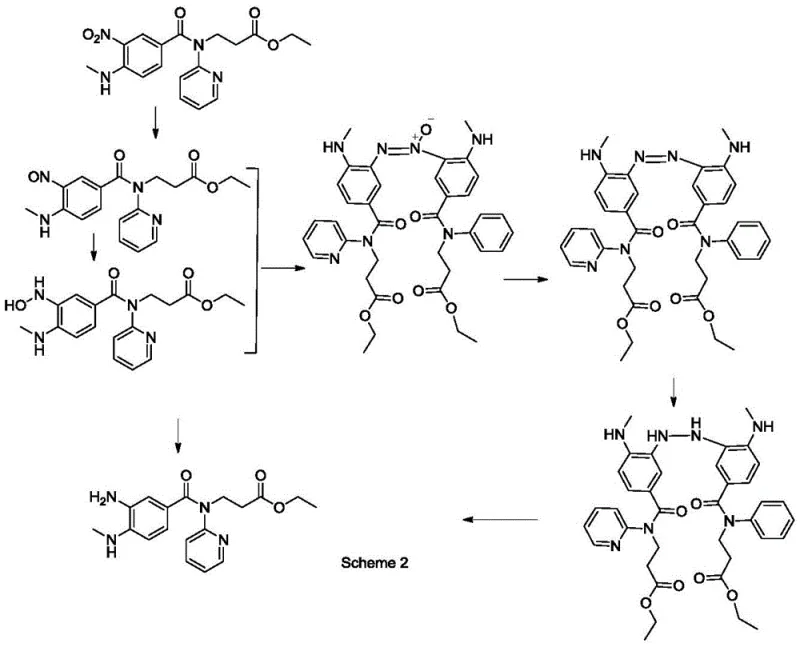
The efficacy of this process lies in the synergistic interaction between the platinum and nickel components within the nano-structured lattice. Platinum serves as the primary active site for hydrogen activation, dissociating molecular hydrogen into reactive species that can attack the nitro group of the substrate. Nickel acts as a secondary catalyst, modifying the electronic environment of the platinum and enhancing the adsorption of the nitro-aromatic substrate. The tungsten disulfide modifier plays a critical role in stabilizing these metal nanoparticles against sintering, a common failure mode in heterogeneous catalysis where particles clump together and lose surface area. This stabilization ensures that the catalyst maintains its high activity over extended reaction times and multiple reuse cycles. The specific solvent system, a mixture of tetrahydrofuran and DMF, further optimizes the solubility of the substrate while facilitating the mass transfer of hydrogen to the catalyst surface, creating an ideal microenvironment for the reduction to proceed rapidly and selectively.
Impurity control is another cornerstone of this mechanistic advantage. In standard nitro reductions, incomplete reduction often leads to the accumulation of hydroxylamine intermediates, which can couple to form stable azoxy dimers that are notoriously difficult to remove. The high selectivity of the Pt-Ni-WS2 system ensures that the reduction proceeds directly to the amine without stalling at these intermediate oxidation states. As illustrated in the reaction pathways, the suppression of these side reactions is vital for meeting regulatory purity standards. The subsequent crystallization step, utilizing n-heptane as an anti-solvent in a controlled heating-cooling cycle, leverages the differences in solubility between the target amine and any trace impurities. This thermodynamic control allows for the growth of large, uniform crystals that exclude impurities from the lattice, resulting in a final product with purity levels exceeding 99%, suitable for direct use in subsequent coupling steps for dabigatran etexilate synthesis.
How to Synthesize Dabigatran Intermediate Efficiently
Implementing this advanced catalytic route requires precise adherence to the preparation of the nanocatalyst and the optimization of the crystallization parameters. The process begins with the synthesis of the catalyst itself, involving the thermal treatment of metal precursors in a high-boiling amine solvent to ensure proper particle size distribution. Once the catalyst is prepared, the hydrogenation is conducted in a standard pressure vessel, but with significantly relaxed safety constraints due to the low operating pressure. The reaction progress is monitored via HPLC to ensure complete consumption of the nitro starting material before proceeding to workup. The critical final stage involves the addition of n-heptane to the reaction filtrate to induce crystallization. Detailed standardized synthetic steps for this procedure are provided below to ensure reproducibility and quality consistency.
- Prepare the catalyst by dispersing platinum acetylacetonate, nickel acetylacetonate, and tungsten disulfide in oleylamine and tetrahydrofuran, followed by high-pressure hydrogen treatment at 200°C.
- Conduct the hydrogenation reaction by mixing the nitro-substrate with the catalyst in a THF/DMF solvent system under 0.3-0.5 MPa hydrogen pressure at 40-45°C.
- Isolate the high-purity solid product by filtering the catalyst and performing a controlled crystallization using n-heptane as an anti-solvent with a specific heating-cooling cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this catalytic technology translates into tangible operational improvements and risk mitigation. The shift from high-pressure hydrogenation to a low-pressure regime reduces the capital expenditure required for specialized reactor vessels and lowers the ongoing maintenance costs associated with high-pressure safety systems. Additionally, the ability to recover and reuse the catalyst multiple times without significant loss of performance drastically reduces the consumption of expensive noble metals, leading to substantial cost savings in raw materials. The generation of a solid product rather than an oil streamlines the logistics of storage and transport, as solids are generally more stable and easier to handle than viscous liquids. These factors combine to create a more resilient and cost-effective supply chain for this critical anticoagulant intermediate.
- Cost Reduction in Manufacturing: The elimination of high-pressure requirements and the recyclability of the catalyst significantly lower both capital and operational expenditures. By avoiding the use of stoichiometric reducing agents like zinc powder, the process also removes the heavy financial burden associated with hazardous waste treatment and disposal. The high yield and selectivity mean that less raw material is wasted, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: The robustness of the catalyst against poisoning ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or off-spec material. The simplified workup procedure, which avoids complex chromatographic separations, shortens the overall manufacturing cycle time. This efficiency allows for faster response to market demand fluctuations and ensures a steady flow of material to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated successfully at the kilogram level with consistent results. The absence of heavy metal waste streams, such as those generated by zinc reduction, aligns perfectly with modern green chemistry principles and strict environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing frameworks.
Q: What are the advantages of the Pt-Ni-WS2 catalyst over traditional Pd/C for this reduction?
A: The Pt-Ni-WS2 catalyst operates effectively at significantly lower hydrogen pressures (0.3-0.5 MPa) compared to traditional methods, prevents catalyst poisoning, and yields a solid product rather than an oil, simplifying downstream purification.
Q: How does this process improve impurity control?
A: The novel catalytic system achieves over 97% selectivity, minimizing the formation of hydroxylamine intermediates and azoxy dimers. Furthermore, the specific crystallization protocol using n-heptane effectively removes residual by-products.
Q: Is the catalyst reusable for commercial scale production?
A: Yes, the patent data indicates that the catalyst can be recovered and reused multiple times (up to 3-4 cycles) with minimal loss in conversion efficiency or selectivity, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving anticoagulant medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic processes described in patent CN108273523B can be seamlessly translated from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance guarantees that the dabigatran intermediates we supply meet the exacting requirements of global pharmaceutical regulators.
We invite you to collaborate with us to optimize your supply chain for anticoagulant drug production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, compliant, and economically efficient source of high-value pharmaceutical intermediates, positioning your organization for success in the competitive cardiovascular therapy market.
