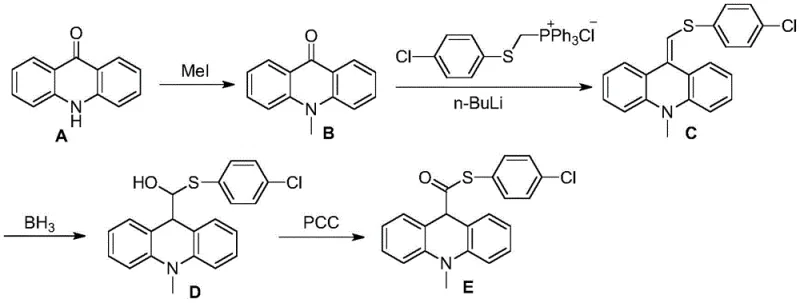
Advanced Manufacturing of APS-5 Key Intermediate via Novel Wittig Strategy
The landscape of chemiluminescent diagnostics is constantly evolving, driven by the demand for substrates with ultra-high sensitivity and stability. Patent CN109928926B, published in late 2022, introduces a significant technological breakthrough in the preparation of the key intermediate for APS-5, a critical acridine dihydrochemiluminescent substrate used in alkaline phosphatase labeling. This specific intermediate, 10-methyl-9,10-acridan-9-thiocarboxylic acid-4-chlorophenyl ester, serves as the foundational building block for generating the light-emitting species upon enzymatic hydroysis. The patent discloses a novel synthetic pathway that fundamentally reimagines the construction of the sulfur-containing side chain, moving away from cumbersome traditional methods towards a more elegant Wittig-based strategy. For R&D directors and procurement specialists in the diagnostic reagent sector, this innovation represents a pivotal shift towards more robust and scalable manufacturing processes. By leveraging acridone as a readily available starting material and employing a strategic sequence of N-methylation, olefination, and controlled oxidation, the inventors have established a route that promises not only chemical efficiency but also substantial operational advantages in a commercial setting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical acridine derivative has been plagued by inefficiencies inherent in earlier methodologies, such as those described in CN1312252A and CN104230804A. Traditional Route 1 often relies on starting with acridine-9-carboxylic acid, necessitating a difficult thioesterification step followed by reduction and methylation, a sequence that frequently suffers from poor atom economy and harsh reaction conditions. Alternatively, Route 2 attempts to utilize acridone but involves a convoluted series of transformations including nucleophilic addition, dehydration, reduction of an olefin to an alcohol, and subsequent oxidation, creating multiple opportunities for yield loss and impurity generation. These legacy processes often require stringent control over multiple distinct reaction environments, increasing the complexity of scale-up and the burden on waste management systems. Furthermore, the use of aggressive reagents in early stages can compromise the integrity of the sensitive acridine core, leading to challenging purification scenarios that drive up production costs and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN109928926B streamlines the entire synthetic architecture by introducing the sulfur-containing aryl fragment through a highly selective Wittig reaction. This approach begins with the straightforward N-methylation of acridone to form N-methyl-9-acridone, followed immediately by the coupling with a specialized phosphonium salt to install the requisite side chain in a single, high-yielding step. This strategic pivot eliminates the need for early-stage thioesterification or complex nucleophilic additions on the carbonyl center, thereby preserving the structural integrity of the molecule throughout the synthesis. The subsequent conversion of the exocyclic double bond to the final ketone functionality is achieved through a mild hydroboration-oxidation sequence followed by a controlled oxidation with PCC, ensuring that the sensitive thioether linkage remains intact. This refined pathway not only reduces the total number of unit operations but also operates under significantly milder conditions, making it inherently safer and more amenable to large-scale commercial production of complex polymer additives and fine chemicals.
Mechanistic Insights into Wittig Olefination and Oxidative Functionalization
The cornerstone of this improved process is the Wittig olefination step, where N-methyl-9-acridone reacts with ((4-chlorophenyl)thio)methyltriphenylphosphine chloride in the presence of a strong base like n-butyllithium. Mechanistically, the low-temperature deprotonation of the phosphonium salt generates a reactive ylide species which then attacks the carbonyl carbon of the acridone derivative. This nucleophilic addition forms a betaine intermediate that collapses to release triphenylphosphine oxide and generate the exocyclic alkene with high stereochemical control. The choice of tetrahydrofuran as the solvent and the precise temperature ramping from -50°C to room temperature are critical for maximizing the conversion while minimizing side reactions such as ylide decomposition or over-alkylation. This step effectively constructs the carbon-sulfur bond framework early in the synthesis, setting the stage for the subsequent functional group manipulations without exposing the thioether moiety to incompatible reagents.
Following the formation of the alkene, the process employs a hydroboration-oxidation protocol to convert the double bond into a secondary alcohol, which is then oxidized to the final ketone. The use of borane-tetrahydrofuran complex allows for anti-Markovnikov addition, although in this symmetric exocyclic context, it primarily serves to hydrate the double bond efficiently. The subsequent oxidation using pyridinium chlorochromate (PCC) in dichloromethane is particularly noteworthy for its selectivity; it oxidizes the benzylic alcohol to the ketone without affecting the sulfide linkage or the aromatic rings. This careful selection of oxidants prevents the formation of sulfoxides or sulfones, which are common impurities in sulfur-containing syntheses and can be detrimental to the performance of the final chemiluminescent substrate. The result is a clean reaction profile that facilitates easier downstream processing and ensures the high purity required for diagnostic applications.
How to Synthesize 10-methyl-9,10-acridan-9-thiocarboxylic acid-4-chlorophenyl ester Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing the target intermediate with high fidelity. The process initiates with the dissolution of acridone in DMF and treatment with sodium hydride, followed by the addition of methyl iodide to secure the N-methyl group. Once the N-methyl-9-acridone is isolated, it undergoes the pivotal Wittig reaction with the chlorophenyl-thio phosphonium salt under inert atmosphere conditions. The resulting alkene is then subjected to hydroboration using borane-THF, followed by oxidative workup with alkaline hydrogen peroxide to yield the alcohol intermediate. Finally, the alcohol is oxidized to the ketone using PCC, and the product is purified via flash column chromatography or recrystallization. For a comprehensive, step-by-step guide including exact stoichiometry, safety precautions, and workup procedures, please refer to the standardized synthesis instructions below.
- N-Methylation of acridone using NaH and methyl iodide in DMF to form N-methyl-9-acridone.
- Wittig reaction between N-methyl-9-acridone and ((4-chlorophenyl)thio)methyltriphenylphosphine chloride using n-BuLi to generate the exocyclic alkene intermediate.
- Hydroboration-oxidation of the alkene intermediate using BH3/THF followed by alkaline hydrogen peroxide to yield the alcohol.
- Oxidation of the alcohol to the final ketone product using PCC in dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. By simplifying the reaction sequence and eliminating the need for multiple distinct transformation types found in legacy routes, the process significantly reduces the consumption of solvents and reagents, leading to a drastic simplification of the manufacturing workflow. This reduction in chemical intensity translates directly into lower raw material costs and decreased waste disposal expenses, addressing the growing pressure for sustainable and cost-effective chemical manufacturing. Furthermore, the use of milder reaction conditions reduces the energy load on the production facility, as there is less requirement for extreme heating or cooling cycles compared to traditional thioesterification methods.
- Cost Reduction in Manufacturing: The streamlined nature of the Wittig-based approach eliminates several unit operations, which inherently lowers the labor and equipment time required per batch. By avoiding the use of expensive or hazardous thioesterification reagents in the initial steps, the process mitigates the risk of costly containment failures and reduces the need for specialized corrosion-resistant equipment. The higher overall yield implied by the fewer steps and milder conditions means that less starting material is wasted, optimizing the cost of goods sold (COGS) for this high-value diagnostic intermediate.
- Enhanced Supply Chain Reliability: The reliance on acridone, a commercially abundant and stable starting material, ensures a secure supply base that is less susceptible to market volatility compared to more exotic precursors. The robustness of the reaction conditions, particularly the tolerance of the Wittig and oxidation steps to standard industrial parameters, enhances the reliability of production schedules. This stability allows for more accurate forecasting and inventory management, reducing the risk of stockouts for critical diagnostic reagent components and ensuring continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents like THF and DCM that are easily recovered and recycled in a modern chemical plant. The avoidance of heavy metal catalysts or extremely toxic reagents simplifies the environmental compliance burden, making it easier to obtain necessary permits for commercial scale-up of complex pharmaceutical intermediates. Additionally, the cleaner reaction profile reduces the load on wastewater treatment facilities, aligning with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this chemiluminescent intermediate. These answers are derived directly from the technical specifications and experimental data provided in patent CN109928926B, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this material into your supply chain or R&D pipeline.
Q: What is the primary advantage of the Wittig-based route for APS-5 intermediate?
A: The primary advantage is the direct installation of the sulfur-containing aryl fragment via a Wittig reaction, which significantly simplifies the synthetic sequence compared to traditional multi-step thioesterification or nucleophilic addition routes, leading to improved overall efficiency.
Q: What are the critical reaction conditions for the key Wittig step?
A: The Wittig reaction requires strict temperature control, initiating at -50°C with n-butyllithium to generate the ylide, followed by gradual warming to room temperature to ensure high selectivity and yield of the exocyclic alkene intermediate.
Q: How does this process impact the purity profile of the final chemiluminescent substrate?
A: By utilizing mild oxidation conditions (PCC) and avoiding harsh thioesterification reagents early in the sequence, the process minimizes side reactions and impurity formation, resulting in a high-purity intermediate crucial for sensitive immunodiagnostic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-methyl-9,10-acridan-9-thiocarboxylic acid-4-chlorophenyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation diagnostic tools. Our team of expert chemists has extensively analyzed the pathway described in CN109928926B and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 10-methyl-9,10-acridan-9-thiocarboxylic acid-4-chlorophenyl ester meets the exacting standards required for immunodiagnostic applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your project's economics. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable supply of this vital chemiluminescent building block.
