Advanced Synthesis of 10-Methyl-9,10-Acridan-9-Thiocarboxylic Acid Ester for Diagnostic Applications
The landscape of chemiluminescent diagnostics is undergoing a significant transformation, driven by the demand for more stable and sensitive substrates like APS-5. At the heart of this technology lies the critical intermediate, 10-methyl-9,10-acridan-9-thiocarboxylic acid-4-chlorophenyl ester. A recent breakthrough detailed in patent CN114213325A introduces a revolutionary preparation method that fundamentally shifts the synthetic paradigm from complex acridone derivatives to a streamlined acridine activation strategy. This innovation addresses long-standing industry pain points regarding operational difficulty and the reliance on hazardous reagents. By leveraging a novel sequence involving 9-position halogenation followed by carbonyldiimidazole activation, the process achieves considerable reaction yields while maintaining exceptional safety standards. For global diagnostic manufacturers, this represents not just a chemical improvement, but a strategic supply chain advantage, ensuring reliable access to high-purity chemiluminescent substrate intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital intermediate has been plagued by inefficient and hazardous pathways that struggle to meet modern GMP and environmental standards. Traditional Route 1, as disclosed in CN1312252A, relies on acridine-9-carboxylic acid, necessitating thioesterification followed by reduction and methylation, which often results in poor control over the reduction step. More problematic are Routes 2 and 3, which utilize acridone as the starting material. These methods involve arduous sequences including N-methylation, nucleophilic addition, dehydration, and subsequent reduction-oxidation cycles. Specifically, Route 2 requires converting an alcohol to a ketone or thioester through multiple redox manipulations, while Route 3 employs Wittig reactions that generate substantial phosphine oxide waste. As illustrated in the reaction schemes below, these pathways are characterized by their structural complexity and the necessity for harsh reaction conditions.
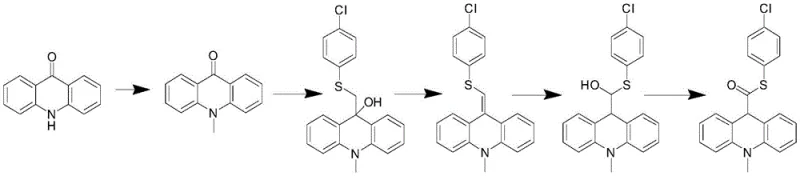
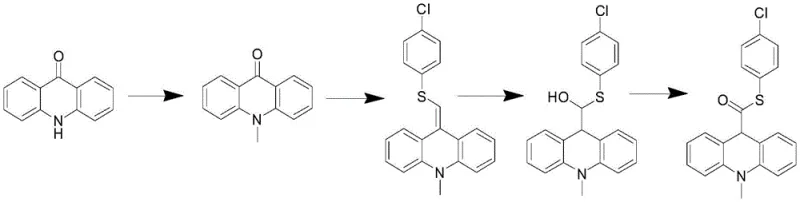
The reliance on acridone derivatives introduces significant bottlenecks, as the functionalization of the 9-position is inherently difficult without resorting to aggressive reagents. Furthermore, the use of highly toxic chemicals in these legacy processes poses severe risks to operator safety and complicates waste disposal, leading to increased operational costs and regulatory scrutiny. The multi-step nature of these conventional methods also accumulates impurities, making the final purification of the chemiluminescent substrate intermediate a costly and yield-limiting endeavor. Consequently, procurement teams have faced challenges in securing consistent, high-quality supplies due to the fragility of these complex manufacturing processes.
The Novel Approach
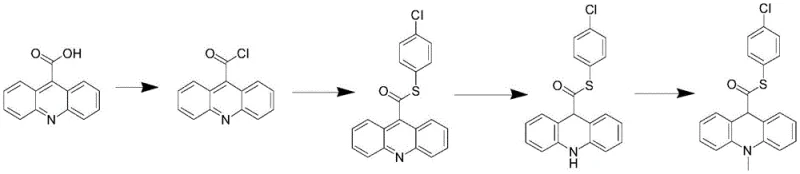
In stark contrast, the methodology presented in CN114213325A offers a streamlined alternative that bypasses the inherent limitations of acridone chemistry. This novel approach initiates with the direct halogenation of acridine at the 9-position using N-bromosuccinimide, a reagent known for its selectivity and ease of handling. The resulting 9-bromoacridine is then activated via a Grignard-type mechanism using magnesium and iodine, followed by coupling with N,N'-carbonyldiimidazole (CDI). This activation strategy creates a highly reactive intermediate that readily undergoes thioesterification with p-chlorothiophenol under mild conditions. By decoupling the ring reduction and N-methylation to the final stages, the process ensures that the sensitive thioester bond is formed on a stable aromatic scaffold before reduction. This logical sequencing drastically simplifies the operation, eliminating the need for complex dehydration or oxidation steps found in prior art.
Mechanistic Insights into Acridine Activation and Thioesterification
The core innovation of this synthesis lies in the unique activation of the 9-position of the acridine ring, which serves as the pivot point for constructing the thiocarboxylic acid ester motif. The mechanism begins with the formation of 9-bromoacridine, where the electron-deficient nature of the acridine ring facilitates selective substitution. Subsequent treatment with magnesium turnings and a catalytic amount of iodine in tetrahydrofuran generates an organomagnesium species in situ. This transient species reacts immediately with CDI to form an acyl-imidazole intermediate, a potent acylating agent. When p-chlorothiophenol is introduced, a nucleophilic attack occurs at the carbonyl carbon, displacing the imidazole leaving group and establishing the crucial carbon-sulfur bond. This thioesterification step is remarkably efficient, proceeding at room temperature without the need for exotic catalysts or extreme temperatures.
Following the formation of the thioester, the aromatic acridine system is converted to the dihydroacridine (acridan) structure through a controlled reduction. The use of zinc powder in the presence of acids such as acetic acid provides a mild yet effective reducing environment that selectively targets the heterocyclic ring without compromising the thioester linkage. This selectivity is paramount, as stronger reducing agents could cleave the sulfur-carbon bond. Finally, N-methylation is achieved using methyl trifluoromethanesulfonate, a powerful methylating agent that ensures quantitative conversion of the secondary amine to the tertiary amine. This mechanistic pathway not only maximizes yield but also minimizes the formation of side products, resulting in a superior impurity profile for the final chemiluminescent substrate intermediate.
How to Synthesize 10-Methyl-9,10-Acridan-9-Thiocarboxylic Acid Ester Efficiently
Implementing this advanced synthesis requires precise control over reaction parameters, particularly during the activation and reduction phases. The process is designed to be robust, allowing for the direct use of crude intermediates in subsequent steps, which significantly reduces solvent consumption and processing time. Operators should focus on maintaining strict temperature control during the initial bromination (0-10°C) to prevent poly-halogenation. The activation step involving magnesium requires careful initiation, typically aided by iodine, to ensure complete formation of the organometallic species. Detailed standardized operating procedures for this high-efficiency pathway are outlined below.
- Brominate acridine using N-bromosuccinimide (NBS) at 0-10°C to obtain 9-bromoacridine.
- Activate 9-bromoacridine via magnesium and iodine in THF, followed by reaction with N,N'-carbonyldiimidazole (CDI).
- React the activated intermediate with p-chlorothiophenol to form the thioester linkage.
- Reduce the acridine ring using zinc powder under acidic conditions (e.g., acetic acid).
- Perform N-methylation using methyl trifluoromethanesulfonate to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio; by shifting from specialized acridone derivatives to commodity acridine and standard reagents like NBS and CDI, the supply chain becomes far more resilient to market fluctuations. This transition effectively mitigates the risk of raw material shortages that often plague complex fine chemical manufacturing. Furthermore, the elimination of highly toxic chemicals reduces the regulatory burden and associated compliance costs, allowing for faster batch release and smoother audits. The operational simplicity of the new route also implies a lower barrier to scale-up, enabling suppliers to respond more agilely to surges in demand for diagnostic reagents.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the reduction in unit operations and reagent costs. By removing the need for expensive Wittig reagents and complex oxidation steps, the overall cost of goods sold is significantly lowered. The ability to use crude intermediates without extensive purification between steps further drives down solvent and energy consumption. Additionally, the high yields reported in the patent examples, such as the 98% yield in the final methylation step, ensure that raw material utilization is maximized, minimizing waste disposal costs. This efficiency creates a competitive pricing structure for the high-purity chemiluminescent substrate intermediate, offering substantial cost savings in diagnostic reagent manufacturing.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of any successful supply chain, and this synthesis method enhances it by relying on widely available starting materials. Acridine and p-chlorothiophenol are established commodities with stable global supply networks, unlike the specialized precursors required for acridone-based routes. The robustness of the reaction conditions means that production is less susceptible to minor variations in utility supply or environmental conditions, ensuring consistent output. This stability allows for longer-term supply contracts and reduces the lead time for high-purity chemiluminescent substrate intermediates, providing downstream manufacturers with the predictability they need for their own production planning.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this route is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The avoidance of hazardous reagents simplifies the engineering controls required for large reactors, reducing capital expenditure on specialized containment systems. Moreover, the generation of less hazardous waste streams aligns with increasingly stringent environmental regulations, facilitating easier permitting and operation in diverse geographic regions. The use of zinc powder and acetic acid for reduction generates benign byproducts that are easier to treat than the heavy metal waste or phosphine oxides associated with traditional methods, supporting a greener and more sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this critical diagnostic intermediate. These answers are derived directly from the technical specifications and comparative data provided in the patent literature, ensuring accuracy and relevance for industry stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this new supply source into your existing quality systems.
Q: Why is the new acridine activation route superior to traditional acridone-based methods?
A: Traditional methods relying on acridone require complex multi-step sequences including Wittig reactions and harsh oxidations. The new route utilizes direct 9-position substitution on acridine, significantly simplifying the synthetic pathway and avoiding highly toxic reagents.
Q: What are the key safety advantages of this preparation method?
A: The process eliminates the use of highly toxic chemicals often found in older protocols. By employing mild reducing agents like zinc powder and standard activating agents like CDI, the method ensures a safer operational environment for large-scale manufacturing.
Q: How does this method impact the purity profile of the APS-5 intermediate?
A: The stepwise approach allows for better control over impurities, particularly by avoiding the side reactions common in dehydration and oxidation steps of prior art. The use of specific activators ensures high conversion rates, leading to a cleaner crude product that is easier to purify.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Methyl-9,10-Acridan-9-Thiocarboxylic Acid Ester Supplier
As the diagnostic industry continues to evolve, the need for reliable partners who can translate cutting-edge patent chemistry into commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team has thoroughly analyzed the activation-reduction-methylation sequence described in CN114213325A and is fully prepared to implement this superior route. We understand that consistency is key in the world of chemiluminescence, which is why our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for in vitro diagnostic applications.
We invite you to collaborate with us to optimize your supply chain for APS-5 and related substrates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new synthesis method can improve your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of this essential intermediate, ensuring your diagnostic products remain competitive in the global market.
