Advanced Semi-Synthesis of Paclitaxel and Docetaxel via Stereochemical Inversion
The pharmaceutical industry has long recognized Paclitaxel and Docetaxel as cornerstone therapies in oncology, yet their supply chain remains constrained by the complexities of extraction and synthesis. Patent CN100586940C introduces a transformative semi-synthetic methodology that addresses these bottlenecks by leveraging a novel stereochemical inversion strategy. This technology fundamentally shifts the paradigm from attempting difficult direct couplings with cis-configured side chains to a more efficient route utilizing readily available trans-chiral side chains. By docking these trans-side chains with protected 10-deacetylbaccatin (10-DAB) and subsequently inverting the stereocenter, the process achieves high purity and yield suitable for large-scale manufacturing. This approach not only simplifies the synthetic route but also enhances the robustness of the supply chain for these critical active pharmaceutical ingredients (APIs). The detailed reaction pathways outlined in the patent provide a clear roadmap for overcoming the historical challenges associated with taxane semi-synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for Paclitaxel and Docetaxel often rely on the direct coupling of 10-DAB with a side chain that already possesses the correct (2'R, 3'S) stereochemistry. However, the synthesis of this specific cis-configured side chain is notoriously difficult, requiring multiple resolution steps or asymmetric catalysis that drive up costs and reduce overall throughput. Furthermore, direct coupling reactions can suffer from low regioselectivity, leading to impurities that are difficult to remove from the highly functionalized taxane core. The reliance on scarce natural precursors and the complexity of maintaining stereochemical integrity throughout a long linear sequence creates significant vulnerability in the commercial scale-up of complex pharmaceutical intermediates. These conventional methods often struggle to meet the stringent purity specifications required for oncology drugs without incurring prohibitive manufacturing expenses.
The Novel Approach
The methodology disclosed in CN100586940C circumvents these issues by employing a "couple-then-invert" strategy. Instead of struggling to synthesize the difficult cis-side chain, the process utilizes a trans-chiral side chain which is synthetically more accessible and easier to produce in bulk. As illustrated in the general reaction scheme below, the protected 10-DAB is first condensed with this trans-side chain to form a stable intermediate. Subsequently, the stereochemistry at the 2'-position is inverted using specific chemical transformations tailored to the target molecule. This decoupling of side chain synthesis from the final stereochemical requirement allows for greater flexibility and efficiency. By separating the coupling step from the stereochemical definition, manufacturers can optimize each stage independently, resulting in a more robust and scalable process for producing high-value taxanes.
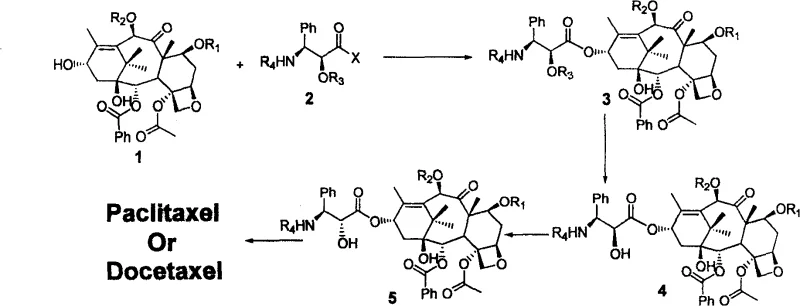
Mechanistic Insights into Stereochemical Inversion and Coupling
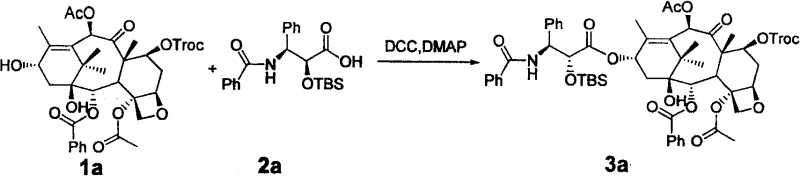
The core of this technological advancement lies in the precise control of stereochemistry during the late stages of synthesis. The initial condensation reaction between the protected 10-DAB (Formula 1) and the chiral side chain (Formula 2) is facilitated by carbodiimide coupling agents such as DCC (dicyclohexylcarbodiimide) in the presence of DMAP (4-dimethylaminopyridine). This step forms the ester linkage at the C13 position of the baccatin core with high efficiency, as evidenced by yields exceeding 86% in the provided embodiments. Following coupling, the protecting group on the side chain hydroxyl (R3) is removed under mild acidic conditions to reveal the free alcohol. This exposure is critical for the subsequent inversion step, which serves as the pivot point for establishing the bioactive conformation of the drug molecule.
For Paclitaxel production, the inversion is achieved through an oxazoline intermediate mechanism. The free hydroxyl group reacts with the adjacent amide carbonyl in the presence of Vilsmeier reagent to form a cyclic oxazoline, which effectively locks the stereochemistry. Subsequent hydrolysis of this oxazoline ring opens it with inversion of configuration, yielding the desired 2'-hydroxyl orientation. In contrast, the Docetaxel route employs a classic Mitsunobu reaction using triphenylphosphine (PPh3) and DEAD (diethyl azodicarboxylate). This powerful transformation allows for the clean inversion of the secondary alcohol with retention of optical purity. The specific reaction pathway for Paclitaxel, detailing the conversion from the coupled intermediate to the final product via these inversion steps, is depicted below, highlighting the chemical precision required for high-purity API intermediate production.
How to Synthesize Paclitaxel and Docetaxel Efficiently
The synthesis protocol described in the patent offers a streamlined approach to generating these complex molecules, minimizing the number of purification steps required between major transformations. The process begins with the careful selection of protecting groups, such as Troc (2,2,2-trichloroethoxycarbonyl) for the 7 and 10 positions of the baccatin core, which are orthogonal to the conditions used for side chain manipulation. The condensation is typically performed in non-polar solvents like toluene at elevated temperatures (30-40°C) to ensure complete conversion. Following the coupling and inversion sequences, the final global deprotection is executed using zinc powder in a mixture of acetic acid and methanol. This reductive step cleanly removes the Troc groups without affecting the sensitive oxetane ring or the ester linkages, delivering the final API in high purity. Detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Condensation of protected 10-deacetylbaccatin (10-DAB) with a novel chiral side chain using DCC and DMAP to form the coupled intermediate.
- Removal of the 2'-hydroxyl protecting group on the side chain under acidic conditions to expose the hydroxyl functionality.
- Stereochemical inversion of the side chain 2'-hydroxyl group using either Vilsmeier reagent (for Paclitaxel) or Mitsunobu reaction (for Docetaxel).
- Final global deprotection of the baccatin core (7-OH and 10-OH) using zinc powder in acetic acid/methanol to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this semi-synthetic route offers substantial strategic benefits by decoupling the availability of the side chain from the complexity of the final assembly. The ability to utilize trans-chiral side chains, which are synthetically simpler and more cost-effective to produce than their cis-counterparts, translates directly into reduced raw material costs. This shift eliminates the need for expensive chiral resolution steps early in the supply chain, thereby stabilizing the supply of critical starting materials. Furthermore, the use of robust coupling reagents and well-understood inversion chemistries ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed reactions or difficult purifications. This reliability is essential for maintaining continuous supply lines for life-saving oncology medications.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of difficult-to-synthesize cis-side chains with easily accessible trans-side chains. By deferring the stereochemical correction to a later stage via efficient inversion reactions, the overall cost of goods sold (COGS) is significantly lowered. The elimination of complex asymmetric synthesis steps for the side chain precursor reduces both material costs and processing time. Additionally, the high yields reported in the patent embodiments indicate minimal material loss during the critical coupling and inversion stages, further enhancing the economic viability of the process for large-scale cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The reliance on trans-chiral side chains mitigates supply risks associated with scarce or difficult-to-source cis-isomers. Since the trans-isomers are easier to synthesize, multiple suppliers can potentially qualify to provide this key starting material, fostering a competitive and resilient supply base. The robustness of the coupling and deprotection steps, which utilize common industrial reagents like DCC and zinc powder, ensures that production is not dependent on exotic or hard-to-procure catalysts. This accessibility of reagents and starting materials drastically simplifies logistics and reduces lead time for high-purity pharmaceutical intermediates, ensuring that manufacturing schedules can be met consistently.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to plant scale. The final deprotection step uses zinc-mediated reduction, which avoids the use of heavy metal catalysts that require complex and costly removal processes to meet regulatory limits. The solvents employed, such as toluene, methanol, and ethyl acetate, are standard industrial solvents with established recovery and recycling protocols, minimizing waste generation. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports sustainable manufacturing practices, making it an attractive option for companies focused on environmental compliance in API production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the described methods. Understanding these details is crucial for technical teams evaluating the integration of this route into existing manufacturing frameworks.
Q: Why is the trans-chiral side chain preferred over the cis-side chain in this synthesis?
A: The patent highlights that synthesizing the trans-chiral side chain is significantly easier and less complex than synthesizing the cis-side chain directly. By coupling the easier-to-make trans-isomer first and then inverting the stereochemistry later in the sequence, the overall process complexity and cost are reduced.
Q: What specific reagents are used for the stereochemical inversion step?
A: The method employs two distinct strategies depending on the target molecule. For Paclitaxel, an oxazoline formation and hydrolysis sequence using Vilsmeier reagent is utilized. For Docetaxel, a Mitsunobu reaction using triphenylphosphine (PPh3) and DEAD is employed to invert the configuration.
Q: How is the final deprotection achieved without damaging the sensitive taxane core?
A: The process utilizes a mild reductive deprotection method using zinc powder in a mixture of acetic acid and methanol. This condition effectively removes the Troc (2,2,2-trichloroethoxycarbonyl) protecting groups at the 7 and 10 positions while maintaining the integrity of the sensitive oxetane ring and ester linkages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel and Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for oncology APIs like Paclitaxel and Docetaxel. Our technical team has extensively analyzed the semi-synthetic routes described in CN100586940C and possesses the expertise to implement these advanced stereochemical inversion strategies at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our facilities are equipped with rigorous QC labs capable of verifying the stereochemical purity and impurity profiles required for stringent purity specifications in the pharmaceutical industry. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to complex synthesis challenges.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized semi-synthetic route can benefit your specific supply chain needs. By leveraging our manufacturing capabilities, you can achieve significant efficiencies and secure a stable source of high-quality taxane intermediates. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
