Advanced Manufacturing of Roflumilast Intermediates via Novel Diacylation Strategy
Advanced Manufacturing of Roflumilast Intermediates via Novel Diacylation Strategy
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments has been significantly shaped by the introduction of Roflumilast, a potent phosphodiesterase-4 (PDE-4) inhibitor approved by the FDA in 2011. As global demand for this critical active pharmaceutical ingredient (API) continues to rise, the efficiency and purity of its synthetic route have become paramount concerns for generic manufacturers and supply chain strategists alike. Patent CN102276522B introduces a transformative methodology that redefines the production of Roflumilast by leveraging a unique diacylation strategy. Unlike traditional approaches that struggle with byproduct management, this invention discloses a method where a specific intermediate compound (I), previously considered a difficult-to-avoid byproduct, is intentionally synthesized in high yield and subsequently converted into the final drug substance. This technical breakthrough not only simplifies the purification process but also establishes a robust framework for industrial-scale production, positioning it as a vital asset for any reliable roflumilast intermediate supplier seeking to optimize their manufacturing portfolio.
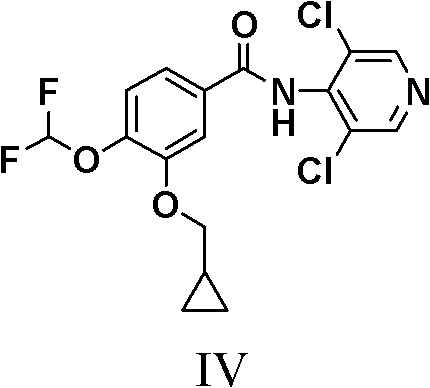
Roflumilast, chemically known as N-(3,5-dichloropyridin-4-yl)-3-cyclopropylmethoxy-4-difluoromethoxybenzamide, represents a complex molecular architecture that demands precise synthetic control. The structural integrity of the final molecule relies heavily on the stability of the amide bond connecting the dichloropyridine ring to the substituted benzoic acid moiety. In the context of large-scale pharmaceutical manufacturing, achieving the stringent purity specifications required for regulatory approval—often exceeding 99.5% assay with tightly controlled impurity profiles—is a formidable challenge. The methodology outlined in CN102276522B addresses these challenges head-on by fundamentally altering the reaction pathway. By shifting the focus from direct amidation to a two-step process involving a stable diacylated intermediate, the process mitigates the risks associated with incomplete reactions and side-product formation, thereby ensuring a consistent supply of high-purity API intermediates for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roflumilast has relied on the direct condensation of 3-cyclopropylmethoxy-4-difluoromethoxybenzoyl chloride with 4-amino-3,5-dichloropyridine. While conceptually straightforward, this conventional route, as described in earlier patents like WO95/01338, presents significant operational hurdles when scaled to industrial levels. A primary limitation is the sensitivity of the reaction to stoichiometry and moisture; even minor deviations can lead to the formation of hydrolysis byproducts such as N-(3,5-dichloropyridin-4-yl)-3-cyclopropylmethoxy-4-hydroxybenzamide. Furthermore, attempts to drive the reaction to completion often require a substantial excess of the amine reactant, which complicates the workup procedure and increases the burden on waste treatment systems. The presence of unreacted starting materials and difficult-to-remove impurities often necessitates multiple recrystallization steps, which erodes overall yield and inflates the cost of goods sold (COGS). These inefficiencies make the conventional route less attractive for cost reduction in pharmaceutical intermediates manufacturing, particularly in a competitive generic market where margin compression is a constant pressure.
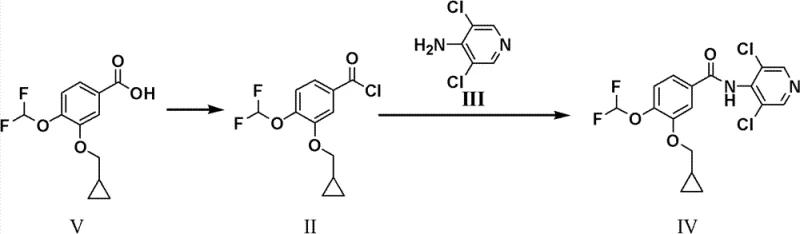
The Novel Approach
In a striking departure from standard practice, the inventors of CN102276522B recognized that the formation of the diacylated byproduct, 4-(N,N-bis(3-cyclopropylmethoxy-4-difluoromethoxybenzoyl))amino-3,5-dichloropyridine (Compound I), could be harnessed rather than avoided. This novel approach deliberately drives the condensation reaction to form this diacylated species almost quantitatively. By utilizing specific reaction conditions—namely the presence of pyridine as an acid binding agent in a polar aprotic solvent like DMF—the process ensures that the amino group on the pyridine ring is fully acylated. This intermediate (I) is remarkably stable and easy to isolate, effectively acting as a "protected" form of the final drug substance. The strategic advantage lies in the subsequent hydrolysis step, where this diacylated intermediate is cleanly converted back to the mono-acylated Roflumilast. This inversion of the problem transforms a liability (the byproduct) into the central pillar of the synthesis, offering a pathway that is inherently more robust and forgiving of minor process variations than the direct amidation route.
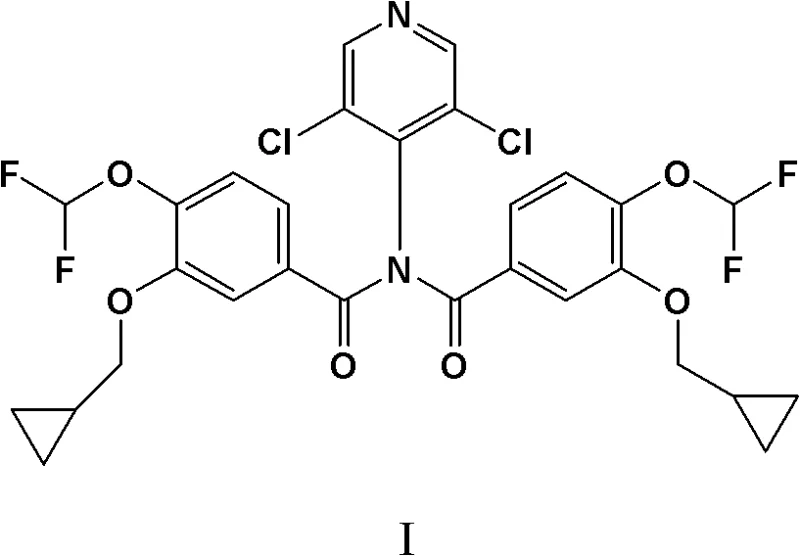
Mechanistic Insights into Diacylation and Selective Hydrolysis
The core of this innovative synthesis lies in the mechanistic interplay between the nucleophilicity of the 4-amino-3,5-dichloropyridine and the electrophilicity of the benzoyl chloride derivative. In the first stage, the reaction between 3-cyclopropylmethoxy-4-difluoromethoxybenzoyl chloride (II) and the amine (III) is facilitated by the basic environment provided by pyridine. The electron-withdrawing chlorine atoms on the pyridine ring reduce the nucleophilicity of the amino group, which typically hinders direct amidation. However, under the optimized conditions of 45°C to 55°C in DMF, the reaction proceeds efficiently to the diacylated stage. The steric and electronic properties of the system favor the formation of the bis-amide structure, preventing the accumulation of the mono-amide until the hydrolysis phase. This controlled progression allows for the isolation of Intermediate (I) with exceptional purity, as evidenced by the sharp melting points and clean NMR spectra reported in the patent examples, providing a solid foundation for the final transformation.
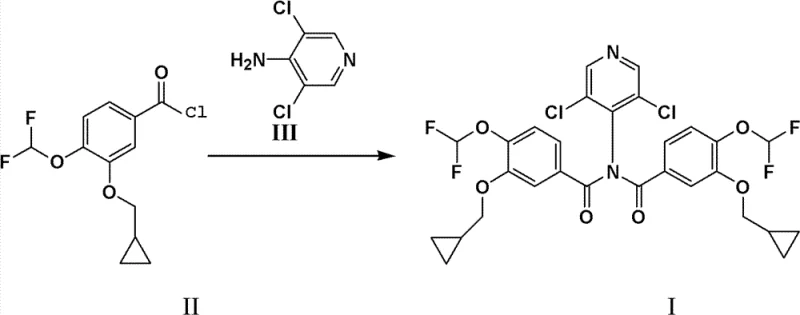
The second mechanistic phase involves the selective hydrolysis of the diacylated intermediate (I) to yield the target Roflumilast (IV). This step exploits the differential stability of the amide bonds under basic conditions. By treating Intermediate (I) with an aqueous alkali solution, such as sodium hydroxide in ethanol, one of the acyl groups is cleaved off, regenerating the starting benzoic acid (V) and leaving behind the desired mono-amide. Crucially, this hydrolysis is highly selective; it does not degrade the sensitive difluoromethoxy or cyclopropylmethoxy groups on the benzene ring. The mechanism allows for a "self-correcting" purification effect: any unreacted acid chloride from the first step is hydrolyzed to the acid, and the diacylated intermediate is converted to the product. This dual outcome ensures that the final crude product is already of high purity, minimizing the need for aggressive chromatographic purification and aligning perfectly with the requirements for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 4-(N,N-bis(3-cyclopropylmethoxy-4-difluoromethoxybenzoyl))amino-3,5-dichloropyridine Efficiently
The preparation of the key diacylated intermediate requires precise control over reaction parameters to maximize yield and minimize side reactions. The process begins with the activation of the benzoic acid derivative using thionyl chloride, a standard yet critical step that must be managed to prevent thermal degradation. Following the formation of the acid chloride, the condensation with the chloropyridine amine is conducted in a mixed solvent system that balances solubility and reactivity. The use of pyridine serves a dual purpose: it acts as a solvent modifier and scavenges the hydrogen chloride generated during the amide bond formation, driving the equilibrium forward. Detailed operational protocols regarding temperature ramps, addition rates, and quenching procedures are essential for reproducibility. For a comprehensive breakdown of the exact molar ratios, solvent volumes, and workup techniques required to achieve the reported 94% yield, please refer to the standardized synthesis guide below.
- Prepare 3-cyclopropylmethoxy-4-difluoromethoxybenzoyl chloride (II) by reacting the corresponding benzoic acid with thionyl chloride in toluene with DMF catalysis at 90°C.
- Condense compound II with 4-amino-3,5-dichloropyridine (III) in DMF using pyridine as an acid binding agent at 45-55°C to form the diacylated intermediate (I).
- Hydrolyze intermediate (I) using aqueous sodium hydroxide in ethanol, followed by pH adjustment to precipitate high-purity Roflumilast and recover the starting benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102276522B offers tangible strategic benefits that extend beyond simple chemical yield. The most significant advantage is the inherent cost reduction in pharmaceutical intermediates manufacturing derived from the recyclability of raw materials. In traditional linear syntheses, unreacted starting materials are often lost in mother liquors or degraded during workup. In contrast, this novel route generates the starting benzoic acid as a co-product of the hydrolysis step. This acid can be precipitated from the filtrate by simple pH adjustment, filtered, and recycled back into the beginning of the process. This closed-loop material flow drastically reduces the net consumption of expensive fluorinated and cyclopropyl-containing building blocks, leading to substantial long-term savings without compromising quality.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of costly purification steps. Because the diacylated intermediate (I) crystallizes readily and the subsequent hydrolysis yields a product with purity exceeding 99%, the need for resource-intensive column chromatography is removed. Furthermore, the recovery of the benzoic acid starting material means that the effective cost per kilogram of the final API is significantly lowered. By converting what was once a waste byproduct into a recoverable asset, the process optimizes atom economy and reduces the financial burden of raw material procurement, making it an ideal candidate for high-volume generic production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on exotic reagents or unstable intermediates. This synthesis utilizes commodity chemicals such as thionyl chloride, pyridine, and common organic solvents like DMF and ethanol, which are readily available from multiple global suppliers. The robustness of the diacylation step means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in reagent quality. This stability ensures that production schedules can be met consistently, reducing the risk of stockouts for downstream formulation partners. Additionally, the ability to store the stable intermediate (I) provides a buffer against supply chain disruptions, allowing manufacturers to build inventory of the key precursor ahead of demand spikes.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to multi-ton production often reveals hidden bottlenecks related to heat transfer and waste management. The exothermic nature of the acylation is well-managed in this protocol through controlled addition rates and the use of high-boiling solvents that facilitate heat dissipation. Moreover, the environmental footprint is minimized by the recycling of the benzoic acid, which reduces the volume of organic waste requiring disposal. The process avoids the use of heavy metal catalysts or hazardous reagents that would trigger strict regulatory scrutiny, simplifying the permitting process for new manufacturing facilities. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its implementation. The following questions address common inquiries regarding the purity profile, scalability, and regulatory implications of this specific manufacturing route. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: How does the diacylation strategy improve purity compared to conventional methods?
A: Conventional methods often struggle with incomplete reactions or hydrolysis byproducts. This novel approach intentionally forms a stable diacylated intermediate (I) which is then selectively hydrolyzed, ensuring product purity exceeds 99% with single impurities below 0.05%.
Q: Can the starting materials be recovered in this process?
A: Yes, a key economic advantage of this patent is the ability to recover 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid from the mother liquor after hydrolysis by acidifying the pH to <3, allowing for recycling and significant raw material cost reduction.
Q: What are the optimal reaction conditions for the condensation step?
A: The patent specifies using DMF as the solvent with pyridine as the acid binding agent. The reaction temperature is optimally maintained between 45°C and 55°C for approximately 5 hours to ensure complete conversion to the diacylated intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in CN102276522B can be reliably replicated on an industrial scale. We understand that for a critical respiratory medication like Roflumilast, consistency is non-negotiable. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, including the critical <0.1% single impurity threshold required for global regulatory filings.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced synthesis technology. Whether you are looking to secure a long-term supply of high-quality intermediates or need assistance in optimizing your own internal processes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal that demonstrates how our manufacturing excellence can drive value for your supply chain.
