Advanced Manufacturing of Roflumilast Key Intermediates via Optimized Alkylation and Oxidation
Advanced Manufacturing of Roflumilast Key Intermediates via Optimized Alkylation and Oxidation
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatment has been significantly shaped by the introduction of Roflumilast, a selective phosphodiesterase 4 (PDE4) inhibitor. Central to the supply chain of this vital medication is the key intermediate, 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid. Patent CN102617340B, published in March 2014, introduces a groundbreaking preparation method that addresses critical bottlenecks in existing manufacturing technologies. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, highlighting its potential to redefine cost structures and supply reliability for global procurement teams. By shifting away from hazardous reagents and complex purification protocols, this methodology offers a robust framework for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry relied on several disparate synthetic routes, each plagued by significant operational deficiencies that hindered efficient mass production. For instance, the method disclosed in WO2005026095 initiates from methyl 3,4-dihydroxybenzoate but suffers from poor regioselectivity during the cyclopropylmethylation step. This lack of control results in a mixture of 3-position, 4-position, and double-alkylated by-products, necessitating rigorous and costly column chromatography for purification, a technique notoriously difficult to translate to multi-ton scales. Similarly, the route described in WO2004033430 requires cryogenic conditions as low as -60°C for bromination and utilizes highly toxic carbon monoxide gas for carbonylation, presenting severe safety hazards and energy consumption challenges. Furthermore, other existing pathways, such as those starting from 3,4-dihydroxybenzaldehyde, often encounter selectivity issues during difluoromethylation, again forcing reliance on chromatographic separation which drastically increases the cost of goods sold (COGS) and extends lead times.
The Novel Approach
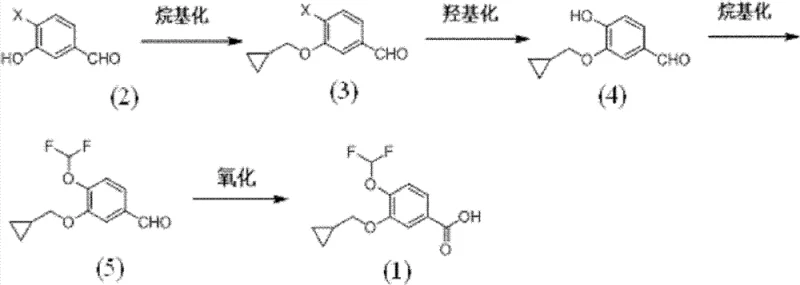
The methodology presented in patent CN102617340B fundamentally re-engineers the synthetic logic to prioritize selectivity and operational simplicity. The process begins with the alkylation of 3-hydroxy-4-halobenzaldehyde, a commercially available or easily accessible starting material, to introduce the cyclopropylmethoxy group with exceptional specificity. This is followed by a metal-catalyzed hydroxylation to install the second oxygen functionality, a step that avoids the harsh conditions of previous nitration-reduction sequences. Subsequent difluoromethylation and final oxidation complete the synthesis. Crucially, every step in this new sequence is designed to yield products that can be purified through standard work-up procedures like extraction and crystallization, completely eliminating the need for column chromatography. This strategic shift not only simplifies the workflow but also ensures that the process is inherently safer and more amenable to continuous manufacturing environments, directly addressing the scalability concerns of modern API production.
Mechanistic Insights into Catalytic Hydroxylation and Oxidation
A deeper examination of the reaction mechanism reveals the sophistication behind the high yields reported in the patent. The pivotal hydroxylation step transforms the aryl halide intermediate into the corresponding phenol using a transition metal catalyst system. The patent details the use of copper salts, such as cuprous iodide, or palladium complexes like Pd(dba)2, in conjunction with specific ligands including 1,10-phenanthroline or 8-hydroxyquinoline. This catalytic cycle facilitates the nucleophilic aromatic substitution under relatively mild thermal conditions ranging from 60°C to 150°C. The choice of base, typically potassium hydroxide or cesium carbonate, plays a critical role in activating the hydroxide nucleophile while maintaining the stability of the sensitive aldehyde moiety. This mechanistic precision ensures that the reaction proceeds with high conversion rates, minimizing the formation of dehalogenated side products that often plague such transformations.
Furthermore, the final oxidation step demonstrates a commitment to green chemistry principles by offering multiple oxidant options. The aldehyde intermediate is converted to the target carboxylic acid using reagents such as sodium hypochlorite, periodic acid, or hydrogen peroxide. Unlike traditional oxidations that might employ heavy metal oxidants like chromium(VI) which generate toxic waste streams, the preferred embodiments utilize hypochlorous acid or periodic acid. These reagents operate effectively at temperatures between 0°C and 50°C, providing excellent control over the exotherm and preventing over-oxidation or degradation of the difluoromethoxy ether linkage. The result is a high-purity final product with yields consistently exceeding 95%, ensuring that the impurity profile remains well within the stringent specifications required for pharmaceutical grade intermediates.
How to Synthesize 3-Cyclopropylmethoxy-4-Difluoromethoxybenzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot plant execution, emphasizing reproducibility and safety. The process is divided into four distinct operational units, each optimized for maximum throughput. The initial alkylation sets the foundation for the molecular architecture, followed by the catalytic installation of the phenolic hydroxyl group. The subsequent difluoromethylation introduces the critical fluorine atoms essential for the biological activity of the final drug, and the sequence concludes with a robust oxidation. For detailed operational parameters, including specific solvent volumes, stirring rates, and quenching procedures, operators should refer to the standardized guidelines below which encapsulate the core technical know-how.
- Alkylate 3-hydroxy-4-halobenzaldehyde with cyclopropylmethyl halide under basic conditions to form the cyclopropylmethoxy intermediate.
- Perform catalytic hydroxylation using copper or palladium catalysts to replace the halogen atom with a hydroxyl group.
- Conduct difluoromethylation on the phenolic hydroxyl group using CHF2Cl or sodium chlorodifluoroacetate.
- Oxidize the resulting aldehyde to the corresponding carboxylic acid using oxidants like sodium hypochlorite or periodic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the downstream processing. By eliminating column chromatography, manufacturers can significantly reduce the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical synthesis. This reduction in material usage directly correlates to a lower environmental footprint and reduced waste disposal costs, aligning with increasingly strict global environmental regulations. Moreover, the avoidance of cryogenic conditions (-60°C) and toxic carbon monoxide removes the need for specialized, expensive reactor infrastructure, allowing production to occur in standard glass-lined or stainless steel vessels found in most multipurpose chemical plants.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the high atom economy and simplified purification. Traditional routes often suffer from yield losses during chromatographic purification, sometimes discarding significant portions of the batch. In contrast, this new method achieves yields of up to 99% in the first step and maintains high efficiency throughout the sequence. The elimination of chromatography means that the process time is drastically shortened, increasing the number of batches that can be produced per year on the same equipment. Additionally, the use of commercially available starting materials like 3-hydroxy-4-iodobenzaldehyde ensures stable raw material pricing, shielding the supply chain from volatility associated with custom-synthesized precursors.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for API manufacturers, and this synthetic route enhances reliability by reducing process complexity. Fewer unit operations mean fewer points of failure and less risk of batch rejection due to out-of-specification impurities. The robustness of the catalytic hydroxylation step, which tolerates a range of ligands and bases, provides flexibility in sourcing raw materials; if a specific ligand becomes scarce, alternatives like 8-hydroxyquinoline can be substituted without compromising the reaction outcome. This flexibility ensures that production schedules remain uninterrupted even when facing minor supply disruptions in the fine chemical market, thereby securing the delivery of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often reveals hidden thermal and safety hazards, but this methodology is explicitly designed for industrial magnification. The reaction temperatures are moderate, and the exotherms are manageable, reducing the risk of thermal runaway incidents. From an environmental perspective, the substitution of toxic reagents with safer alternatives like sodium hypochlorite simplifies wastewater treatment protocols. The ability to recycle solvents such as DMF or ethyl acetate further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only reduces regulatory compliance burdens but also appeals to end-clients who are increasingly prioritizing suppliers with strong environmental, social, and governance (ESG) credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on this superior manufacturing capability.
Q: How does this new method improve purification compared to prior art?
A: Unlike previous methods requiring column chromatography due to poor selectivity, this process utilizes highly selective alkylation and hydroxylation steps, allowing for simple extraction and crystallization purification suitable for industrial scale.
Q: What are the reaction conditions for the hydroxylation step?
A: The hydroxylation is conducted at moderate temperatures between 60-150°C using copper or palladium catalysts with ligands like 1,10-phenanthroline, avoiding the extreme cryogenic conditions (-60°C) found in older patents.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to high yields (up to 99% in early steps), mild reaction conditions, and the avoidance of toxic gases like carbon monoxide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclopropylmethoxy-4-Difluoromethoxybenzoic Acid Supplier
As the global demand for respiratory therapies continues to grow, securing a stable supply of high-quality intermediates is critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN102617340B to deliver superior value. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both clinical trial demands and full-scale commercial requirements seamlessly. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this novel manufacturing process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive down costs and enhance the reliability of your Roflumilast supply chain.
