Advanced One-Pot Synthesis of Sulbactam: Enhancing Purity and Scalability for Global API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational efficiency, particularly for critical beta-lactamase inhibitors like Sulbactam. Patent CN102702228A introduces a transformative one-pot synthesis strategy that fundamentally alters the production landscape for this essential antibiotic adjuvant. By initiating the reaction from 6-aminopenicillanic acid (6-APA) and utilizing a sequential bromination-oxidation-reduction cascade, this method eliminates the cumbersome isolation steps characteristic of legacy processes. The core innovation lies in substituting hazardous elemental bromine with hydrobromic acid and sodium nitrite, thereby generating the reactive brominating species in situ under controlled low-temperature conditions ranging from -15°C to 5°C. This approach not only mitigates severe environmental hazards associated with volatile halogens but also streamlines the workflow into a continuous liquid-phase operation, ensuring that the final Sulbactam solid is obtained with exceptional purity levels exceeding 99.5% HPLC content.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for Sulbactam, often referred to as Route 1 in technical literature, rely heavily on a double-bromination protocol that presents significant logistical and safety challenges for large-scale producers. As illustrated in the reaction scheme below, the conventional pathway necessitates the use of elemental bromine ($Br_2$), a substance known for its extreme toxicity, corrosivity, and difficulty in containment, which imposes rigorous safety protocols and expensive infrastructure requirements on manufacturing facilities. Furthermore, the intermediate products in these older methods frequently precipitate as solids during the reaction, complicating stirring and heat transfer, which inevitably leads to inconsistent reaction kinetics and reduced overall yields. The necessity to isolate and purify these solid intermediates adds multiple unit operations, including filtration and drying, which not only extends the production cycle time but also increases the risk of product degradation and mechanical loss, ultimately inflating the cost of goods sold for the final API intermediate.
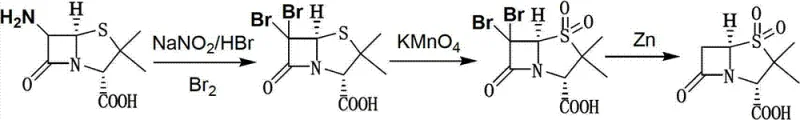
The Novel Approach
In stark contrast, the novel methodology described in the patent data employs a sophisticated single-bromination technique that maintains the reaction mixture in a homogeneous liquid phase throughout the critical transformation stages. By generating the brominating agent in situ through the reaction of sodium nitrite and hydrobromic acid, the process avoids the handling of free bromine entirely, thereby drastically reducing the potential for workplace exposure and environmental release. This "one-kettle" or one-pot design allows for the direct conversion of 6-APA to the final Sulbactam acid solid without the need to isolate the 6-alpha-bromo penicillanic acid or the 6-alpha-bromo sulbactam acid intermediates. The seamless transition from bromination to oxidation using potassium permanganate, and finally to reductive debromination with zinc powder, ensures that the molecular integrity of the sensitive beta-lactam ring is preserved while maximizing atom economy and throughput efficiency.
Mechanistic Insights into Sequential Bromination and Oxidation-Reduction
The chemical elegance of this process is rooted in the precise control of oxidation states and stereochemistry at the C-6 position of the penicillin nucleus. The initial step involves the diazotization of the amino group on 6-APA followed by nucleophilic substitution with bromide ions, occurring efficiently at temperatures between -10°C and 0°C to prevent ring opening or epimerization. Following the formation of the 6-alpha-bromo intermediate, the sulfur atom in the thiazolidine ring is selectively oxidized to the sulfone state using potassium permanganate in a phosphate buffer system, a critical transformation that defines the biological activity of the final Sulbactam molecule. The reaction conditions are meticulously tuned, with pH adjustments to 1.2 using hydrochloric acid and the addition of sodium bisulfite to quench excess oxidant, ensuring that the sensitive beta-lactam moiety remains intact during this aggressive oxidative step.
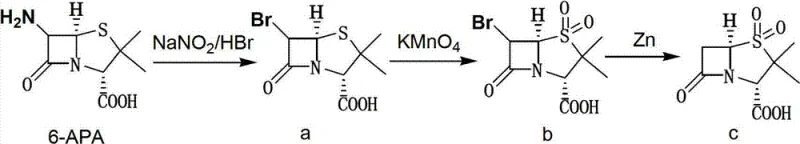
Impurity control is inherently built into the mechanistic design of this one-pot system, primarily through the avoidance of solid-state transitions that often trap impurities within crystal lattices. The final reductive debromination step utilizes zinc powder in an aqueous ethyl acetate system at -5°C to 0°C, which cleanly removes the bromine atom introduced in the first step without affecting the newly formed sulfone group or the beta-lactam ring. The use of zinc powder in batches, combined with simultaneous pH regulation to 3.5-4.0, prevents the accumulation of exothermic heat that could otherwise trigger decomposition pathways. Subsequent workup involving salting out with saturated sodium chloride and extraction with ethyl acetate effectively partitions the organic product from inorganic zinc salts and manganese byproducts, resulting in a crude product that requires minimal recrystallization to achieve pharmaceutical-grade specifications.
How to Synthesize Sulbactam Efficiently
The execution of this synthesis requires strict adherence to thermal profiles and reagent stoichiometry to replicate the high yields reported in the patent examples. Operators must prepare the reaction vessel with water and 48wt% hydrobromic acid, cooling the mixture to below 5°C before introducing ethanol and 6-APA to establish the correct solvent environment for the diazotization reaction. The addition of sodium nitrite solution must be controlled over a period of 3 to 4 hours while maintaining the temperature between -10°C and 0°C, followed by an insulation period of 6 to 8 hours to ensure complete conversion to the bromo-intermediate.
- Perform single bromination of 6-APA using NaNO2 and HBr at -15 to 5°C to form 6-alpha-bromo penicillanic acid.
- Oxidize the intermediate using potassium permanganate at -5 to 5°C to generate 6-alpha-bromo sulbactam acid.
- Execute reductive debromination using zinc powder at -5 to 0°C, followed by crystallization to obtain high-purity Sulbactam solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented one-pot methodology offers substantial strategic advantages by fundamentally restructuring the cost and risk profile of Sulbactam production. The elimination of elemental bromine from the raw material list removes a major hazardous chemical from the supply chain, simplifying logistics, storage, and regulatory compliance requirements associated with transporting and storing volatile halogens. Furthermore, the consolidation of three distinct reaction steps into a single vessel significantly reduces the capital expenditure required for reactor trains, as fewer units are needed to achieve the same annual output, directly translating to lower depreciation costs and a smaller physical footprint for the manufacturing plant.
- Cost Reduction in Manufacturing: The shift from a multi-step isolation process to a continuous one-pot operation drastically reduces the consumption of solvents and energy typically required for drying and re-dissolving solid intermediates. By avoiding the use of expensive palladium catalysts often found in hydrogenation reduction steps and substituting them with cost-effective zinc powder, the direct material costs are significantly optimized. Additionally, the simplified workflow minimizes labor hours and equipment occupancy time, allowing for faster batch turnover and improved asset utilization rates without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Relying on hydrobromic acid and sodium nitrite as primary reagents enhances supply security, as these commodities are more readily available and easier to source globally compared to specialized catalysts or highly regulated elemental bromine. The robustness of the liquid-phase reaction reduces the likelihood of batch failures caused by filtration issues or solid handling errors, ensuring a more consistent and predictable production schedule that can reliably meet the demanding delivery timelines of downstream API manufacturers.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its homogeneous nature, which facilitates efficient heat and mass transfer even in large-scale reactors, mitigating the risks associated with scaling up exothermic reactions. From an environmental perspective, the reduction in hazardous waste generation, particularly the avoidance of bromine vapors and the simplified treatment of aqueous waste streams containing zinc and manganese salts, aligns with increasingly stringent global environmental regulations, reducing the long-term liability and disposal costs for the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this Sulbactam synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is critical for process engineers aiming to adapt this laboratory-scale success to commercial production environments while maintaining strict quality control standards.
Q: How does the new one-pot method improve upon traditional Sulbactam synthesis?
A: The new method replaces the traditional double-bromination process involving hazardous elemental bromine with a single-bromination step using hydrobromic acid. This eliminates the need for intermediate isolation, significantly simplifying the workflow and reducing environmental pollution while maintaining high yields above 82%.
Q: What are the critical temperature controls required for this synthesis?
A: Precise temperature control is vital throughout the process. The initial bromination must occur between -15°C and 5°C, the oxidation step requires -5°C to 5°C, and the final reduction with zinc powder is conducted at -5°C to 0°C to ensure stability and prevent side reactions.
Q: Why is replacing elemental bromine with hydrobromic acid significant for manufacturing?
A: Elemental bromine is highly corrosive, volatile, and difficult to handle safely on an industrial scale. Substituting it with hydrobromic acid drastically improves operational safety, reduces the need for specialized corrosion-resistant equipment, and lowers the overall environmental footprint of the production facility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulbactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies like the one-pot Sulbactam process to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in our manufacturing facilities. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of Sulbactam meets the highest international standards for beta-lactamase inhibitors, providing our partners with a secure and high-quality supply of this essential antibiotic adjuvant.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient production method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of our high-purity Sulbactam into your drug development pipeline.
