Revolutionizing Sulfentrazone Intermediate Production via Isomer-Telescoped Synthesis
Introduction to Patent CN113402472B
The global demand for effective herbicides continues to drive innovation in agrochemical intermediate manufacturing, specifically for difluoromethyl triazolinone derivatives like sulfentrazone. Patent CN113402472B, published in September 2022, introduces a transformative synthetic methodology that fundamentally alters the production landscape for the critical intermediate 1-(2,4-dichlorophenyl)-3-methyl-1H-1,2,4-triazol-5(4H)-one. Unlike conventional processes that rely on the expensive and environmentally burdensome 2,4-dichloroaniline, this invention utilizes a telescoped sequence starting from commodity chlorobenzene. This strategic shift not only mitigates raw material supply risks but also streamlines the reaction pathway by eliminating the need for separating isomeric mixtures at early stages. The structural complexity of the final target, as illustrated below, demands precise control over regioselectivity and purity, challenges which this new protocol addresses through ingenious process design.
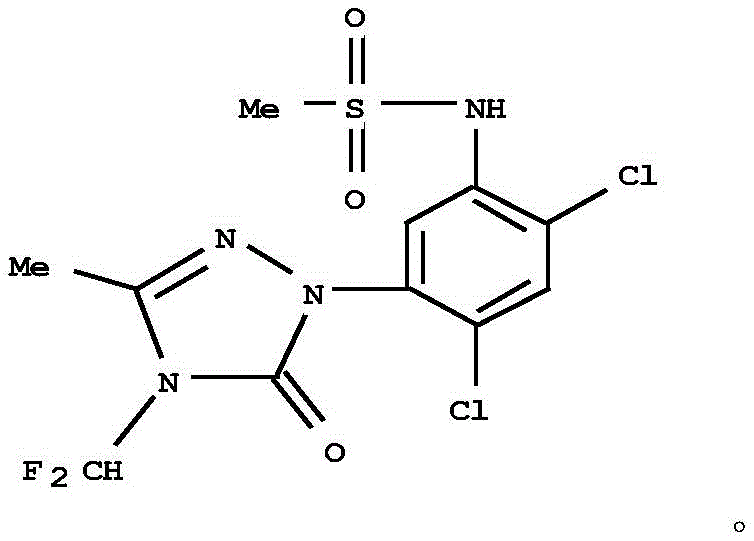
For R&D directors and procurement strategists, understanding the implications of this patent is vital for maintaining competitive advantage in the herbicide market. The ability to synthesize high-purity intermediates without the bottlenecks associated with traditional aniline derivatives offers a compelling value proposition. By integrating catalytic hydrogenation and controlled electrophilic chlorination, the process achieves a total yield reaching 70% with product purity exceeding 95%, setting a new benchmark for efficiency in agrochemical synthesis. This report delves into the mechanistic advantages and commercial viability of this novel route, providing actionable insights for scaling production and optimizing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfentrazone intermediates has been plagued by reliance on 2,4-dichloroaniline as the primary starting material. As depicted in the traditional reaction scheme below, this precursor is typically derived from m-dinitrobenzene chlorination or 2,4-dichlorobenzamide degradation, processes characterized by high production risks and severe environmental pollution. The use of sodium hypochlorite in oxidation steps generates substantial wastewater and hazardous sodium chloride waste, complicating disposal and increasing operational costs. Furthermore, the separation of isomers during the production of 2,4-dichloroaniline is often inefficient, leading to yield losses and limiting the scalability of the entire operation. These factors collectively contribute to volatile pricing and supply instability, making the conventional route less attractive for large-scale commercial manufacturing.
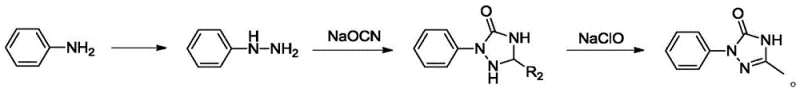
The Novel Approach
In stark contrast, the methodology disclosed in CN113402472B presents a streamlined, five-step sequence that bypasses the need for isolated 2,4-dichloroaniline entirely. The process initiates with the nitration of chlorobenzene to yield a mixture of ortho- and para-chloronitrobenzenes, which are carried forward without separation. This mixture undergoes catalytic hydrogenation to form the corresponding anilines, followed by diazotization and condensation with aldehyde to construct the triazole ring. The final step involves a selective chlorination to install the second chlorine atom at the desired position. This approach, visualized in the comprehensive reaction scheme below, leverages the concept of isomer telescoping to maximize atom economy and minimize unit operations. By deferring separation until the final stages, the process drastically reduces energy consumption and solvent usage, offering a robust alternative for cost reduction in agrochemical intermediates manufacturing.
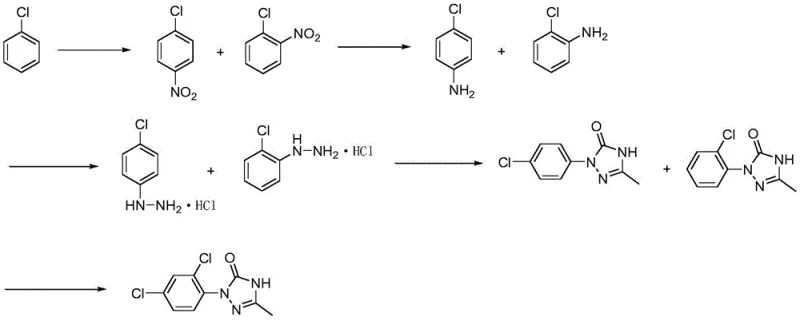
Mechanistic Insights into Telescoped Isomer Synthesis
The core innovation of this patent lies in its ability to manage isomeric mixtures throughout the synthetic sequence without compromising the integrity of the final product. In the initial nitration step, chlorobenzene reacts with a nitrating agent, such as a mixture of fuming nitric acid and sulfuric acid, at controlled temperatures between 20°C and 30°C. This generates a mixture of o-chloronitrobenzene and p-chloronitrobenzene, which is directly subjected to catalytic hydrogenation using Pt/C or Raney nickel. The hydrogenation proceeds smoothly at mild pressures (0.5-0.7 MPa), reducing both nitro groups to amines while preserving the chloro substituents. This tolerance for mixed isomers eliminates the need for energy-intensive fractional distillation after every step, a common bottleneck in fine chemical synthesis. The subsequent diazotization utilizes sodium nitrite and hydrochloric acid in the presence of copper sulfate, converting the aniline mixture into hydrazines which are then condensed with acetaldehyde.
The formation of the triazolinone ring is achieved through cyclization with sodium cyanate followed by oxidation with air or oxygen. This oxidative cyclization is critical for establishing the heterocyclic core, and the use of molecular oxygen as the oxidant represents a greener alternative to stoichiometric metal oxidants. Finally, the regioselective chlorination of the triazole mixture is performed using chlorine gas in the presence of a Lewis acid catalyst like ferric chloride. This step is pivotal, as it converts the mono-chlorinated phenyl ring into the required 2,4-dichloro pattern found in the target intermediate. The mechanism likely involves electrophilic aromatic substitution directed by the existing substituents, ensuring high selectivity. By controlling the reaction temperature between 60°C and 70°C, the process minimizes over-chlorination and side reactions, resulting in a crude product with purity suitable for direct use in downstream herbicide synthesis.
How to Synthesize 1-(2,4-Dichlorophenyl)-3-methyl-1H-1,2,4-triazol-5(4H)-one Efficiently
The practical implementation of this synthesis requires careful attention to reaction conditions, particularly during the nitration and chlorination phases where exothermicity and selectivity are concerns. The patent outlines a robust protocol where solvents like methanol or ethanol are recovered and recycled, further enhancing the economic profile of the process. Operators must ensure strict temperature control during the addition of nitrating agents and chlorine gas to maintain safety and product quality. The detailed standard operating procedures for each of the five steps, including specific molar ratios and workup techniques, are essential for replicating the high yields reported in the examples. For a complete breakdown of the standardized synthesis steps, please refer to the guide below.
- Nitration of chlorobenzene to obtain a mixture of o-chloronitrobenzene and p-chloronitrobenzene without separation.
- Catalytic hydrogenation of the nitro mixture to yield o-chloroaniline and p-chloroaniline.
- Diazotization of the aniline mixture followed by condensation with aldehyde and cyclization to form the triazole ring mixture.
- Final electrophilic chlorination of the triazole mixture using chlorine gas and a Lewis acid catalyst to yield the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain heads looking to optimize their agrochemical portfolios. The primary driver for cost reduction is the substitution of 2,4-dichloroaniline with chlorobenzene, a widely available commodity chemical with a stable global supply chain. This shift removes the dependency on a specialized intermediate that is often subject to environmental regulations and production caps. Additionally, the elimination of intermediate separation steps translates directly into reduced utility costs, as less energy is required for distillation and heating. The ability to recycle solvents and catalysts, such as the Pt/C used in hydrogenation, further contributes to substantial cost savings in raw material consumption. These efficiencies collectively lower the cost of goods sold (COGS), allowing for more competitive pricing in the final herbicide market.
- Cost Reduction in Manufacturing: The telescoped nature of the synthesis significantly reduces the number of unit operations, thereby lowering labor and equipment utilization costs. By avoiding the isolation of unstable or hazardous intermediates, the process also reduces waste disposal fees associated with hazardous byproducts. The use of air or oxygen for oxidation instead of expensive stoichiometric oxidants like sodium hypochlorite eliminates the generation of large volumes of saline wastewater, simplifying effluent treatment and reducing environmental compliance costs. Furthermore, the high atom economy of the route ensures that a greater proportion of raw materials ends up in the final product, minimizing waste generation at the source.
- Enhanced Supply Chain Reliability: Relying on chlorobenzene as the starting material insulates the supply chain from the volatility associated with specialized aniline derivatives. Chlorobenzene is produced on a massive scale for various industrial applications, ensuring consistent availability and price stability. The simplified process flow also reduces the risk of production delays caused by equipment bottlenecks or purification failures. With fewer steps requiring isolation and quality control testing, the overall lead time for batch production is shortened, enabling faster response to market demand fluctuations. This reliability is crucial for maintaining continuous production schedules for downstream herbicide formulations.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard chemical transformations like nitration, hydrogenation, and chlorination, which are well-understood at industrial scales. The avoidance of heavy metal oxidants and the reduction of saline waste streams align with increasingly stringent environmental regulations, facilitating easier permitting and operation in regulated jurisdictions. The ability to operate at moderate temperatures and pressures enhances process safety, reducing the risk of thermal runaways or pressure vessel incidents. This combination of scalability and environmental stewardship makes the technology highly attractive for long-term investment and capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and process descriptions provided in CN113402472B, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is key to evaluating the feasibility of adopting this route for commercial production.
Q: Why is avoiding 2,4-dichloroaniline beneficial for supply chain stability?
A: Traditional routes rely on 2,4-dichloroaniline, which involves hazardous nitration and chlorination steps with high environmental risks and supply constraints. The new method uses commodity chlorobenzene, ensuring consistent availability and lower regulatory hurdles.
Q: How does the isomer-telescoping strategy impact production costs?
A: By carrying ortho- and para- isomers through nitration, hydrogenation, and diazotization without separation, the process eliminates energy-intensive distillation and purification steps, significantly reducing operational expenditure and solvent consumption.
Q: What represents the key quality control checkpoint in this synthesis?
A: The final chlorination step is critical, where the triazole ring mixture is selectively chlorinated to form the 2,4-dichloro pattern. Rigorous control of temperature and catalyst loading ensures high purity (>95%) and minimizes over-chlorinated byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfentrazone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the agrochemical sector. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN113402472B can be successfully translated to industrial reality. We are committed to delivering high-purity sulfentrazone intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex multi-step syntheses with the utmost attention to safety and quality, providing our partners with a secure and reliable source of critical crop protection ingredients.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your agrochemical manufacturing operations.
