Advanced Manufacturing of Mepivacaine Intermediates: A Safer, Scalable Reductive Amination Route
The pharmaceutical landscape for local anesthetics is constantly evolving, driven by the need for safer, more efficient manufacturing processes that comply with stringent global regulatory standards. A pivotal advancement in this domain is detailed in patent CN103073483A, which discloses a novel preparation method for Mepivacaine and its optical enantiomers. This technology represents a significant departure from traditional methylation strategies, utilizing a reductive amination protocol that employs formaldehyde and formic acid. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patent is critical. The disclosed method not only mitigates the safety risks associated with hazardous alkylating agents but also delivers exceptional yields, often exceeding 90%, thereby addressing key pain points in cost reduction in pharmaceutical intermediates manufacturing. By leveraging formic acid as a dual-purpose solvent and reducing agent, the process simplifies the operational workflow, ensuring that the production of high-purity Mepivacaine is both economically viable and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mepivacaine has relied on methylation strategies that pose significant challenges in terms of safety, cost, and impurity profiles. One conventional approach, as illustrated in prior art, utilizes dimethyl sulfate as the methylating agent. While chemically effective, dimethyl sulfate is a potent carcinogen and poses severe occupational health hazards, necessitating expensive containment infrastructure and rigorous waste treatment protocols. Another prevalent method involves the use of iodomethane (methyl iodide). Although capable of producing chiral targets, this route suffers from prohibitively low yields, reported as low as 32% in some literature, primarily due to the formation of unwanted quaternary ammonium salt by-products. Furthermore, iodomethane is a costly reagent, drastically inflating the raw material costs for large-scale production. A third alternative employs sodium cyanoborohydride for reductive amination; however, this reagent is unstable, prone to generating toxic hydrogen cyanide upon contact with acids, and requires strictly anhydrous conditions, complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN103073483A introduces a streamlined Eschweiler-Clarke type reaction that fundamentally reshapes the production economics. By substituting hazardous alkyl halides with inexpensive formaldehyde and formic acid, the process achieves a dramatic improvement in safety and efficiency. The reaction proceeds smoothly at moderate temperatures between 60°C and 100°C, specifically optimized around 90°C to 95°C in the provided examples. Crucially, this method inherently prevents the over-alkylation that leads to quaternary ammonium salts, stopping cleanly at the tertiary amine stage. This selectivity translates directly into higher product purity and simplified purification steps, eliminating the need for complex chromatographic separations often required to remove quaternary impurities. The versatility of this approach allows for the synthesis of both racemic Mepivacaine and its optically pure enantiomers, such as S-Mepivacaine, with excellent stereochemical retention.
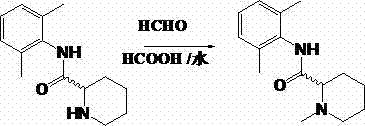
Mechanistic Insights into Formic Acid-Mediated Reductive Amination
To fully appreciate the robustness of this synthetic route, one must delve into the mechanistic underpinnings that drive the transformation of N-(2,6-dimethylphenyl)-2-piperidinecarboxamide into Mepivacaine. The reaction initiates with the condensation of the secondary amine group on the piperidine ring with formaldehyde. In the acidic environment provided by the formic acid solvent, this condensation rapidly generates an reactive iminium ion intermediate. This electrophilic species is the cornerstone of the transformation, poised for nucleophilic attack. Unlike hydride-based reductions that require external reducing agents like sodium borohydride, this system utilizes the formate ion itself as the hydride source. The formate ion transfers a hydride to the iminium carbon, effectively reducing the double bond and establishing the N-methyl group characteristic of Mepivacaine. This intramolecular redox process is highly efficient, as the formic acid is oxidized to carbon dioxide, a gas that easily escapes the reaction mixture, driving the equilibrium towards product formation according to Le Chatelier's principle.
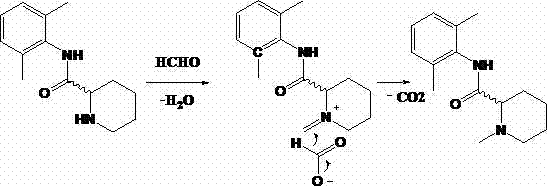
Beyond the primary transformation, the mechanism offers distinct advantages regarding impurity control, a critical metric for R&D Directors focused on quality. The specificity of the formate reduction ensures that the reaction terminates at the tertiary amine stage. In conventional alkylation with methyl iodide, the resulting tertiary amine can act as a nucleophile for a second alkylation event, generating a quaternary ammonium salt that is structurally similar to the product and difficult to separate. In the formic acid system, the tertiary amine product is significantly less nucleophilic towards the iminium intermediate under these specific acidic conditions, effectively self-limiting the reaction. Furthermore, the use of chiral starting materials, such as (S)-N-(2,6-dimethylphenyl)-2-piperidinecarboxamide, ensures that the stereocenter at the 2-position of the piperidine ring remains untouched. The mild acidic conditions do not promote racemization, allowing for the production of S-Mepivacaine with enantiomeric excess (ee) values reaching up to 99.7%, as demonstrated in the patent examples. This mechanistic fidelity is essential for meeting the strict chirality specifications required for modern anesthetic APIs.
How to Synthesize Mepivacaine Efficiently
Implementing this synthesis route in a pilot or commercial plant requires precise control over reaction parameters to maximize the benefits outlined in the patent. The process is remarkably forgiving regarding water content, unlike cyanoborohydride reductions, which simplifies the handling of raw materials like paraformaldehyde or aqueous formaldehyde solutions. The standard operating procedure involves dissolving the piperidine carboxamide precursor in formic acid, followed by the addition of the formaldehyde source. The mixture is then heated to reflux or maintained at 90-95°C for a duration of approximately 8 hours. Workup is straightforward, involving neutralization and extraction, avoiding the need for specialized quenching of dangerous hydrides. For a detailed breakdown of the specific molar ratios, temperature ramps, and isolation techniques validated in the patent, please refer to the standardized synthesis guide below.
- Dissolve N-(2,6-dimethylphenyl)-2-piperidinecarboxamide in anhydrous or concentrated formic acid at ambient temperature (20-25°C).
- Add paraformaldehyde or aqueous formaldehyde solution to the reaction mixture and heat to 90-95°C for approximately 8 hours.
- Quench the reaction with hydrochloric acid, extract with toluene after basification, and purify to obtain Mepivacaine with yields exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the transition to this formic acid-based methodology offers compelling strategic advantages that extend far beyond simple chemistry. The primary driver for adoption is the substantial reduction in raw material costs. By replacing expensive reagents like iodomethane or specialized reducing agents like sodium cyanoborohydride with commodity chemicals like formaldehyde and formic acid, the direct material cost of goods sold (COGS) is drastically lowered. These commodity reagents are produced on a massive global scale, ensuring consistent availability and shielding the supply chain from the volatility often seen with niche fine chemical reagents. Furthermore, the elimination of hazardous reagents reduces the regulatory burden and insurance costs associated with storing and transporting toxic substances, contributing to a leaner operational budget.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the complete removal of high-cost alkylating agents. Iodomethane, previously a standard reagent, is not only expensive but also results in low yields (around 32%), meaning a significant portion of the valuable chiral starting material is wasted. In contrast, the new method achieves yields consistently above 90%, effectively tripling the output per unit of starting material. This efficiency gain, combined with the low cost of formic acid and formaldehyde, results in a significantly reduced cost per kilogram of the final API intermediate. Additionally, the simplicity of the workup—avoiding complex purification steps to remove quaternary salts—reduces solvent consumption and labor hours, further enhancing the overall cost competitiveness of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Formaldehyde and formic acid are bulk industrial chemicals with robust global supply networks, unlike specialized reagents that may have single-source suppliers or long lead times. This ubiquity ensures that production schedules are not disrupted by raw material shortages. Moreover, the process does not require strictly anhydrous conditions, allowing for the use of standard grade solvents and reagents without the need for energy-intensive drying processes. This flexibility simplifies logistics and inventory management, allowing manufacturers to maintain lower safety stocks while ensuring continuous production capability, which is vital for meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior. The avoidance of dimethyl sulfate and cyanide-based reagents eliminates the generation of highly toxic waste streams, simplifying wastewater treatment and reducing the environmental footprint of the facility. The only by-product of the reduction step is carbon dioxide, which vents harmlessly. This green chemistry profile facilitates easier permitting for capacity expansion and aligns with the increasing ESG (Environmental, Social, and Governance) mandates of multinational pharmaceutical companies. The reaction is exothermic but manageable, and the lack of sensitive reagents makes the scale-up from laboratory to multi-ton production straightforward and safe, minimizing the risk of batch failures during technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN103073483A, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and purity. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own supply chains.
Q: Why is the formic acid method superior to dimethyl sulfate for Mepivacaine production?
A: The formic acid method eliminates the use of dimethyl sulfate, a highly toxic and hazardous methylating agent. Additionally, it avoids the formation of quaternary ammonium salt by-products common in iodomethane routes, resulting in higher purity and easier downstream processing.
Q: What are the typical reaction conditions for this reductive amination process?
A: The reaction typically proceeds in formic acid acting as both solvent and reductant, with formaldehyde as the methyl source. Optimal temperatures range from 90°C to 95°C, achieving high conversion rates within 8 hours without requiring anhydrous conditions or expensive catalysts.
Q: Can this process be used for chiral S-Mepivacaine synthesis?
A: Yes, the process is stereospecific. Starting with chiral (S)-N-(2,6-dimethylphenyl)-2-piperidinecarboxamide preserves the optical purity, yielding S-Mepivacaine with ee values up to 99.7% and high isolated yields, making it ideal for enantiopure API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mepivacaine Supplier
The technological advancements detailed in patent CN103073483A underscore the potential for optimizing the production of critical anesthetic intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Mepivacaine intermediate meets the exacting standards required for pharmaceutical applications, providing our partners with absolute confidence in product quality.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis route for your supply chain needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this method can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your strategic partner in the reliable supply of high-quality pharmaceutical intermediates.
