Advanced Green Synthesis of Mepivacaine Intermediates for Commercial Pharmaceutical Production
Advanced Green Synthesis of Mepivacaine Intermediates for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking safer, more efficient pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in patent CN103073484A, which discloses a novel preparation method for mepivacaine and its optical enantiomers. This technology represents a paradigm shift from traditional, hazardous methylation protocols to a greener, base-catalyzed approach utilizing dimethyl carbonate (DMC). For R&D directors and procurement specialists, this innovation offers a compelling value proposition: it replaces toxic reagents like dimethyl sulfate and iodomethane with a benign, cost-effective alternative while maintaining high yields and exceptional stereochemical integrity. As a leading player in the fine chemical sector, understanding the nuances of this patent is essential for optimizing supply chains and ensuring regulatory compliance in the manufacturing of local anesthetics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mepivacaine has relied on methylation strategies that pose significant operational and environmental challenges. Traditional routes often employ dimethyl sulfate or iodomethane as the methylating agent. As illustrated in the background art of the patent, the use of dimethyl sulfate introduces severe toxicity risks, requiring specialized containment equipment and rigorous waste management protocols to handle sulfur-containing byproducts. Furthermore, alternative methods involving sodium cyanoborohydride reduction necessitate strictly anhydrous conditions and present explosion hazards due to the generation of hydrogen cyanide upon contact with acids. These legacy processes not only inflate manufacturing costs through expensive reagent procurement but also create bottlenecks in production throughput due to the complexity of purification and safety monitoring.
The Novel Approach
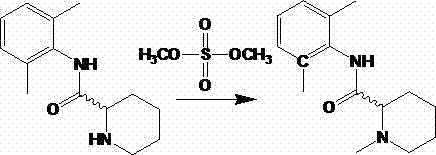
In stark contrast, the methodology outlined in CN103073484A leverages dimethyl carbonate as a sustainable methylating reagent under alkaline catalysis. This approach operates under mild conditions, typically between 30°C and 80°C, eliminating the need for extreme temperatures or pressures. The reaction mechanism is inherently cleaner, producing methanol and carbon dioxide as primary byproducts rather than hazardous sulfur or cyanide waste. By utilizing readily available starting materials such as N-(2,6-dimethylphenyl)-2-piperidinecarboxamide, the process simplifies the supply chain logistics. The transition to this chemistry allows manufacturers to drastically reduce the environmental footprint of their operations while simultaneously improving the economic viability of producing high-purity mepivacaine intermediates.

Mechanistic Insights into Base-Catalyzed N-Methylation
To fully appreciate the robustness of this synthetic route, one must examine the detailed catalytic cycle facilitated by the alkaline environment. The reaction initiates with the deprotonation of the secondary amine nitrogen in the piperidine ring by the base catalyst (B), generating a reactive nitrogen anion. This nucleophile then attacks the carbonyl carbon of the dimethyl carbonate, forming a transient carbamate intermediate and releasing a methoxide ion. Crucially, the methoxide ion acts as a base to regenerate the catalyst by neutralizing the conjugate acid formed in the first step, creating a self-sustaining catalytic loop. Subsequent intramolecular rearrangement and decarboxylation steps lead to the formation of a quaternary ammonium species, which finally decomposes to yield the target N-methylated product, mepivacaine, along with carbon dioxide and regenerated base.
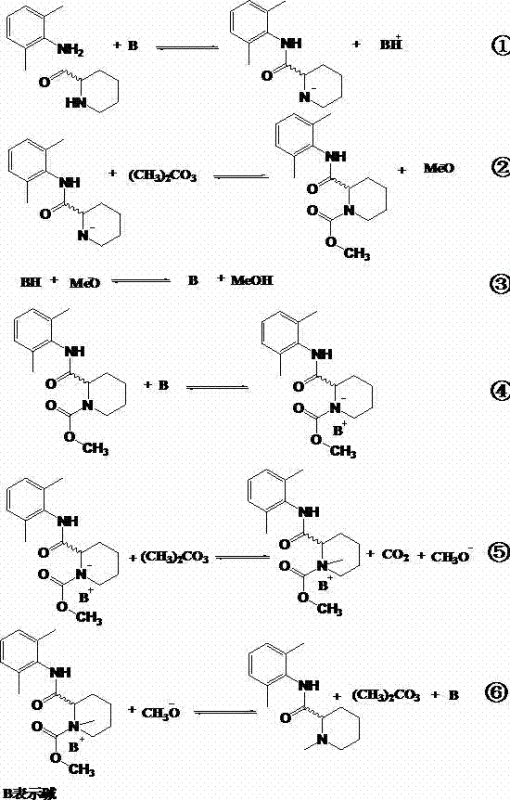
From an impurity control perspective, this mechanism offers distinct advantages over reductive amination or alkylation with halides. The absence of strong reducing agents prevents the formation of over-reduced byproducts or ring-opened impurities often seen with borohydrides. Additionally, the mild basicity ensures that the chiral center at the 2-position of the piperidine ring remains intact, preventing racemization. This is critical for the production of optical enantiomers like S-mepivacaine or R-mepivacaine, where maintaining >99% enantiomeric excess (ee) is a stringent quality requirement. The clean reaction profile minimizes the burden on downstream purification units, allowing for simpler crystallization or extraction protocols to achieve pharmaceutical-grade purity.
How to Synthesize Mepivacaine Efficiently
Implementing this synthesis in a pilot or production setting requires precise control over reaction parameters to maximize yield and minimize side reactions. The patent provides multiple embodiments demonstrating the versatility of the process across different solvents (methanol, ethanol, isopropanol) and bases (NaOH, K2CO3, KOH, basic zeolites). The general procedure involves dissolving the amide precursor in the chosen alcohol solvent, adding the base, and then introducing dimethyl carbonate at controlled temperatures. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical documentation below.
- Dissolve N-(2,6-dimethylphenyl)-2-piperidinecarboxamide in a suitable alcohol solvent (methanol or ethanol) and add an alkaline catalyst such as sodium hydroxide or potassium carbonate.
- Heat the mixture to 30-80°C and slowly add dimethyl carbonate while maintaining stirring to initiate the methylation reaction.
- Maintain the reaction temperature for 1-10 hours, then cool, acidify, remove solvent, and adjust pH to isolate the pure mepivacaine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dimethyl carbonate-based route translates into tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio. Dimethyl carbonate is a commodity chemical with a stable global supply, unlike specialized alkylating agents that may be subject to regulatory restrictions or price volatility. By shifting to this greener chemistry, companies can mitigate supply risks associated with hazardous material transport and storage. Furthermore, the elimination of toxic reagents significantly lowers the barrier for regulatory approval in key markets, accelerating time-to-market for generic formulations of mepivacaine.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous reagents with low-cost dimethyl carbonate. Traditional methods utilizing iodomethane or sodium cyanoborohydride incur high costs not only for the reagents themselves but also for the specialized waste disposal required. By removing the need for heavy metal scavengers or complex sulfur waste treatment, the overall cost of goods sold (COGS) is significantly optimized. Additionally, the high yields reported in the patent embodiments, reaching up to 96%, ensure maximum material utilization, further driving down the unit cost of the final API intermediate.
- Enhanced Supply Chain Reliability: Reliability in the supply of pharmaceutical intermediates is paramount. This synthesis route utilizes robust, non-hazardous chemicals that are easier to source and store in bulk quantities. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity to moisture, which are common pitfalls in anhydrous alkylation processes. Consequently, manufacturers can maintain consistent production schedules and shorter lead times, ensuring a steady flow of high-purity mepivacaine to downstream formulation partners without the interruptions caused by safety incidents or reagent shortages.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this method is inherently designed for sustainability. The byproducts—methanol and carbon dioxide—are manageable and less harmful than cyanide or sulfate wastes. This aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The simplicity of the workup, involving standard acid-base extraction and crystallization, facilitates easy scale-up from laboratory to multi-ton production without the need for exotic reactor configurations. This scalability ensures that the supply chain can flexibly respond to market demand surges while maintaining a strong environmental, social, and governance (ESG) profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in CN103073484A, providing clarity on process feasibility and product quality.
Q: What are the safety advantages of using dimethyl carbonate over dimethyl sulfate?
A: Dimethyl carbonate is a non-toxic, biodegradable green reagent, whereas dimethyl sulfate is highly toxic and carcinogenic. The new process eliminates the severe safety hazards and complex waste treatment associated with sulfur-based reagents.
Q: Can this method preserve the optical purity of chiral mepivacaine enantiomers?
A: Yes, the mild reaction conditions (30-80°C) and the specific mechanism prevent racemization. Patent data confirms that starting with chiral precursors yields enantiomers with high optical purity (e.g., >99% ee).
Q: Is this process scalable for industrial API production?
A: Absolutely. The process uses commercially available, low-cost raw materials and avoids hazardous reagents like sodium cyanoborohydride, making it highly suitable for large-scale commercial manufacturing with simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mepivacaine Supplier
The technological advancements described in patent CN103073484A underscore the potential for more sustainable and efficient production of local anesthetic intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of mepivacaine intermediate meets the highest pharmacopeial standards required by global regulatory bodies.
We invite pharmaceutical partners to leverage our expertise for their next project. Whether you are looking to optimize an existing route or develop a new supply chain for complex intermediates, our team is ready to provide a Customized Cost-Saving Analysis. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
