Revolutionizing Steroid Synthesis: A Deep Dive into Enzymatic High-Purity Cholesterol Manufacturing
Revolutionizing Steroid Synthesis: A Deep Dive into Enzymatic High-Purity Cholesterol Manufacturing
The pharmaceutical and fine chemical industries are witnessing a paradigm shift in the synthesis of critical steroid intermediates, driven by the urgent need for greener, safer, and higher-purity manufacturing processes. Patent CN115678953A introduces a groundbreaking method for preparing high-purity cholesterol through multi-enzyme catalysis, addressing long-standing challenges in stereochemical control and process safety. This technology is particularly vital for the production of injection-grade pharmaceutical excipients, liposomes, and advanced drug delivery systems where impurity profiles are strictly regulated. By leveraging a sophisticated cascade of biocatalysts alongside precise organometallic coupling, this route offers a compelling alternative to traditional, hazardous synthetic pathways. As a leading entity in the sector, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier partnership that guarantees both quality and continuity.
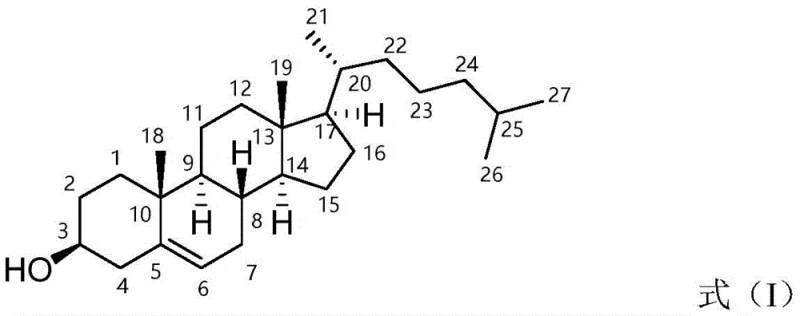
Cholesterol, chemically defined as (3 beta)-cholest-5-en-3-ol, serves as a fundamental building block in modern medicine, yet its synthesis has historically been plagued by inefficiencies. The rigid planar molecule, characterized by its four condensed rings, requires precise functionalization to ensure biological compatibility. The structural integrity shown in Formula I is paramount, as even minor deviations in stereochemistry can render the material unsuitable for sensitive applications like liquid crystals or parenteral formulations. This patent elucidates a dual-route strategy that not only optimizes yield but fundamentally rethinks how we approach the reduction of ketone groups in complex steroid scaffolds, moving away from brute-force chemistry towards elegant biological precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of plant-derived cholesterol has relied heavily on methods that are increasingly untenable in a modern regulatory environment. Traditional routes often involve the use of sodium borohydride for the critical reduction step, a reagent known for its lack of stereoselectivity which frequently generates unwanted 3-alpha hydroxy isomers that are notoriously difficult to separate from the desired 3-beta product. Furthermore, many existing processes depend on high-pressure hydrogenation, a dangerous operation that necessitates specialized infrastructure and carries inherent safety risks regarding explosion and leakage. The reliance on toxic reagents and harsh conditions not only complicates waste treatment but also introduces heavy metal contaminants that require expensive and time-consuming purification steps to meet pharmacopeial standards. Additionally, routes starting from stigmasterol often suffer from long synthetic sequences and limited availability of high-quality starting materials, creating bottlenecks in the supply chain. These cumulative factors result in lower overall yields, higher production costs, and a final product that may struggle to meet the stringent purity requirements of high-value applications like electronic materials or injectables.
The Novel Approach
In stark contrast, the methodology disclosed in CN115678953A presents a transformative solution by integrating multi-enzyme catalysis into the synthetic workflow. This innovative approach replaces hazardous chemical reduction with a highly specific enzymatic system comprising ester hydrolase, 3-beta-steroid dehydrogenase, and glucose dehydrogenase, operating under remarkably mild conditions. The process allows for the effective control of isomer impurities during the reduction phase, ensuring that the stereochemistry at the C3 position is preserved with exceptional fidelity. By avoiding high-pressure hydrogenation and toxic reducing agents, the new route significantly simplifies the downstream processing and reduces the environmental footprint of the manufacturing facility. The flexibility of the patent, which offers two distinct preparation lines (coupling before or after enzymatic reduction), provides manufacturers with strategic options to optimize based on their specific equipment and raw material availability. This adaptability, combined with the ability to achieve purities exceeding 99.9%, positions this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing while simultaneously elevating product quality.
Mechanistic Insights into Multi-Enzyme Catalytic Reduction
The core brilliance of this patent lies in its mechanistic execution, particularly the synergistic action of the enzyme cocktail used for the final transformation. The system employs 3-beta-steroid dehydrogenase to catalyze the stereoselective reduction of the ketone group, a step that is traditionally prone to error in chemical synthesis. This enzyme works in tandem with glucose dehydrogenase, which serves to regenerate the essential cofactor NAD+ in situ, driving the reaction equilibrium towards the desired alcohol product without the need for stoichiometric amounts of expensive cofactors. The reaction is conducted in a biphasic or aqueous-organic system at a controlled temperature of 28-32°C and a pH of 6.8-7.5, conditions that are gentle enough to preserve the integrity of the sensitive steroid backbone while maximizing enzymatic activity. This biological precision ensures that the formation of the 3-alpha isomer is virtually eliminated, solving a persistent purification headache that has plagued the industry for decades. The result is a clean reaction profile that minimizes the generation of by-products, thereby reducing the load on crystallization and filtration units.
Complementing the biocatalytic step is the robust organometallic coupling strategy used to construct the cholesterol side chain. The process utilizes a lithium tetrachlorocuprate reagent to mediate the coupling of a sulfonylated intermediate with a Grignard reagent derived from bromoisopentane. This copper-lithium mediated coupling is highly efficient and allows for the precise installation of the isoheptyl side chain at the C17 position. The patent details two viable sequences: one where the side chain is coupled prior to enzymatic reduction, and another where the reduction occurs first. Both routes demonstrate high operability and yield, showcasing the versatility of the chemistry. The careful control of temperature during the coupling step, specifically cooling to -20°C to -10°C before adding the cuprate solution, prevents side reactions and ensures high conversion rates. This combination of advanced organometallic chemistry and green biocatalysis creates a hybrid synthetic platform that leverages the strengths of both disciplines to achieve high-purity cholesterol with unmatched efficiency.
How to Synthesize High-Purity Cholesterol Efficiently
Implementing this dual-route synthesis requires a clear understanding of the critical process parameters outlined in the patent to ensure reproducibility and scale-up success. The procedure begins with the activation of the starting di-dehydro alcohol via sulfonylation, followed by the divergent paths of coupling and enzymatic transformation. Each step, from the preparation of the lithium tetrachlorocuprate solution to the final recrystallization, demands precise temperature control and stoichiometry to maintain the high purity profile. The following guide summarizes the operational framework necessary for executing this advanced manufacturing protocol effectively.
- Perform sulfonylation of the starting di-dehydro alcohol using p-toluenesulfonyl chloride to activate the hydroxyl group for subsequent coupling.
- Execute a copper-lithium mediated coupling reaction with a bromoisopentane-derived Grignard reagent to construct the cholesterol side chain.
- Apply a multi-enzyme cascade (ester hydrolase, 3-beta-steroid dehydrogenase, glucose dehydrogenase) to achieve highly stereoselective ketone reduction and ester hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits that extend far beyond simple unit cost metrics. The elimination of high-pressure hydrogenation equipment reduces capital expenditure and lowers the barrier to entry for manufacturing partners, thereby diversifying the potential supplier base and enhancing supply security. Furthermore, the mild reaction conditions and reduced toxicity of reagents lead to simpler waste streams, significantly lowering the costs associated with environmental compliance and hazardous waste disposal. The high specificity of the enzymatic steps means that less material is lost to isomer impurities, directly improving the mass balance and overall throughput of the production line. These factors collectively contribute to a more resilient and cost-effective supply chain for critical steroid intermediates.
- Cost Reduction in Manufacturing: The transition from chemical reduction to enzymatic catalysis removes the need for expensive and hazardous reagents like sodium borohydride and high-pressure hydrogen gas. By operating at ambient pressure and moderate temperatures, the process drastically reduces energy consumption and eliminates the need for specialized high-pressure reactors, leading to substantial operational cost savings. Additionally, the high stereoselectivity minimizes the loss of valuable intermediates to isomeric by-products, effectively increasing the yield per batch and reducing the cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and robust enzymatic systems ensures a stable and continuous production flow, mitigating the risks associated with raw material scarcity. The simplified purification process, which avoids complex chromatographic separations often required to remove isomers, shortens the overall production cycle time and accelerates time-to-market for downstream customers. This reliability is crucial for maintaining the continuity of supply for high-demand applications such as liposomal drug formulations, where interruptions can have significant clinical and commercial impacts.
- Scalability and Environmental Compliance: The green nature of this multi-enzyme process aligns perfectly with global sustainability goals, making it easier to obtain regulatory approvals in environmentally strict jurisdictions. The reduction in toxic waste and the avoidance of heavy metal catalysts simplify the effluent treatment process, allowing for easier commercial scale-up of complex pharmaceutical intermediates. This environmental advantage not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand value of the end-product by associating it with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented cholesterol synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity, safety, and scalability. Understanding these details is key for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: How does the enzymatic route improve purity compared to chemical reduction?
A: Traditional chemical reduction using sodium borohydride often generates difficult-to-remove 3-alpha hydroxy isomers. The patented multi-enzyme system utilizes 3-beta-steroid dehydrogenase, which possesses inherent stereospecificity, effectively suppressing isomer formation and achieving purities exceeding 99.9% without complex chromatographic purification.
Q: What are the safety advantages of this process over conventional hydrogenation?
A: Conventional routes often rely on high-pressure hydrogenation, which poses significant explosion risks and requires specialized, expensive equipment. This novel method operates under mild atmospheric conditions (28-32°C) using biocatalysis, eliminating high-pressure hazards and reducing the need for heavy metal catalyst removal steps.
Q: Is this process scalable for industrial production of liposome excipients?
A: Yes, the process is designed for scalability. It utilizes robust enzymatic reactions and standard organic synthesis steps like sulfonylation and coupling that are easily transferred from laboratory to pilot and commercial scales, ensuring a consistent supply of high-quality cholesterol for drug delivery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholesterol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the multi-enzyme catalytic route described in CN115678953A for producing high-purity cholesterol. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace isomers down to 0.05%, guaranteeing that every batch meets the exacting standards required for injection-grade excipients and electronic materials.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis method can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
