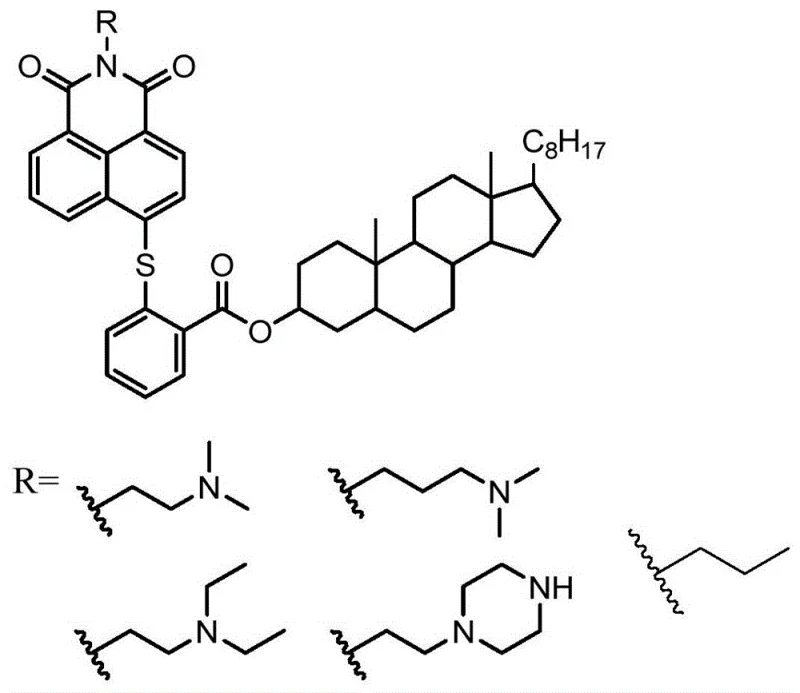
Advanced Synthesis of Thioether-Cholesterol Naphthalimides for Oncology Applications
Advanced Synthesis of Thioether-Cholesterol Naphthalimides for Oncology Applications
The pharmaceutical landscape is constantly evolving towards more targeted and potent anticancer agents, and patent CN109096357B represents a significant leap forward in this domain by disclosing a novel class of naphthalimide derivatives containing both thioether linkages and cholesteryl esters. This technology merges the potent DNA-intercalating capabilities of the naphthalimide scaffold with the lipophilic transport advantages of cholesterol, creating a hybrid molecule designed to penetrate tumor cell membranes with high efficiency. As a leading entity in fine chemical manufacturing, we recognize the immense potential of this synthetic route for developing next-generation oncology therapeutics. The patent outlines a robust, three-step methodology that transforms readily available starting materials into complex bioactive structures, offering a reliable pharmaceutical intermediate supplier pathway for research and development teams globally. By leveraging this specific chemical architecture, manufacturers can access a versatile platform for generating libraries of compounds with tunable anticancer properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of naphthalimide-based anticancer agents often struggles with poor water solubility and non-specific distribution within the body, which limits their therapeutic index and clinical utility. Conventional routes frequently rely on harsh reaction conditions that can degrade sensitive functional groups or result in complex mixtures of regioisomers that are difficult to separate. Furthermore, direct conjugation of large lipophilic groups like steroids to the naphthalimide core without specialized linkers can lead to steric hindrance, reducing the molecule's ability to intercalate into DNA effectively. Many existing processes also utilize toxic heavy metal catalysts or require extreme temperatures that pose significant safety risks and environmental burdens during commercial scale-up of complex pharmaceutical intermediates. These factors collectively increase the cost of goods and extend the timeline for bringing new drug candidates to preclinical evaluation.
The Novel Approach
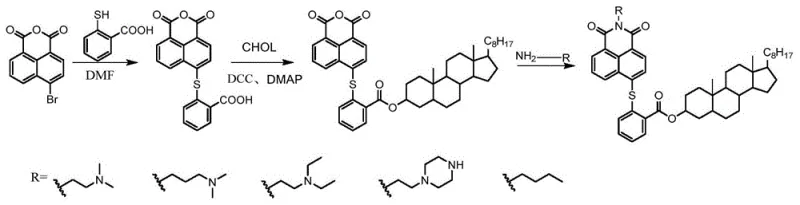
The methodology described in CN109096357B overcomes these hurdles through a modular, stepwise construction that prioritizes mild conditions and high selectivity. The process initiates with a nucleophilic aromatic substitution to install a thioether linker, followed by a Steglich esterification to attach the cholesterol moiety, and concludes with an imidization reaction to finalize the pharmacophore. This strategic sequence ensures that the delicate steroid skeleton remains intact throughout the synthesis, preserving its biological function as a membrane-targeting vector. The use of DCC and DMAP as coupling agents facilitates the esterification at room temperature, drastically reducing energy consumption compared to thermal methods. This novel approach not only simplifies the purification workflow but also enhances the overall yield and purity of the final active pharmaceutical ingredients, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Thioether Linkage and Steglich Esterification
The core of this synthetic innovation lies in the precise control of nucleophilic substitutions and coupling reactions. The first step involves the displacement of the bromine atom on 4-bromo-1,8-naphthalic anhydride by the thiol group of mercaptosalicylic acid in a polar aprotic solvent like DMF at 100°C. This reaction proceeds via an addition-elimination mechanism where the electron-withdrawing anhydride group activates the aromatic ring for nucleophilic attack, resulting in the formation of a stable carbon-sulfur bond. The subsequent esterification utilizes dicyclohexylcarbodiimide (DCC) to activate the carboxylic acid of the intermediate, forming an O-acylisourea species that is highly reactive towards the hydroxyl group of cholesterol. The addition of 4-dimethylaminopyridine (DMAP) acts as a nucleophilic catalyst, accelerating the acyl transfer and preventing side reactions such as N-acylurea formation. This mechanistic precision ensures that the final product retains the structural integrity required for biological activity.
Impurity control is rigorously managed through the selection of specific solvents and purification techniques tailored to the polarity of each intermediate. The patent details the use of thin-layer chromatography (TLC) to monitor reaction progress in real-time, allowing operators to quench reactions immediately upon completion to prevent degradation. Following the esterification step, the removal of dicyclohexylurea (DCU) byproduct is achieved through filtration and careful solvent evaporation, followed by silica gel column chromatography. This purification strategy effectively separates the target cholesterol ester from unreacted starting materials and coupling byproducts, ensuring a high-purity intermediate for the final imidization step. Such attention to detail in the downstream processing is critical for meeting the stringent quality standards required for high-purity pharmaceutical intermediates intended for biological testing.
How to Synthesize Cholesterol-Naphthalimide Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable compounds with reproducible results. It begins with the preparation of the thioether-linked anhydride, proceeds to the conjugation of cholesterol, and finishes with the introduction of various aliphatic amines to modulate solubility and potency. Each step is optimized for yield and ease of execution, utilizing standard laboratory equipment and commercially available reagents. For process chemists looking to implement this route, the detailed conditions regarding temperature, stoichiometry, and workup procedures offer a solid foundation for scaling. The following guide summarizes the standardized operational steps derived directly from the patent examples to facilitate immediate adoption in your R&D facilities.
- React 4-bromo-1,8-naphthalic anhydride with mercaptosalicylic acid in DMF at 100°C for 3 hours to form the thioether-linked naphthalic anhydride intermediate.
- Perform esterification of the intermediate with cholesterol using DCC and DMAP catalysts in dichloromethane at room temperature for 24 hours.
- React the resulting cholesterol ester anhydride with specific aliphatic amines (e.g., N,N-dimethylethylenediamine) in ethanol under reflux to yield the final naphthalimide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial benefits driven by the availability of raw materials and the efficiency of the reaction conditions. The starting materials, including 4-bromo-1,8-naphthalic anhydride and cholesterol, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. The elimination of precious metal catalysts removes the need for expensive scavenging steps and reduces the burden of heavy metal testing in the final product, which translates directly into lower operational costs. Additionally, the ability to perform key coupling reactions at room temperature significantly lowers the energy footprint of the manufacturing process, aligning with modern sustainability goals and reducing utility expenses. These factors combine to create a highly cost-effective production model that enhances margin potential for downstream drug developers.
- Cost Reduction in Manufacturing: The process avoids the use of expensive transition metal catalysts and high-pressure equipment, relying instead on standard organic coupling reagents like DCC and DMAP which are cost-efficient at scale. By eliminating the need for complex metal removal protocols, the downstream processing becomes simpler and faster, reducing labor and material costs associated with purification. The moderate reaction temperatures also mean that standard glass-lined reactors can be used without specialized heating or cooling infrastructure, further driving down capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: Since the synthesis relies on widely available bulk chemicals rather than proprietary or scarce reagents, the supply chain is inherently more resilient to market fluctuations. The modular nature of the synthesis allows for the stocking of key intermediates, such as the thioether-anhydride, enabling a flexible response to demand spikes without restarting the entire process from scratch. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, providing a competitive edge in fast-paced drug development timelines.
- Scalability and Environmental Compliance: The reactions described generate minimal hazardous waste, with the primary byproduct being dicyclohexylurea, which is easily filtered and disposed of according to standard protocols. The use of common solvents like dichloromethane and ethanol facilitates solvent recovery and recycling, minimizing environmental impact and disposal costs. This green chemistry profile simplifies regulatory compliance and permits easier scaling from kilogram to multi-ton production volumes without encountering significant environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these naphthalimide derivatives. The answers are derived from the experimental data and background information provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing pipeline.
Q: What is the primary advantage of incorporating a cholesterol moiety into naphthalimide derivatives?
A: Cholesterol acts as a lipophilic carrier that enhances cell membrane permeability and targets tumor cells more effectively due to the high cholesterol demand of rapidly dividing cancer cells, thereby improving the anticancer efficacy of the naphthalimide DNA intercalator.
Q: How does the thioether linkage contribute to the stability of the compound?
A: The thioether bond formed between the naphthalene core and the salicylic acid linker provides robust chemical stability under physiological conditions while maintaining the planar rigidity required for effective DNA intercalation.
Q: What purification methods are recommended for these intermediates?
A: The patent specifies silica gel column chromatography using gradients of ethyl acetate/methanol or dichloromethane/methanol, ensuring high purity removal of urea byproducts from the DCC coupling and unreacted starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholesterol-Naphthalimide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of cholesterol-naphthalimide derivatives meets the highest industry standards. Our commitment to quality assurance means that you can rely on us for the continuous supply of critical intermediates required for your oncology drug development programs.
We invite you to collaborate with us to explore the full potential of this synthetic technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and project timelines. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your path to market while optimizing your overall production costs.
