Advanced One-Pot Synthesis of Solifenacin Succinate for Commercial Scale-Up and Cost Reduction
Advanced One-Pot Synthesis of Solifenacin Succinate for Commercial Scale-Up and Cost Reduction
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance safety, efficiency, and economic viability, particularly for high-volume urology medications like solifenacin succinate. Patent CN103450183A introduces a transformative approach to synthesizing this critical active pharmaceutical ingredient by leveraging a novel condensation strategy involving substituted phosphine oxides and triphosgene. This methodology fundamentally shifts the paradigm from hazardous, multi-step procedures to a streamlined, one-pot operation that significantly enhances process safety and environmental compliance. By replacing traditional, dangerous reagents with a recyclable phosphine oxide system, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains while maintaining stringent quality standards. The structural integrity of the final product, as depicted below, is preserved with exceptional optical purity, ensuring therapeutic efficacy.
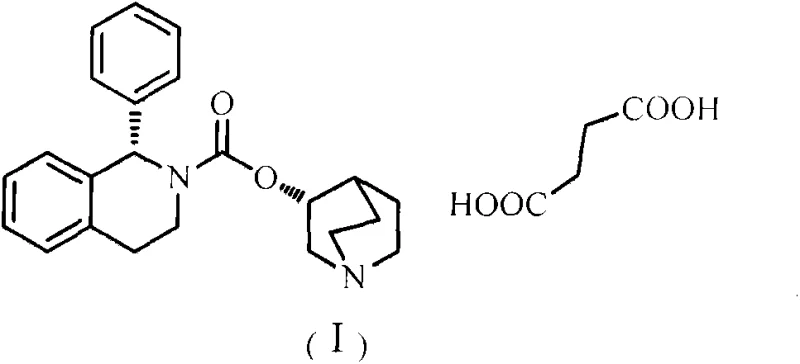
This innovation addresses the critical bottlenecks found in legacy synthetic routes, providing a reliable pharmaceutical intermediates supplier with a distinct competitive edge in the global market. The ability to execute this synthesis under mild conditions without compromising yield represents a significant technological leap forward. For procurement and technical teams, understanding the mechanistic advantages of this route is essential for evaluating long-term sourcing strategies and risk mitigation plans. The following analysis dissects the technical superiority of this patent-protected method, highlighting its potential to redefine cost structures and operational safety in the production of antimuscarinic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of solifenacin free base has relied heavily on methodologies that pose substantial safety risks and operational complexities. For instance, earlier patents such as WO2012001481 describe routes utilizing ethyl chloroformate in conjunction with sodium hydride. This combination is problematic because ethyl chloroformate is a highly toxic substance subject to strict regulatory controls, necessitating elaborate handling protocols and specialized containment infrastructure. Furthermore, sodium hydride is a pyrophoric reagent that ignites spontaneously upon exposure to moisture, creating severe hazards during storage, dosing, and quenching phases on a large scale. These safety concerns are compounded by the relatively low reaction yields, often hovering around 60%, which drastically inflates the cost of goods sold and generates excessive chemical waste. The requirement for refluxing in toluene for extended periods, sometimes exceeding 24 hours, further exacerbates energy consumption and reduces overall plant throughput.
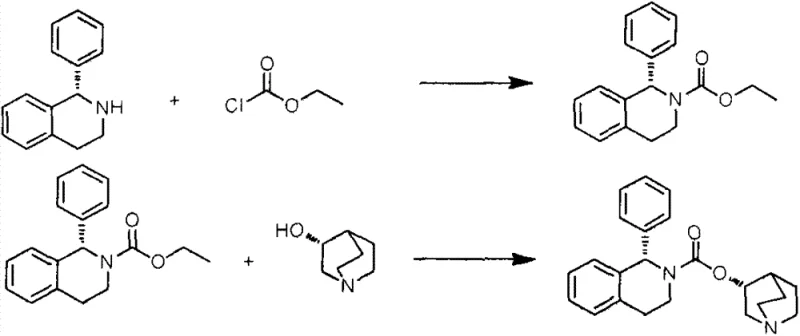
Alternative routes reported in literature, such as those in WO2011/086003A1, attempt to mitigate some issues by using triphosgene but still retain the dangerous sodium hydride step. These processes suffer from cumbersome purification requirements and moderate total yields of approximately 75%. Another approach involving bis(p-nitrophenyl)carbonate generates stoichiometric amounts of p-nitrophenol as a byproduct, which requires multiple extraction and washing steps to remove, leading to significant solvent usage and wastewater treatment burdens. The cumulative effect of these inefficiencies is a manufacturing process that is fragile, expensive, and difficult to scale safely, creating vulnerabilities in the supply chain for this essential medication.
The Novel Approach
In stark contrast, the methodology disclosed in CN103450183A utilizes a sophisticated activation system based on the reaction between solid phosgene (triphosgene) and substituted phosphine oxides, such as triphenylphosphine oxide or tributylphosphine oxide. This innovative combination generates a highly reactive intermediate in situ that facilitates the coupling of S-1-phenyl-1,2,3,4-tetrahydroisoquinoline and R-3-quinuclidinol without the need for hazardous bases like sodium hydride. The reaction proceeds smoothly in a one-pot fashion, where the activation, intermediate formation, and final coupling occur sequentially in the same vessel. This consolidation of steps not only simplifies the operational workflow but also dramatically reduces the cycle time and solvent consumption. The mild reaction conditions, typically ranging from 20°C to 80°C, minimize thermal stress on the equipment and reduce the risk of thermal runaway incidents. Moreover, the phosphine oxide byproduct can be easily separated and recycled back into the process, offering a sustainable loop that drastically cuts raw material expenses.
Mechanistic Insights into Phosphine Oxide Mediated Carbamoylation
The core of this technological breakthrough lies in the unique activation mechanism where the phosphine oxide acts as a nucleophilic catalyst or activator for the triphosgene. Upon mixing triphosgene with the phosphine oxide in toluene at ambient temperatures, a reactive acyl-phosphonium species or similar activated complex is formed. This transient intermediate possesses enhanced electrophilicity, allowing it to react efficiently with the amine group of the tetrahydroisoquinoline derivative to form a stable carbamate intermediate. This activation step is crucial as it bypasses the need for strong deprotonating agents, thereby preserving the stereochemical integrity of the chiral centers in the molecule. The subsequent addition of R-3-quinuclidinol triggers a nucleophilic attack on the activated carbamate, completing the formation of the solifenacin backbone. The entire sequence is kinetically favorable and can be monitored precisely using HPLC or TLC to ensure complete conversion before proceeding to the next stage.
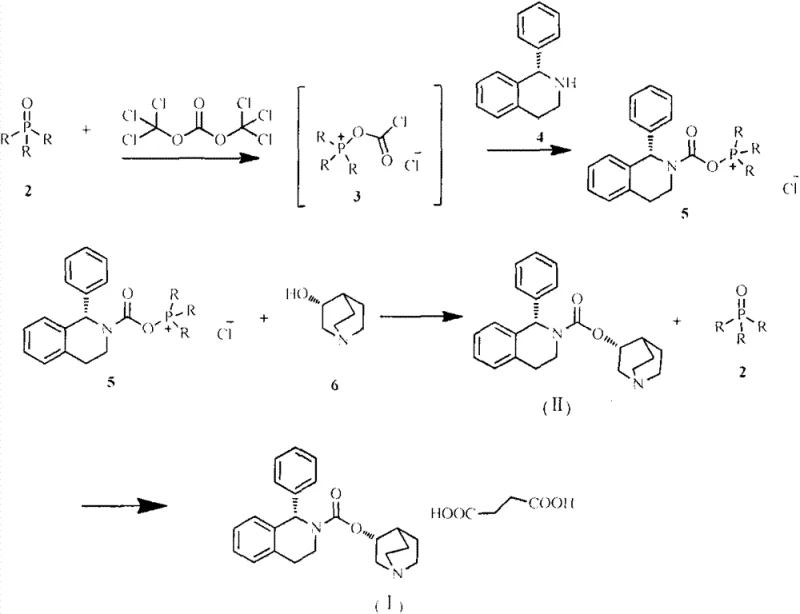
From an impurity control perspective, this mechanism offers superior selectivity. The absence of strong bases prevents epimerization or degradation of the sensitive quinoline and quinuclidine moieties, which are common failure modes in sodium hydride-mediated reactions. The workup procedure is elegantly simple: water is added to quench any remaining reactive species, and the organic phase containing the product and the recoverable phosphine oxide is separated. The aqueous phase is then basified to liberate the free base, which is extracted into fresh toluene. This phase separation strategy effectively isolates the product from the phosphine oxide, which remains in the initial organic layer for recycling. The result is a crude product with purity levels exceeding 98%, requiring minimal downstream purification to meet pharmacopeial standards. This high level of control over the reaction pathway ensures consistent batch-to-batch quality, a critical metric for regulatory compliance.
How to Synthesize Solifenacin Free Base Efficiently
Implementing this advanced synthesis route requires precise control over stoichiometry and temperature profiles to maximize the benefits of the phosphine oxide activation system. The process begins with the preparation of the activating agent by reacting triphosgene with the chosen phosphine oxide in toluene, followed by the sequential addition of the amine and alcohol components. Careful monitoring of reaction progress is essential to determine the optimal endpoints for each stage, ensuring that intermediates are fully consumed before the next reagent is introduced. The final isolation involves a straightforward aqueous workup and crystallization or salt formation step to yield the high-purity succinate salt. For detailed operational parameters, stoichiometric ratios, and specific temperature ramps validated through experimental examples, please refer to the standardized synthesis guide below.
- Activate triphosgene with substituted phosphine oxide (e.g., triphenylphosphine oxide) in toluene at 20-30°C to form the reactive condensing intermediate.
- Add S-1-phenyl-1,2,3,4-tetrahydroisoquinoline to the reaction mixture and stir to form the activated carbamate intermediate in situ.
- Introduce R-3-quinuclidinol to the system, raise temperature to 50-80°C, and complete the coupling to yield solifenacin free base with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phosphine oxide-mediated synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of sodium hydride removes a major safety liability from the manufacturing site, reducing insurance premiums, safety training costs, and the need for specialized inert atmosphere equipment. Furthermore, the ability to recycle the phosphine oxide reagent creates a closed-loop material flow that insulates the production cost from volatile raw material price fluctuations. This process intensification leads to a significant reduction in the overall carbon footprint and waste disposal costs, aligning with modern green chemistry initiatives and corporate sustainability goals. The simplified workflow also shortens the manufacturing lead time, allowing for faster response to market demand spikes and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the recyclability of the phosphine oxide activator. Unlike traditional coupling reagents that are consumed stoichiometrically and generate waste, the phosphine oxide can be recovered from the organic phase and reused in subsequent batches, effectively lowering the variable cost per kilogram of API produced. Additionally, the avoidance of expensive and regulated reagents like ethyl chloroformate reduces procurement complexity and regulatory compliance costs. The higher reaction yields observed in this method mean that less raw material is required to produce the same amount of finished product, directly improving the gross margin profile of the manufacturing operation.
- Enhanced Supply Chain Reliability: By removing reliance on hazardous materials that often face shipping restrictions and supply disruptions, this route ensures a more robust and continuous supply of solifenacin intermediates. The use of common, non-controlled solvents like toluene and readily available starting materials simplifies logistics and reduces the risk of production stoppages due to regulatory hold-ups. The mild reaction conditions also reduce wear and tear on reactor vessels and ancillary equipment, leading to higher asset availability and lower maintenance downtime. This reliability is crucial for maintaining uninterrupted supply to downstream formulation partners and meeting contractual delivery obligations consistently.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis makes it inherently scalable, as it minimizes the number of unit operations and transfer steps required between stages. This reduction in processing steps lowers the potential for human error and cross-contamination, facilitating a smoother transition from pilot plant to commercial scale production. The significant reduction in three wastes (wastewater, waste gas, and solid waste) due to the absence of nitrophenol byproducts and the recycling of reagents simplifies environmental permitting and wastewater treatment requirements. This environmental friendliness positions the manufacturer favorably in regions with strict ecological regulations, ensuring long-term operational license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel solifenacin synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific manufacturing contexts and regulatory environments.
Q: Why is the phosphine oxide method superior to sodium hydride routes for solifenacin?
A: The phosphine oxide method eliminates the use of sodium hydride, a pyrophoric and hazardous reagent that requires strict safety protocols and specialized equipment. Additionally, the phosphine oxide can be recovered and recycled, significantly reducing raw material costs and waste generation compared to traditional methods.
Q: What represents the key impurity control advantage in this new process?
A: By avoiding harsh basic conditions associated with sodium hydride and utilizing a mild one-pot protocol, the formation of side products is minimized. The process allows for easy purification via aqueous workup where the phosphine oxide remains in the organic phase for recovery, yielding solifenacin with purity exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scale-up. It operates under mild temperatures (20-80°C), uses common solvents like toluene, and avoids controlled toxic substances like ethyl chloroformate, making it safer and more economically viable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solifenacin Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, safe, and cost-effective synthetic routes for high-value pharmaceutical intermediates like solifenacin succinate. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the phosphine oxide activation method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for optical purity and chemical identity. Our commitment to process innovation allows us to offer clients a secure supply of high-quality intermediates that support their drug development and commercialization timelines.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, demonstrating our transparency and dedication to fostering long-term, collaborative relationships in the global pharmaceutical supply chain.
