Advanced Solifenacin Succinate Manufacturing: High Purity and Scalable Industrial Routes
Advanced Solifenacin Succinate Manufacturing: High Purity and Scalable Industrial Routes
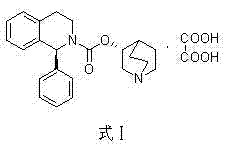
The pharmaceutical landscape for Overactive Bladder (OAB) treatments demands rigorous standards for Active Pharmaceutical Ingredient (API) quality and supply chain stability. Patent CN103896938A introduces a transformative methodology for the preparation of Solifenacin Succinate, chemically known as (3R)-1-azabicyclo[2.2.2]octane-3-yl (1S)-1-phenyl-3,4-dihydroisoquinoline-2-(1H)-carboxylate succinate. This innovation addresses critical bottlenecks in existing manufacturing protocols by leveraging eco-friendly ionic liquids and optimizing reaction stoichiometry to achieve exceptional purity levels exceeding 99.8%. For R&D directors and procurement specialists, this patent represents a pivotal shift away from hazardous, low-yield processes toward a streamlined, industrially viable pathway that ensures consistent supply of this high-value muscarinic M3 receptor antagonist.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Solifenacin Succinate has been plagued by significant economic and technical hurdles that compromise commercial viability. One prominent prior art route, disclosed in WO2007076116, relies on Rs-3-quinuclidinol as a primary starting material. This reagent is prohibitively expensive, costing approximately four to six times more than the alternative tetrahydroisoquinoline precursors used in newer methods. Furthermore, this legacy process suffers from selectivity issues during the reaction of two chlorine atoms with hydroxyl and nitrogen groups, leading to complex byproduct profiles that are difficult to remove. The necessity for extensive purification not only drives up costs but also results in lower final product purity, making it unsuitable for modern GMP standards.
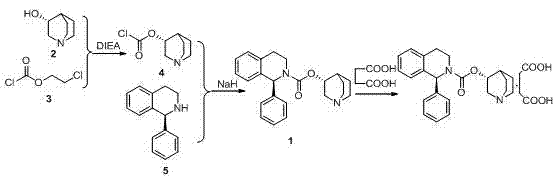
Another conventional approach involves the use of chiral chromatographic columns to separate enantiomers, as described in earlier literature. While this method utilizes cheaper raw materials initially, the reliance on chiral HPLC for resolution introduces massive capital expenditure risks. The equipment requirements are stringent, and the process is inherently difficult to scale for multi-ton production. If a separation fails, the entire batch value is lost, creating unacceptable supply chain volatility. Additionally, other methods utilizing triphosgene or explosive nitro compounds pose severe safety risks and environmental liabilities, further disqualifying them from sustainable industrial application.
The Novel Approach
The methodology outlined in CN103896938A fundamentally reengineers the synthetic pathway to prioritize safety, cost-efficiency, and scalability. By initiating the synthesis with the more economical (S)-1-phenyl-1,2,3,4-tetrahydroisoquinoline, the process immediately reduces raw material costs. The core innovation lies in the utilization of ionic liquids, such as [bmim]BF4, as reaction media. These solvents are not only recyclable, significantly reducing waste discharge, but they also accelerate reaction rates and improve transformation efficiency. The three-step sequence—carbamation, transesterification, and salification—is tightly controlled to minimize isomer impurities, ensuring that the final Solifenacin Succinate meets strict pharmacopeial standards without the need for expensive chiral separation columns.
Mechanistic Insights into Ionic Liquid-Mediated Transesterification
The heart of this advanced synthesis lies in the second step, where the ethyl carbamate intermediate undergoes transesterification with (R)-3-quinuclidinol. In traditional organic solvents, this reaction often requires harsh conditions that degrade sensitive functional groups. However, the use of ionic liquids creates a unique microenvironment that stabilizes the transition state. The reaction is conducted at a precise temperature range of 100~105°C in the presence of sodium hydride and a catalytic amount of DMF. The ionic liquid facilitates the nucleophilic attack of the quinuclidinol hydroxyl group on the carbamate carbonyl carbon, promoting efficient displacement of the ethoxy group. This mechanism is crucial for maintaining the stereochemical integrity of the (1S) and (3R) configurations, which are essential for the biological activity of the drug.
Impurity control is rigorously managed through stoichiometric precision and solvent selection. In the first step, the molar ratio of the tetrahydroisoquinoline derivative to the acid binding agent is maintained between 1:1 and 1:1.5. This prevents over-reaction and the formation of bis-carbamate byproducts. Similarly, in the transesterification step, the ratio of reactants is optimized to 1:1~1.5 to ensure complete conversion while minimizing side reactions. The final crystallization step utilizes a specific volume ratio of ethanol to ethyl acetate (1:4), which selectively precipitates the succinate salt while keeping impurities in solution. This multi-layered approach to purification ensures that the final product consistently achieves an HPLC purity greater than 99.8%, a benchmark that is difficult to reach with older technologies.
How to Synthesize Solifenacin Succinate Efficiently
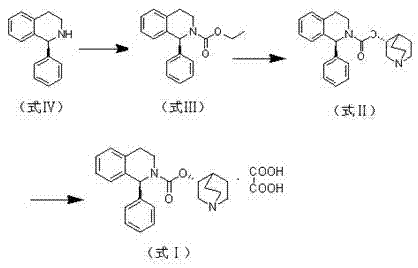
The synthesis of Solifenacin Succinate via this patented route involves a logical progression of carbamate protection, stereoselective coupling, and salt formation. The process is designed to be robust, utilizing readily available reagents and avoiding extreme conditions that complicate scale-up. Detailed below is the strategic overview of the operational parameters required to replicate this high-yield pathway in a pilot or production setting. For the complete standard operating procedure including exact addition rates and workup details, please refer to the technical guide section.
- React (S)-1-phenyl-1,2,3,4-tetrahydroisoquinoline with vinyl chloroformate in ionic liquid or toluene to form the ethyl carbamate intermediate.
- Perform transesterification between the ethyl carbamate and (R)-3-quinuclidinol using sodium hydride in ionic liquid at 100-105°C.
- Salify the resulting base with succinic acid in an ethanol/ethyl acetate mixture, followed by cooling crystallization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-based synthesis offers profound strategic advantages beyond mere technical feasibility. The shift away from expensive chiral starting materials and hazardous reagents directly translates to a more resilient and cost-effective supply chain. By eliminating the dependency on chiral chromatography, manufacturers can drastically reduce capital expenditure on specialized equipment and lower the risk of batch failures. This reliability is critical for maintaining continuous supply to downstream formulation partners, especially in the competitive OAB therapeutic market where demand is steady and growing.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by raw material optimization and process simplification. By substituting the costly Rs-3-quinuclidinol starter with the more affordable tetrahydroisoquinoline derivative, the overall bill of materials is significantly reduced. Furthermore, the ability to recycle ionic liquids minimizes solvent consumption and waste disposal costs. The elimination of chiral column chromatography removes a major cost center associated with high-pressure liquid chromatography resins and energy consumption, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of stable, non-explosive reagents. Unlike routes that rely on zinc ethyl or nitro compounds, which pose storage and transport hazards, the reagents in this process are commercially available and safe to handle. The high yield of each step—often exceeding 90% for intermediates and 80% for the final product—ensures that less raw material is needed to produce the same amount of API. This efficiency buffers the supply chain against raw material price fluctuations and availability issues, providing a more predictable delivery schedule for global clients.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor types without the need for specialized high-pressure or cryogenic equipment. The use of ionic liquids aligns with green chemistry principles, reducing volatile organic compound (VOC) emissions and facilitating easier solvent recovery. This environmental compliance reduces the regulatory burden on manufacturing sites and future-proofs the production facility against tightening environmental regulations. The robust nature of the reaction conditions allows for seamless transition from kilogram-scale development to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Solifenacin Succinate using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality assurances associated with this synthesis route. Understanding these details is essential for technical teams evaluating supplier capabilities and process robustness.
Q: Why is the ionic liquid method superior for Solifenacin Succinate production?
A: The ionic liquid method eliminates the need for expensive chiral chromatography and toxic reagents like triphosgene. It allows for solvent recycling, reduces environmental pollution, and achieves product purity greater than 99.8% with high yields suitable for industrial scale-up.
Q: What are the key cost drivers in traditional Solifenacin synthesis?
A: Traditional routes often rely on expensive starting materials like Rs-3-quinuclidinol, which can be four to six times more costly than tetrahydroisoquinoline derivatives. Additionally, processes requiring chiral separation columns involve high equipment investment and risk of batch failure, significantly driving up manufacturing costs.
Q: Is this synthesis route safe for large-scale manufacturing?
A: Yes, this route avoids explosive nitro compounds and highly flammable organometallic reagents like zinc ethyl found in other methods. The use of stable ionic liquids and controlled temperatures (100-105°C) enhances operational safety and makes the process robust for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solifenacin Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and unwavering quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We are committed to delivering Solifenacin Succinate that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards before it leaves our facility.
We invite you to collaborate with us to optimize your supply chain for this critical OAB medication. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
