Advanced Enzymatic Resolution of C-24 Hydroxy Vitamin D Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust methodologies for the production of high-purity active pharmaceutical ingredients (APIs) and their precursors, particularly when dealing with complex stereochemical challenges. Patent CN100404686C introduces a groundbreaking approach for the separation and isomerization of vitamin D derivative isomers, specifically targeting the critical C-24 hydroxyl branch bond. This patent addresses a longstanding bottleneck in the synthesis of bioactive vitamin D analogs, where the C-24 position acts as a chiral center, resulting in a mixture of C-24R and C-24S hydroxyl isomers. Given that the C-24S configuration typically exhibits superior physiological activity, the ability to efficiently isolate this specific epimer is paramount for drug efficacy. The disclosed technology utilizes selective enzymatic esterification and solvolysis, coupled with a novel recycling mechanism, to achieve high diastereomeric purity without the excessive costs associated with traditional asymmetric synthesis or inefficient chromatographic methods.
Furthermore, the integration of this technology into commercial manufacturing pipelines offers significant advantages for a reliable pharmaceutical intermediates supplier. By shifting from harsh chemical conditions to mild enzymatic processes, manufacturers can reduce energy consumption and minimize the formation of hazardous by-products. The patent details specific embodiments using lipases from Alcaligenes sp. and Pseudomonas sp., demonstrating that these biocatalysts can distinguish between the subtle steric differences at the C-24 position with remarkable precision. This level of selectivity is crucial for meeting the stringent purity specifications required by global regulatory bodies, ensuring that the final vitamin D derivatives are safe and effective for therapeutic use. The method not only simplifies the purification workflow but also enhances the overall sustainability of the production process, aligning with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of C-24 hydroxy vitamin D derivative isomers has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional approaches often relied on asymmetric reduction of C-24 keto groups or direct synthetic coupling of the side chain to the vitamin D skeleton. These methods frequently necessitate严苛 reaction conditions, expensive chiral catalysts, and multi-step protection-deprotection sequences that drive up production costs and extend lead times. Moreover, conventional column chromatography, while theoretically capable of separating isomers, proves practically ineffective for C-24R and C-24S forms due to their nearly identical polarity and minimal structural divergence. This results in poor resolution, low recovery rates, and substantial product loss, making it an unsustainable option for large-scale manufacturing. Additionally, older enzymatic methods often discarded the less biologically active C-24R isomer as waste, leading to increased environmental burden and reduced atom economy, which is unacceptable in modern cost-sensitive supply chains.
The Novel Approach
In stark contrast, the methodology outlined in patent CN100404686C presents a transformative solution that overcomes these legacy inefficiencies through a clever combination of biocatalysis and chemical recycling. The core innovation lies in the selective enzymatic esterification of the C-24(R) hydroxyl group, leaving the desired C-24(S) hydroxyl group untouched, or vice versa via solvolysis, creating a significant chemical difference that facilitates easy separation. 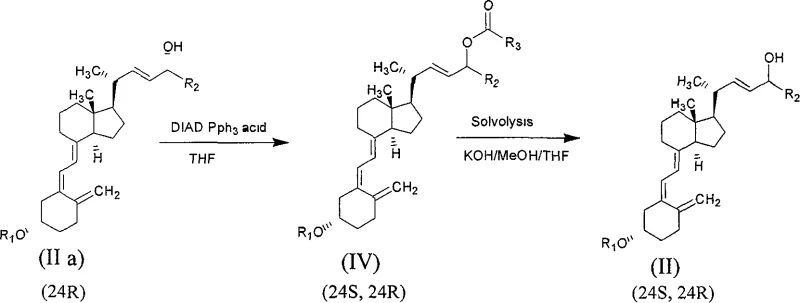 As illustrated in the reaction pathways, the process allows for the recovery and reuse of the C-24(R) isomer through a Mitsunobu inversion reaction, effectively converting the 'waste' stream back into a usable mixture of isomers. This closed-loop system drastically improves the total yield of the valuable C-24(S) form. By employing mild reaction temperatures ranging from 10°C to 60°C and common organic solvents like hexane or ethyl acetate, the process ensures operational safety and scalability, providing a clear pathway for cost reduction in API manufacturing while maintaining high stereochemical integrity.
As illustrated in the reaction pathways, the process allows for the recovery and reuse of the C-24(R) isomer through a Mitsunobu inversion reaction, effectively converting the 'waste' stream back into a usable mixture of isomers. This closed-loop system drastically improves the total yield of the valuable C-24(S) form. By employing mild reaction temperatures ranging from 10°C to 60°C and common organic solvents like hexane or ethyl acetate, the process ensures operational safety and scalability, providing a clear pathway for cost reduction in API manufacturing while maintaining high stereochemical integrity.
Mechanistic Insights into Lipase-Catalyzed Stereoselective Esterification
The success of this separation strategy hinges on the exquisite stereoselectivity of lipase enzymes, which function as highly specific biocatalysts capable of distinguishing between the C-24(R) and C-24(S) configurations. When a mixture of C-24 hydroxy vitamin D derivatives is exposed to a lipase such as Alcaligenes sp. or Pseudomonas sp. in the presence of an acylating agent like vinyl acetate, the enzyme preferentially binds to and esterifies the C-24(R) hydroxyl group. This selectivity is driven by the spatial arrangement of the enzyme's active site, which accommodates the R-isomer's side chain orientation more favorably than the S-isomer, or vice versa depending on the specific enzyme variant used. The reaction proceeds under mild conditions, typically between 20°C and 40°C, preventing thermal degradation of the sensitive vitamin D triene system. The result is a reaction mixture containing C-24(R)-acetate esters and unreacted C-24(S)-alcohols, which possess sufficiently different polarities to be easily separated by standard silica gel chromatography. 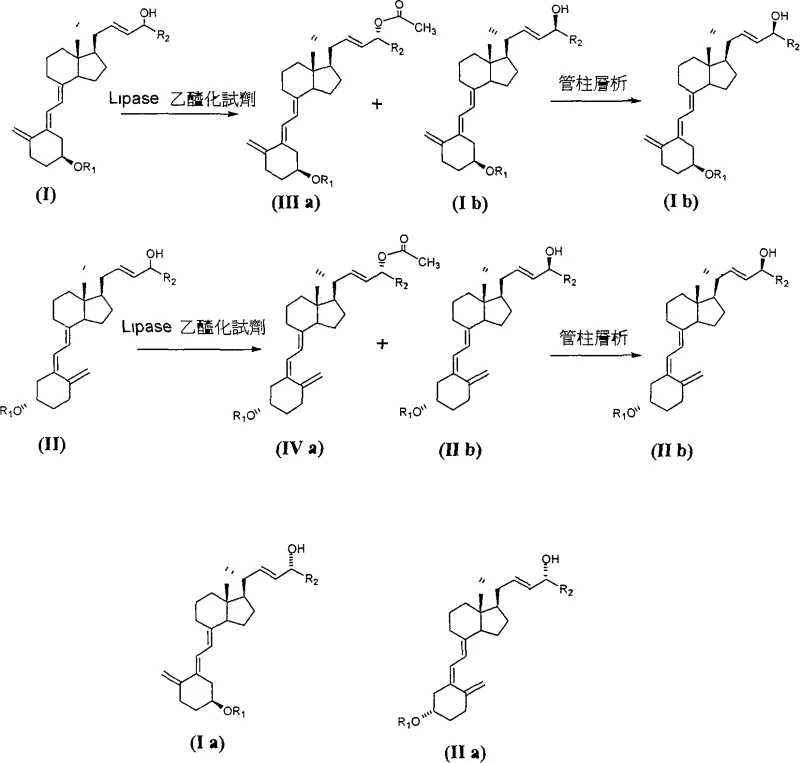 This mechanistic pathway ensures that the diastereomeric excess (d.e.) value can exceed 90%, and in optimized embodiments, reach greater than 95%, guaranteeing the high purity required for downstream pharmaceutical applications.
This mechanistic pathway ensures that the diastereomeric excess (d.e.) value can exceed 90%, and in optimized embodiments, reach greater than 95%, guaranteeing the high purity required for downstream pharmaceutical applications.
Impurity control is further enhanced by the specificity of the enzymatic reaction, which ignores other hydroxyl groups present on the vitamin D skeleton, such as those on the A-ring, provided they are protected or sterically hindered. The patent explicitly notes that even if the A-ring contains a free hydroxyl group, the lipase selectively targets the C-24 position, eliminating the need for complex orthogonal protection strategies that add steps and cost. Furthermore, the subsequent recycling of the C-24(R) isomer via Mitsunobu chemistry involves the use of reagents like triphenylphosphine and diisopropyl azodicarboxylate (DIAD) in aprotic solvents such as THF. This chemical inversion step converts the separated R-isomer back into a mixture of R and S esters, which upon hydrolysis regenerates the starting material mixture. This capability to chemically manipulate the stereochemistry post-separation provides a robust safety net against yield loss, ensuring that the impurity profile remains manageable and that the overall process mass intensity is optimized for industrial viability.
How to Synthesize C-24 Hydroxy Vitamin D Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize the diastereomeric ratio and overall throughput. The process begins with the preparation of a reaction mixture containing the C-24(R,S) hydroxy vitamin D derivative substrate, an organic solvent such as n-hexane or diisopropyl ether, and a vinyl ester acylating agent. The choice of solvent is critical, as it influences enzyme activity and substrate solubility; the patent highlights that solvents like tert-butyl methyl ether and ethyl acetate are also highly effective. Once the mixture is prepared, a specific lipase, either in free or immobilized form, is introduced to initiate the selective esterification. The reaction is monitored via HPLC to track the conversion of the C-24(R) isomer into its acetate form, with the endpoint determined when the diastereomeric excess reaches the desired threshold, typically above 80% and ideally exceeding 95%.
- Dissolve the mixture of C-24(R,S) hydroxy vitamin D derivative isomers in an organic solvent such as hexane or ethyl acetate, adding a vinyl ester acylating agent.
- Introduce a selective lipase enzyme, such as Alcaligenes sp. or Pseudomonas sp., and maintain the reaction temperature between 20°C to 40°C to achieve stereoselective esterification of the R-isomer.
- Separate the resulting C-24(R)-ester and unreacted C-24(S)-alcohol via column chromatography, then optionally recycle the R-isomer through Mitsunobu inversion to maximize overall yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic resolution technology translates into tangible strategic benefits that extend beyond simple technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow, which directly correlates to substantial cost savings in manufacturing operations. By replacing inefficient chromatographic separations with a highly selective enzymatic step, companies can reduce the consumption of expensive silica gel and large volumes of elution solvents, thereby lowering both material costs and waste disposal fees. Furthermore, the ability to recycle the C-24(R) isomer means that the effective yield of the valuable C-24(S) product is significantly increased without requiring additional raw material inputs. This improvement in atom economy reduces the dependency on costly starting materials and mitigates the risk of supply shortages, ensuring a more stable and predictable production schedule for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of harsh chemical reagents and the reduction in solvent usage inherent to this enzymatic process lead to a leaner production model. Traditional methods often require cryogenic temperatures or expensive chiral auxiliaries, whereas this method operates at near-ambient temperatures using commercially available lipases. The recycling loop for the R-isomer effectively turns a waste stream into a value-generating asset, minimizing the cost per kilogram of the final active isomer. This efficiency allows for more competitive pricing structures and higher margins in the supply of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process contributes to greater supply chain resilience. Lipases are stable, readily available biocatalysts that do not suffer from the supply volatility often associated with precious metal catalysts or specialized chiral ligands. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment corrosion, ensuring consistent quality and on-time delivery. Additionally, the flexibility to use various solvents and acylating agents provides procurement teams with the agility to adapt to market fluctuations in raw material availability without compromising the integrity of the synthesis.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard reactor equipment and the absence of hazardous by-products. The enzymatic nature of the reaction aligns with increasingly strict environmental regulations regarding volatile organic compounds (VOCs) and heavy metal residues. By minimizing waste generation and avoiding toxic reagents, manufacturers can streamline their environmental compliance protocols and reduce the carbon footprint of their operations. This sustainability profile is increasingly becoming a key differentiator for suppliers seeking partnerships with major multinational pharmaceutical corporations committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented separation technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. The answers are derived directly from the experimental data and claims presented in patent CN100404686C, providing a factual basis for decision-making.
Q: Why is enzymatic resolution preferred over traditional chromatography for Vitamin D isomers?
A: Traditional column chromatography struggles to separate C-24R and C-24S isomers due to their nearly identical polarity and structural similarity. Enzymatic resolution leverages the steric specificity of lipases to chemically differentiate the isomers, converting one into an ester while leaving the other as an alcohol, thereby drastically increasing the separation factor and purity.
Q: How does the patent address the waste of the less active C-24(R) isomer?
A: The process includes a crucial recycling step where the separated C-24(R) hydroxy derivative, which has lower biological activity, is subjected to a Mitsunobu reaction. This inverts the stereochemistry or creates a mixture that can be re-subjected to the enzymatic resolution, effectively converting waste material back into valuable starting material and improving atom economy.
Q: What specific enzymes and conditions are optimal for this separation?
A: The patent identifies Alcaligenes sp. Lipase and Pseudomonas sp. Lipase as highly effective biocatalysts. Optimal conditions involve reaction temperatures ranging from 20°C to 40°C in solvents like hexane, diisopropyl ether, or ethyl acetate, achieving diastereomeric excess (d.e.) values greater than 90% within 42 to 72 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin D Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the development of next-generation vitamin D therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated enzymatic resolution techniques described in patent CN100404686C can be seamlessly transferred to an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and chiral analysis capabilities to guarantee that every batch of C-24 hydroxy vitamin D derivatives meets the highest international standards. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate use in downstream API synthesis, minimizing validation time and accelerating time-to-market for new drug candidates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative separation technology can be tailored to your specific project requirements. By leveraging our expertise in biocatalysis and process optimization, we can provide a Customized Cost-Saving Analysis that demonstrates the economic viability of switching to this enzymatic route. We encourage you to request specific COA data and route feasibility assessments to verify the superior diastereomeric excess and yield improvements achievable through our manufacturing processes. Together, we can drive efficiency and innovation in the production of high-value pharmaceutical intermediates, securing a competitive edge in the global marketplace.
