Scalable Palladium-Catalyzed Synthesis of Vitamin D Derivatives for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex active ingredients, particularly for dermatological treatments where vitamin D analogs play a pivotal role. Patent CN101316813B discloses a groundbreaking conversion method for preparing specific vitamin D derivatives, notably compounds exhibiting potent human keratinocyte proliferation inhibitory activity with reduced hypercalcemic effects. This technology represents a significant departure from legacy synthesis routes, offering a chemically elegant solution to long-standing production bottlenecks. The core innovation lies in a convergent strategy that couples two distinct molecular fragments under palladium catalysis, bypassing the need for cumbersome photochemical rearrangements.  By leveraging this advanced organic synthesis technique, manufacturers can achieve higher throughput and consistency, addressing the critical demand for reliable vitamin D intermediate suppliers in the global market. The following analysis details the technical merits and commercial implications of this patented process.
By leveraging this advanced organic synthesis technique, manufacturers can achieve higher throughput and consistency, addressing the critical demand for reliable vitamin D intermediate suppliers in the global market. The following analysis details the technical merits and commercial implications of this patented process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of vitamin D derivatives has been plagued by significant technical hurdles that constrain supply and inflate costs. Traditional routes often depend on starting materials like 1α,3β-dihydroxy-5-androsten-17-one, which typically require specialized fermentation processes to produce in sufficient quantities, creating a fragile supply chain dependent on biological variability. Furthermore, the critical step of converting the steroid skeleton to the vitamin D secosteroid structure traditionally necessitates photochemical reactions. These photo-reactions require specialized equipment that is difficult to operate on a large scale due to light penetration limits and safety concerns associated with high-energy UV radiation. Consequently, the overall yield of these multi-step processes is often low, and the batch-to-batch reproducibility suffers, making it challenging to meet the stringent quality standards required for pharmaceutical intermediates. These inherent limitations have created a pressing need for cost reduction in pharmaceutical manufacturing through more robust chemical methodologies.
The Novel Approach
The methodology described in CN101316813B introduces a transformative chemical synthesis route that circumvents the drawbacks of fermentation and photochemistry. Instead of relying on light-induced bond cleavage, this novel approach utilizes a palladium-catalyzed cross-coupling reaction to join a bromo-substituted CD-ring fragment (Formula 2) with a protected A-ring alkyne fragment (Formula 3). 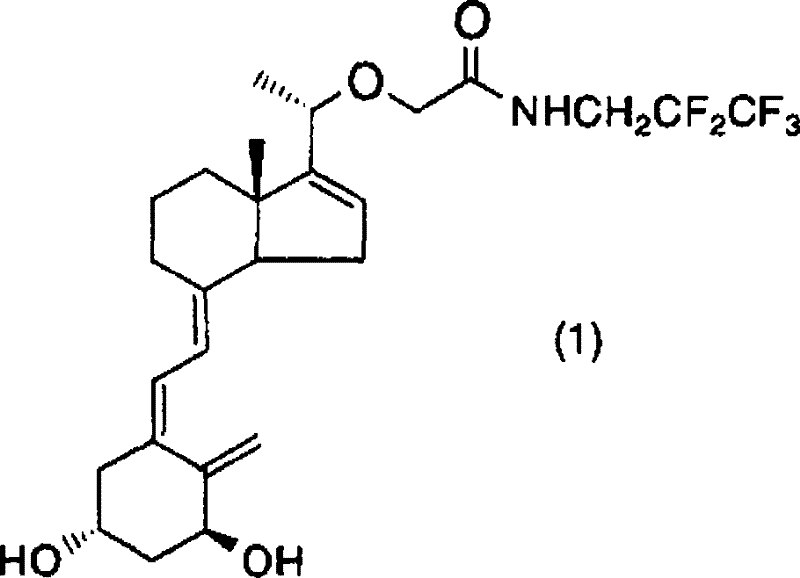 This convergent strategy allows for the independent optimization of both molecular halves before the final assembly, significantly enhancing process control. The reaction proceeds under thermal conditions in standard organic solvents like toluene, utilizing common bases and ligands, which makes the process inherently safer and easier to scale. By eliminating the dependency on specialized photoreactors and fermentation-derived precursors, this method offers a streamlined pathway that drastically simplifies the production workflow, thereby enhancing supply chain reliability for high-purity vitamin D derivatives.
This convergent strategy allows for the independent optimization of both molecular halves before the final assembly, significantly enhancing process control. The reaction proceeds under thermal conditions in standard organic solvents like toluene, utilizing common bases and ligands, which makes the process inherently safer and easier to scale. By eliminating the dependency on specialized photoreactors and fermentation-derived precursors, this method offers a streamlined pathway that drastically simplifies the production workflow, thereby enhancing supply chain reliability for high-purity vitamin D derivatives.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
The heart of this synthetic innovation is the palladium-catalyzed coupling reaction, which forms the crucial carbon-carbon bond linking the A-ring and CD-ring systems. The mechanism involves the oxidative addition of the palladium catalyst, such as palladium acetate or tetrakis(triphenylphosphine)palladium, into the carbon-halogen bond of the bromo-intermediate (Formula 2). This is followed by transmetallation with the alkyne species derived from Formula 3, facilitated by the presence of a base like triethylamine or diisopropylethylamine. The choice of ligand is critical; triphenylphosphine is particularly effective in stabilizing the palladium center and promoting the reductive elimination step that releases the coupled product (Formula 4).  The reaction is typically conducted at elevated temperatures, preferably between 80°C and 100°C, ensuring complete conversion of the starting materials within a reasonable timeframe of 8 to 10 hours. This thermal stability is a key advantage, allowing the use of standard heating jackets on industrial reactors rather than complex cooling or irradiation systems.
The reaction is typically conducted at elevated temperatures, preferably between 80°C and 100°C, ensuring complete conversion of the starting materials within a reasonable timeframe of 8 to 10 hours. This thermal stability is a key advantage, allowing the use of standard heating jackets on industrial reactors rather than complex cooling or irradiation systems.
Impurity control is another vital aspect addressed by the mechanistic design of this process. The A-ring fragment (Formula 3) incorporates protecting groups, such as tert-butyldimethylsilyl (TBS) ethers, at the 1α and 3β hydroxyl positions. These groups shield the reactive alcohol functionalities from participating in side reactions during the harsh coupling conditions, preventing the formation of ether byproducts or elimination impurities. After the coupling is complete, these protecting groups can be cleanly removed under mild acidic conditions, such as using hydrochloric acid in ethanol, or with fluoride sources like tetra-n-butylammonium fluoride. This strategic use of protection-deprotection chemistry ensures that the final active pharmaceutical ingredient meets rigorous purity specifications, minimizing the burden on downstream purification steps like silica gel chromatography and enhancing the overall economic viability of the process.
How to Synthesize Vitamin D Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities. The process begins with the preparation of the bromo-intermediate by alkylating the corresponding alcohol with 2-bromo-N-(2,2,3,3,3-pentafluoropropyl)acetamide in the presence of a strong base like sodium hexamethyldisilazide. Simultaneously, the A-ring fragment is synthesized with appropriate silyl protection. The critical coupling step involves mixing these two fragments with a palladium catalyst and ligand in an anhydrous solvent system under an inert atmosphere. Careful monitoring of the reaction temperature and time is essential to prevent catalyst degradation. Once the coupling is complete, the workup involves standard aqueous extraction and concentration, followed by the deprotection step to reveal the final diol functionality.
- Preparation of the bromo-substituted CD-ring intermediate (Formula 2) via alkylation of the corresponding alcohol with 2-bromo-N-(2,2,3,3,3-pentafluoropropyl)acetamide.
- Synthesis of the protected A-ring alkyne fragment (Formula 3) containing silyl protecting groups to ensure stability during coupling.
- Execution of the palladium-catalyzed cross-coupling reaction between Formula 2 and Formula 3 in toluene with triethylamine to form the protected vitamin D skeleton.
- Final deprotection of the silyl groups using acidic conditions or fluoride sources to yield the target active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this palladium-catalyzed synthesis offers profound strategic benefits that extend beyond simple chemistry. The primary advantage lies in the decoupling of production from biological constraints. By replacing fermentation-dependent starting materials with fully synthetic precursors, manufacturers gain complete control over raw material sourcing, mitigating the risks of crop failures or biological contamination that often plague semi-synthetic routes. This shift ensures a more predictable and stable supply of intermediates, which is crucial for maintaining continuous production schedules for downstream drug products. Furthermore, the elimination of photochemical steps removes the need for capital-intensive specialized equipment, allowing production to be housed in standard multipurpose chemical plants, thereby reducing fixed asset investment and operational overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of unit operations. Traditional photochemical steps often suffer from low throughput due to the physical limitations of light penetration in large vessels, requiring long processing times or multiple small batches. In contrast, the thermal coupling reaction described here can be run in large standard reactors with efficient heat transfer, significantly increasing batch size and throughput. Additionally, the use of commercially available palladium catalysts and ligands, which can often be recovered or used in low loadings, avoids the high costs associated with exotic reagents. The streamlined workflow reduces labor hours and utility consumption, leading to substantial cost savings in the overall cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the use of robust, non-perishable chemical starting materials. Unlike biological ferments which have short shelf-lives and strict storage requirements, the synthetic intermediates involved in this pathway are stable solids or liquids that can be stockpiled safely. This stability allows for better inventory management and buffer stock creation, protecting against market fluctuations or logistics disruptions. Moreover, the reaction conditions are tolerant to minor variations in input quality, providing a wider operating window that reduces the rate of batch failures. This reliability translates directly into shorter lead times for high-purity pharmaceutical intermediates, enabling faster response to market demand spikes.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner profile. The avoidance of photochemical waste streams and the potential for solvent recycling in the thermal coupling step align with green chemistry principles. The reaction generates fewer hazardous byproducts compared to older methods, simplifying waste treatment and disposal compliance. Scalability is inherent to the design; the reaction kinetics are not limited by photon flux, meaning that scaling from pilot kilograms to commercial tons is a matter of geometric reactor scaling rather than re-engineering the light source. This ease of scale-up facilitates rapid commercialization and ensures that the supply can grow in tandem with the clinical success of the final drug product.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its adoption. Common inquiries often revolve around the specific catalyst systems, the handling of sensitive intermediates, and the purification strategies required to meet pharmacopeial standards. The following answers address these critical operational questions based on the detailed disclosures within the patent literature, providing clarity on the feasibility and robustness of the technology for industrial application.
Q: Why is this palladium-catalyzed method superior to traditional photochemical synthesis?
A: Traditional methods rely on difficult-to-scale photochemical reactions and fermentation-derived starting materials. This new process utilizes standard thermal conditions and readily available chemical precursors, significantly improving manufacturability and supply chain reliability.
Q: What are the key impurities controlled in this synthesis?
A: The use of specific silyl protecting groups on the A-ring fragment prevents unwanted side reactions at the hydroxyl positions during the high-temperature coupling step, ensuring a cleaner impurity profile and higher final purity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions (80-100°C, standard solvents like toluene) are fully compatible with existing industrial reactor infrastructure, eliminating the need for specialized photoreactors and facilitating easy scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin D Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route disclosed in CN101316813B for the production of high-value vitamin D analogs. As a dedicated CDMO partner, we possess the technical expertise to translate this patented laboratory methodology into a robust, GMP-compliant commercial process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We employ stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate or API meets the highest international standards, minimizing risk for your regulatory filings.
We invite you to collaborate with us to optimize your supply chain for these critical dermatological therapeutics. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, identifying further opportunities for efficiency within this synthetic framework. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective manufacturing partner in the industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
