Optimizing Procaterol Hydrochloride Production via Stable Intermediate Synthesis
Introduction to Advanced Intermediate Synthesis
The pharmaceutical landscape continuously demands more robust and scalable pathways for critical bronchodilators like Procaterol Hydrochloride. Patent CN114195712A introduces a transformative approach to synthesizing key intermediates, specifically targeting the structural backbone required for beta-2 receptor agonists. This innovation addresses long-standing inefficiencies in traditional manufacturing, offering a pathway that enhances both chemical stability and process controllability. By shifting away from volatile acylating agents and optimizing the sequence of functional group transformations, this methodology provides a reliable foundation for large-scale production. The strategic redesign of the synthetic route ensures that manufacturers can achieve consistent quality while mitigating the risks associated with hazardous reagents.
For R&D directors and procurement specialists, understanding the nuances of this new intermediate is vital for securing supply chains. The core innovation lies in the utilization of 8-butyryloxyquinolone as a stable starting material, which undergoes a carefully orchestrated series of reactions to yield 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone. This compound serves as a pivotal precursor, bridging the gap between simple quinolone derivatives and the complex stereochemistry required for the final active pharmaceutical ingredient. The implications for commercial manufacturing are profound, as the process eliminates several purification bottlenecks that have historically plagued this sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Procaterol has relied on routes that introduce the butyryl side chain using highly reactive and unstable halides such as 2-bromobutyryl chloride or 2-bromobutyryl bromide. These reagents are notoriously difficult to handle on an industrial scale due to their susceptibility to hydrolysis and uncontrolled side reactions. The harsh conditions required for their deployment often lead to poor regioselectivity and the formation of complex impurity profiles that are challenging to remove. Furthermore, the original research routes disclosed in earlier literature often struggle with reproducibility when scaled beyond the laboratory bench, creating significant supply chain vulnerabilities.
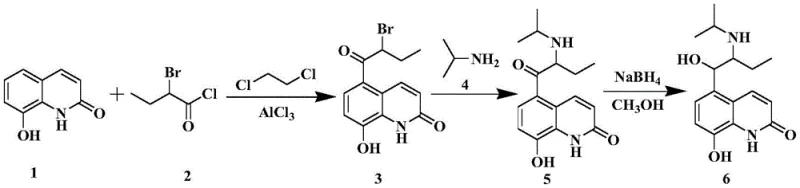
As illustrated in the conventional pathways, the direct acylation of 8-hydroxyquinolone derivatives frequently results in low yields and requires extensive downstream processing to meet pharmaceutical purity standards. The reliance on these unstable intermediates necessitates strict anhydrous conditions and specialized equipment, driving up capital expenditure and operational costs. Additionally, the environmental footprint of disposing of bromine-containing waste streams from these traditional methods adds another layer of complexity for compliance officers. Consequently, there is a pressing industry need for a synthesis strategy that circumvents these inherent chemical instabilities while maintaining high efficiency.
The Novel Approach
The patented methodology fundamentally reengineers the synthetic logic by employing 8-butyryloxyquinolone as the primary substrate, effectively decoupling the acylation step from the introduction of the bromine functionality. This approach utilizes a Fries rearrangement to migrate the acyl group to the desired position on the quinolone ring, followed by a protective benzylation step that stabilizes the phenolic hydroxyl group. This sequence ensures that the sensitive bromination occurs on a protected scaffold, drastically reducing side reactions and improving overall yield. The result is a streamlined process that is not only safer but also significantly more amenable to continuous manufacturing protocols.
By avoiding the direct use of 2-bromobutyryl halides in the initial stages, the new route achieves superior process stability and reproducibility. The intermediate 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone is obtained with high purity through straightforward crystallization techniques, eliminating the need for costly chromatographic separations. This shift represents a paradigm change in how bronchodilator intermediates are produced, offering a clear path toward cost reduction in pharmaceutical intermediate manufacturing. The robustness of this chemistry allows for wider operating windows, making it an ideal candidate for technology transfer to large-scale production facilities.
Mechanistic Insights into Fries Rearrangement and Regioselectivity
The success of this novel synthesis hinges on the precise execution of the Fries rearrangement, a Lewis acid-catalyzed migration of an acyl group from oxygen to the aromatic ring. In this specific application, the reaction of 8-butyryloxyquinolone with butyryl chloride in the presence of anhydrous aluminum trichloride generates a mixture of 5-butyryl and 7-butyryl isomers. The mechanistic preference for the 5-position is driven by the electronic activation of the quinolone ring and the steric environment surrounding the nitrogen atom. Careful control of reaction temperature, typically between 60°C and 120°C, is essential to maximize the ratio of the desired 5-isomer while minimizing thermal degradation of the sensitive heterocyclic core.
Crucially, the order of operations regarding the benzyl protection group is non-negotiable for successful synthesis. Comparative studies reveal that attempting to protect the hydroxyl group prior to the Fries rearrangement leads to complete process failure. When 8-benzyloxyquinolone is subjected to Friedel-Crafts acylation conditions, the reaction does not proceed to the desired intermediate; instead, it yields a complex mixture of unreacted starting material and debenzylated by-products. This failure underscores the electronic deactivation caused by the benzyloxy group, which hinders the electrophilic attack required for acylation at the 5-position.
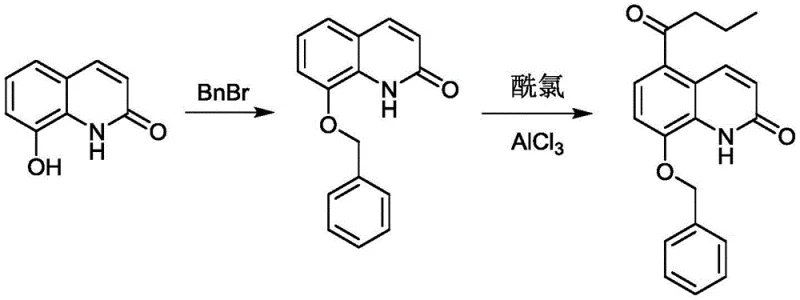
The mechanistic data confirms that the free phenolic hydroxyl group generated after the Fries rearrangement is essential for the subsequent benzylation step to proceed efficiently. Once the acyl group is securely positioned at C-5, the introduction of the benzyl protecting group using benzyl bromide and a base like potassium carbonate proceeds smoothly. This protection is vital for the subsequent bromination step, as it prevents unwanted side reactions at the oxygen atom and stabilizes the molecule during the radical or electrophilic bromination process. Understanding these electronic and steric subtleties is key for R&D teams aiming to replicate this high-yielding process in their own facilities.
How to Synthesize 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone Efficiently
The synthesis of this critical intermediate is achieved through a logical four-step sequence that prioritizes stability and ease of purification. The process begins with the Fries rearrangement to establish the carbon skeleton, followed by protection, functionalization, and finally amination. Each step has been optimized to utilize common industrial solvents and reagents, ensuring that the barrier to entry for adoption is low. The detailed standardized synthesis steps below outline the specific molar ratios, temperatures, and workup procedures required to achieve the reported high purity levels.
- Perform Fries rearrangement on 8-butyryloxyquinolone with butyryl chloride and Lewis acid catalyst to obtain 5-butyryl-8-hydroxyquinolone mixture.
- React the hydroxyquinolone mixture with a benzyl compound and base to form 5-butyryl-8-benzyloxyquinolone without isolation.
- Brominate the benzyloxy intermediate using pyridinium tribromide in tetrahydrofuran to yield 5-(2-bromobutyryl)-8-benzyloxyquinolone.
- Conduct aminolysis with isopropylamine to finalize the 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible benefits that extend beyond mere chemical elegance. The elimination of unstable and hazardous reagents like 2-bromobutyryl chloride significantly reduces the safety risks associated with raw material storage and handling. This inherently safer design translates to lower insurance premiums and reduced regulatory burdens, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the robustness of the process ensures a consistent supply of high-quality material, mitigating the risk of production delays caused by batch failures.
- Cost Reduction in Manufacturing: The new route eliminates the need for expensive and difficult-to-handle acylating agents, replacing them with stable and commercially available alternatives like butyryl chloride. By simplifying the purification process to basic crystallization and filtration, the method removes the dependency on resource-intensive chromatography. This streamlining of downstream processing drastically lowers the cost of goods sold (COGS) and improves overall margin potential for the final API.
- Enhanced Supply Chain Reliability: Utilizing stable starting materials such as 8-butyryloxyquinolone ensures that raw material sourcing is less susceptible to market volatility. The process tolerance for wider reaction parameters means that manufacturing can continue even with minor fluctuations in utility conditions, guaranteeing on-time delivery. This reliability is crucial for maintaining uninterrupted production schedules for life-saving bronchodilator medications.
- Scalability and Environmental Compliance: The synthesis is designed with industrial scale-up in mind, utilizing solvents and conditions that are easily managed in multi-ton reactors. The avoidance of excessive bromine waste and the reduction of complex by-product streams simplify wastewater treatment and environmental compliance. This green chemistry approach aligns with modern sustainability goals, making it an attractive option for companies focused on reducing their environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on why this route outperforms historical methods. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new standard.
Q: Why is 2-bromobutyryl chloride avoided in this new synthesis route?
A: Traditional routes utilizing 2-bromobutyryl chloride or bromide suffer from reagent instability and harsh reaction conditions, leading to significant side reactions and poor reproducibility. This novel method bypasses these unstable acylating agents entirely.
Q: What is the advantage of performing Fries rearrangement before benzyl protection?
A: Experimental data demonstrates that reversing the order (protecting before rearrangement) fails to yield the desired intermediate due to competitive side reactions and inability to isolate the target compound, making the specific sequence critical for success.
Q: What purity levels can be achieved with this manufacturing process?
A: The optimized process consistently delivers intermediates with purity exceeding 97% to 99% as verified by HPLC, significantly reducing the burden on downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate supply chains for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-(2-isopropylaminobutyryl)-8-benzyloxyquinolone meets the highest international standards. Our commitment to quality assurance means you can rely on us for consistent performance in your final drug formulation.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more stable manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your production volumes. Let us help you secure a sustainable and efficient source for your critical bronchodilator intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
