Revolutionizing Procaterol Synthesis: A Stable Intermediate Route for Industrial Scale-Up
Revolutionizing Procaterol Synthesis: A Stable Intermediate Route for Industrial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency with safety, particularly for critical bronchodilators like Procaterol Hydrochloride. A significant breakthrough in this domain is detailed in patent CN114105872A, which introduces a novel intermediate capable of streamlining the production of this essential medication. This patent addresses long-standing challenges associated with the instability of traditional acylating agents, proposing a method that utilizes 8-butyryloxyquinolone as a foundational raw material. By shifting away from volatile and reactive halides, this technology promises to enhance the reliability of the supply chain for respiratory therapeutics. The strategic implementation of this route allows manufacturers to bypass complex purification steps typically required for unstable intermediates, thereby optimizing the overall production workflow. For stakeholders in the fine chemical sector, this represents a pivotal shift towards more sustainable and controllable manufacturing processes for high-value API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
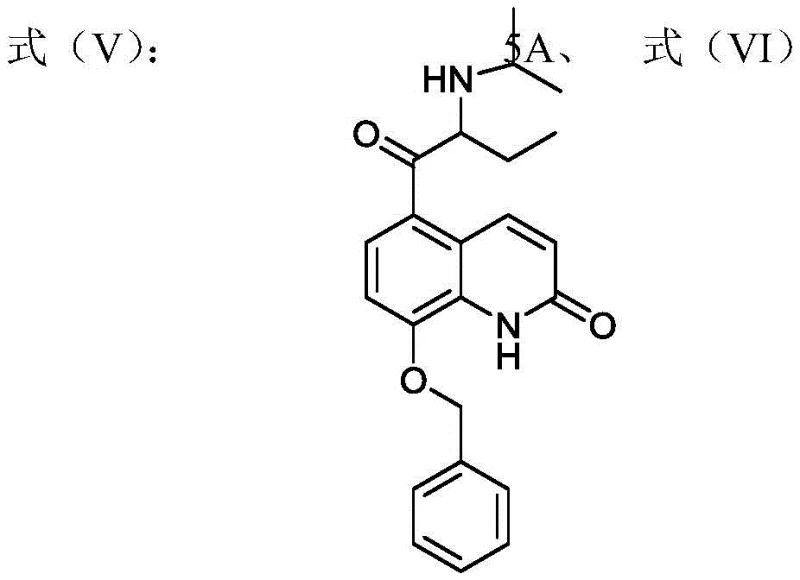
Historically, the synthesis of Procaterol has relied heavily on the use of 2-bromobutyryl chloride or 2-bromobutyryl bromide as key acylation reagents. As illustrated in the original research process, these reagents are inherently unstable and prone to generating significant side reactions during the acylation of 8-hydroxyquinolone. The harsh conditions required to manage these reactive species often lead to poor process stability and reproducibility, creating bottlenecks in large-scale production. Furthermore, the difficulty in controlling the bromination reaction precisely results in inconsistent yields and complicates the downstream purification efforts. These technical hurdles not only inflate production costs but also introduce variability that is unacceptable for GMP-compliant pharmaceutical manufacturing. The reliance on such sensitive reagents poses a continuous risk to supply continuity, making the conventional route less desirable for modern industrial applications seeking reliability.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114105872A employs 8-butyryloxyquinolone as a stable starting material, effectively circumventing the issues associated with unstable acid halides. This innovative approach utilizes a Fries rearrangement followed by a telescoped benzylation and bromination sequence, which drastically simplifies the operational complexity. By avoiding the direct use of 2-bromobutyryl reagents in the initial stages, the process achieves a higher degree of control over reaction parameters. The ability to carry forward crude mixtures without intermediate isolation further enhances the efficiency of the workflow, reducing solvent usage and processing time. This novel pathway not only resolves the reproducibility issues plaguing older methods but also ensures that the final intermediate meets stringent purity specifications required for API synthesis. It stands as a testament to how strategic reagent selection can transform a challenging synthesis into a robust industrial process.
Mechanistic Insights into Fries Rearrangement and Selective Bromination
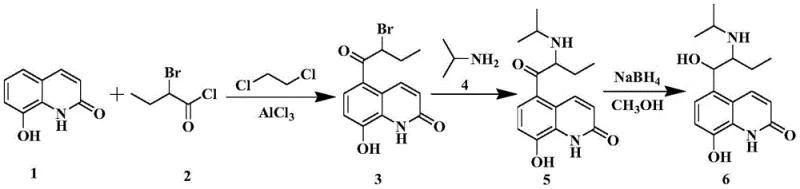
The core of this technological advancement lies in the precise execution of a Fries rearrangement followed by a highly selective bromination using pyridinium tribromide. The process begins with the rearrangement of 8-butyryloxyquinolone in the presence of a Lewis acid catalyst, such as anhydrous aluminum trichloride, to generate a mixture of 5-butyryl and 7-butyryl hydroxyquinolones. Crucially, this mixture is not purified but is directly subjected to benzylation, where the phenolic hydroxyl group is protected. The subsequent bromination step is the mechanistic highlight, utilizing pyridinium tribromide in tetrahydrofuran to selectively introduce the bromine atom at the alpha-position of the butyryl side chain. This reagent choice is superior to liquid bromine as it provides a controlled release of bromine, minimizing over-bromination and ring substitution side reactions. The mechanism ensures that even if the 7-isomer is present in the mixture, the reaction conditions favor the formation of the desired 5-substituted product or allow for its easy separation later.
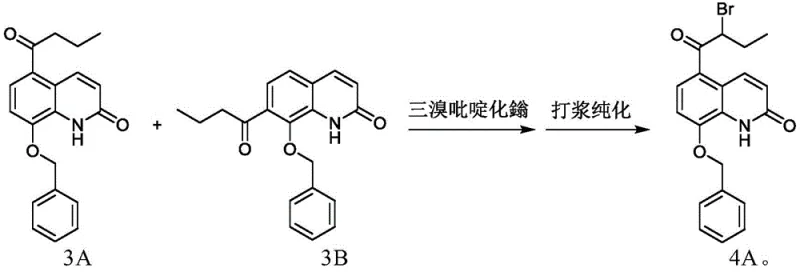
Impurity control is intrinsically built into this synthetic design through the physical properties of the intermediates and the specificity of the reagents. The use of pyridinium tribromide prevents the formation of complex poly-brominated by-products that are common when using elemental bromine. Additionally, the process leverages the solubility differences between the target intermediate and its isomers or by-products. For instance, the final product, 5-(2-bromobutyryl)-8-benzyloxyquinolone, can be effectively purified via slurry techniques in solvents like ethyl acetate, removing unreacted starting materials and minor impurities. The patent data indicates that even if the isomeric mixture from the Fries rearrangement is carried through, the final crystallization or slurry steps yield a product with purity exceeding 98%. This robustness against isomeric impurities reduces the need for expensive chromatographic separations, making the process economically viable and chemically elegant for large-scale operations.
How to Synthesize 5-(2-bromobutyryl)-8-benzyloxyquinolone Efficiently
The synthesis of this critical intermediate involves a streamlined three-step sequence that maximizes yield while minimizing waste. The process initiates with a Fries rearrangement under controlled thermal conditions, followed immediately by protection of the hydroxyl group to prevent unwanted side reactions. The final bromination step requires careful temperature management to ensure selectivity. Detailed operational parameters, including specific molar ratios and solvent volumes, are critical for achieving the high purity levels reported in the patent literature. Manufacturers looking to implement this route should pay close attention to the choice of solvent in the bromination step, as tetrahydrofuran has been identified as the optimal medium for this transformation.
- Perform Fries rearrangement on 8-butyryloxyquinolone with butyryl chloride and Lewis acid catalyst to obtain a mixture of butyryl-hydroxyquinolones.
- React the crude mixture directly with a benzyl compound and base to form 5-butyryl-8-benzyloxyquinolone without intermediate isolation.
- Treat the benzyloxy derivative with pyridinium tribromide in tetrahydrofuran to achieve selective alpha-bromination, yielding the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the stabilization of the raw material supply chain; by replacing unstable and hazardous acid halides with stable ester precursors, the risk of raw material degradation during storage and transport is virtually eliminated. This stability translates directly into reduced inventory losses and more predictable procurement cycles. Furthermore, the telescoping of reaction steps—specifically carrying the crude Fries product directly into benzylation—significantly reduces the consumption of solvents and the energy required for intermediate isolation. This reduction in unit operations leads to a leaner manufacturing footprint and lower overall production costs. The process is designed to be scalable, ensuring that supply can be ramped up to meet market demand without the technical bottlenecks associated with purifying unstable intermediates.
- Cost Reduction in Manufacturing: The elimination of unstable reagents like 2-bromobutyryl chloride removes the need for specialized handling equipment and rigorous safety protocols associated with corrosive and volatile chemicals. By utilizing 8-butyryloxyquinolone, the process inherently lowers the cost of goods sold through improved yield consistency and reduced waste disposal costs. The ability to skip the isolation of intermediate mixtures further drives down operational expenses by saving on solvent recovery and drying times. Consequently, the overall manufacturing economics are optimized, allowing for more competitive pricing in the global API market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stable raw materials is a cornerstone of a resilient supply chain. The new route relies on commercially available and stable precursors, mitigating the risk of supply disruptions caused by the shelf-life limitations of reactive acid halides. This stability ensures that production schedules can be maintained with greater certainty, reducing the likelihood of delays due to raw material quality issues. Additionally, the robustness of the synthesis against isomeric impurities means that variations in upstream feedstock quality are less likely to halt production, providing a buffer against supply chain volatility and ensuring consistent delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently greener due to the reduction in solvent usage and the avoidance of hazardous liquid bromine in favor of the safer pyridinium tribromide complex. This shift aligns with increasingly stringent environmental regulations regarding volatile organic compounds and hazardous waste generation. The simplified workup procedures, such as slurry purification instead of complex chromatography, facilitate easier scale-up from pilot plant to commercial tonnage. This scalability ensures that the technology can support the growing global demand for respiratory medications while maintaining a compliant and sustainable manufacturing profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new methodology for their production lines.
Q: Why is the new synthesis route for procaterol intermediates considered superior to conventional methods?
A: The new route avoids unstable reagents like 2-bromobutyryl chloride, utilizing stable 8-butyryloxyquinolone instead, which significantly improves process stability and reproducibility.
Q: Does the presence of isomers affect the purity of the final procaterol intermediate?
A: No, the process is designed such that even if isomer 3B is present in the raw material mixture, it does not negatively impact the quality or purity of the final intermediate 4A after purification.
Q: What are the key advantages of using pyridinium tribromide over liquid bromine in this synthesis?
A: Pyridinium tribromide offers safer handling and more controlled reaction conditions compared to liquid bromine, reducing side reactions and simplifying the isolation of the target product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-bromobutyryl)-8-benzyloxyquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our expertise aligns perfectly with the advanced synthesis techniques described in patent CN114105872A, allowing us to deliver this complex quinolone derivative with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a reliable supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-(2-bromobutyryl)-8-benzyloxyquinolone adheres to the highest industry standards. We are committed to supporting your drug development and commercialization goals with materials that facilitate smooth downstream processing.
We invite you to collaborate with us to leverage this superior synthetic route for your Procaterol projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this stable intermediate can optimize your overall manufacturing economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, cost-effective, and high-quality supply chain for your respiratory therapeutic portfolios.
