Scalable Synthesis of High-Purity BTK Inhibitor Intermediates via Mild IBX Oxidation
Scalable Synthesis of High-Purity BTK Inhibitor Intermediates via Mild IBX Oxidation
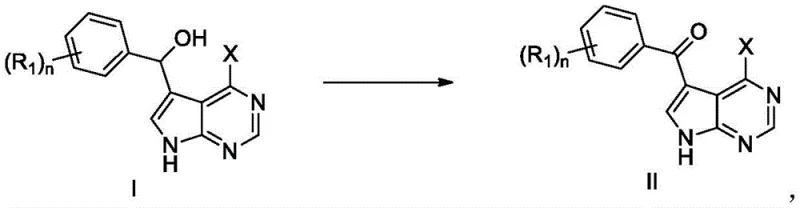
The development of Bruton's tyrosine kinase (BTK) inhibitors represents a cornerstone in modern oncology therapeutics, particularly for treating B-cell malignancies. However, the commercial viability of these life-saving drugs often hinges on the efficiency of synthesizing their critical intermediates. Recent intellectual property, specifically patent CN115353519A, introduces a transformative refining method for producing the key intermediate known as Formula II. This technical breakthrough shifts the paradigm from hazardous, cryogenic lithiation processes to a mild, high-yield oxidation strategy using 2-iodoxybenzoic acid (IBX). For global pharmaceutical manufacturers, this evolution signals a move towards more sustainable, cost-effective, and scalable production capabilities, directly addressing the chronic bottlenecks associated with complex heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrolopyrimidine-based BTK inhibitor intermediates has relied heavily on organolithium chemistry. Prior art, such as methods described in CN108699062A, necessitates the use of tert-butyllithium under extreme cryogenic conditions, typically around -78°C. This requirement imposes severe constraints on industrial infrastructure, demanding specialized low-temperature reactors and significant energy expenditure for cooling. Furthermore, these conventional routes often suffer from mediocre yields, frequently capping at approximately 41%, which drastically inflates the cost of goods sold (COGS) due to material loss. Perhaps most critically for supply chain continuity, these legacy methods depend on column chromatography for purification, a technique that is notoriously difficult to scale, solvent-intensive, and inefficient for multi-ton manufacturing campaigns.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353519A utilizes IBX as a selective oxidant to convert the alcohol precursor (Formula I) directly into the ketone target (Formula II). This reaction proceeds efficiently at ambient temperatures between 20°C and 30°C, eliminating the need for energy-intensive cryogenic systems. The process achieves exceptional conversion rates with yields reaching up to 93% and purity levels exceeding 99.5%. Crucially, the purification strategy abandons column chromatography in favor of a sophisticated multi-step slurry beating process. This shift not only simplifies the operational workflow but also drastically reduces solvent consumption and processing time, making the route inherently more robust for commercial scale-up.
Mechanistic Insights into IBX-Catalyzed Oxidation and Impurity Control
The core chemical transformation involves the oxidation of a secondary alcohol adjacent to the pyrrolopyrimidine ring system. IBX acts as a hypervalent iodine reagent that facilitates the removal of hydrogen atoms to form the carbonyl group without affecting other sensitive functionalities on the molecule. The reaction mechanism is clean and thorough, ensuring no starting material remains, which simplifies downstream processing. The choice of solvent, preferably dimethyl sulfoxide (DMSO), plays a vital role in solubilizing both the substrate and the oxidant, ensuring homogeneous reaction kinetics. This mechanistic elegance allows for a narrow operating window where side reactions are minimized, directly contributing to the high crude purity observed before any purification steps are even initiated.
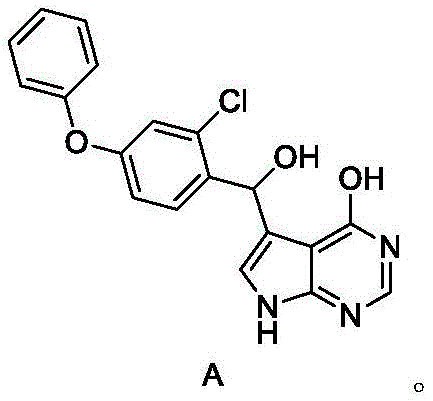
A critical aspect of this technology is the management of Impurity A, a hydrolysis byproduct that can compromise the quality of the final API. Structural analysis reveals that Impurity A arises from the hydrolysis of the chlorine atom on the pyrrolopyrimidine core under certain conditions. The patent data indicates that the choice of alkaline solution during the slurry purification step is paramount. By employing weak alkaline solutions such as potassium carbonate, sodium carbonate, or dipotassium hydrogen phosphate, the process effectively suppresses the formation of this hydrolysis impurity. Stronger bases or improper pH control can accelerate hydrolysis, but the optimized weak base protocol maintains the integrity of the chloro-substituent while effectively washing away organic byproducts.
How to Synthesize BTK Inhibitor Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the dissolution of the Formula I substrate in DMSO, followed by the controlled addition of IBX. The reaction mixture is maintained at mild temperatures to ensure steady oxidation. Upon completion, the workup involves a quenching step with sodium thiosulfate to reduce excess oxidant, followed by a series of filtration and slurry washing steps designed to crystallize the product while leaving impurities in the mother liquor. For a detailed breakdown of the standardized operating procedures and specific stoichiometric ratios required for GMP manufacturing, please refer to the technical guide below.
- Oxidize Formula I compound with IBX in DMSO at 20-30°C for 18-24 hours.
- Quench reaction with sodium thiosulfate solution and perform initial slurry beating.
- Purify via sequential slurry treatments using acetonitrile/weak base and DMSO/water to remove Impurity A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this IBX-based synthesis route offers tangible strategic advantages beyond mere chemical elegance. The elimination of cryogenic requirements translates to a significant reduction in utility costs and capital expenditure on specialized equipment. Furthermore, the removal of column chromatography addresses one of the most persistent bottlenecks in fine chemical manufacturing, allowing for faster batch turnover and reduced solvent disposal costs. The high yield and purity profile also mean less raw material is needed per kilogram of output, optimizing inventory management and reducing the environmental footprint of the production campaign.
- Cost Reduction in Manufacturing: The transition from cryogenic lithiation to ambient temperature oxidation fundamentally alters the cost structure of the intermediate. By removing the need for -78°C cooling and expensive organolithium reagents, the direct material and utility costs are substantially lowered. Additionally, replacing column chromatography with slurry purification eliminates the massive solvent volumes associated with chromatographic columns, leading to drastic savings in solvent procurement and waste treatment expenses. The high yield of over 90% ensures that raw material utilization is maximized, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Relying on stable, shelf-stable reagents like IBX rather than pyrophoric tert-butyllithium enhances operational safety and supply security. The simplified process flow reduces the risk of batch failures associated with complex low-temperature operations. Moreover, the robustness of the slurry purification method ensures consistent product quality across different batches, minimizing the risk of out-of-specification results that could delay shipments. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial expansion, avoiding techniques that are difficult to translate from lab to plant. The reduction in solvent usage and the avoidance of hazardous cryogenic fluids align with increasingly stringent environmental regulations and green chemistry principles. This makes the technology future-proof against tightening emissions standards and waste disposal restrictions. The ability to scale from kilograms to metric tons without changing the fundamental purification logic ensures that supply can grow in lockstep with market demand for BTK inhibitors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and quality control measures.
Q: How does the IBX oxidation method improve upon traditional lithiation routes?
A: Unlike traditional methods requiring tert-butyllithium at -78°C, the IBX method operates at mild temperatures (20-30°C), significantly reducing energy costs and safety risks while improving yield from ~41% to over 90%.
Q: What is the primary strategy for controlling Impurity A in this synthesis?
A: Impurity A is a hydrolysis byproduct. The process controls it by utilizing specific weak alkaline solutions (like potassium carbonate) during the slurry purification step, which prevents hydrolysis of the sensitive chloro-pyrrolopyrimidine core.
Q: Is column chromatography required for purification in this new method?
A: No. A key advantage of this patent is the replacement of column chromatography with a multi-step slurry purification process, making it highly suitable for industrial scale-up and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BTK Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs depends on the reliability of the supply chain for critical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of the IBX oxidation process, including rigorous QC labs capable of detecting trace impurities like Impurity A at ppm levels. We adhere to stringent purity specifications to ensure that every batch of BTK inhibitor intermediate meets the exacting standards required for subsequent coupling reactions in API synthesis.
We invite potential partners to engage with our technical team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this refined method. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your project timelines and volume requirements.
