Scalable Manufacturing of Heteroaromatic BTK Inhibitor Intermediates via Novel Copper Catalysis
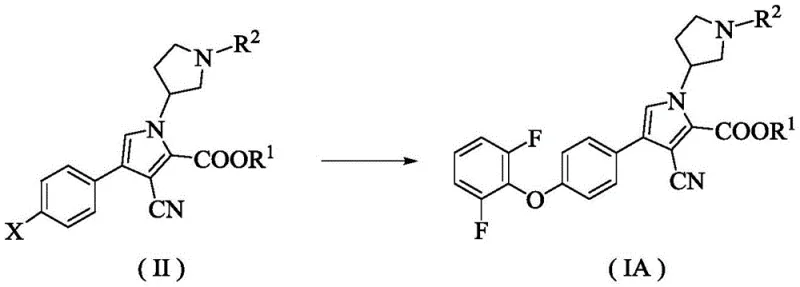
The pharmaceutical landscape for treating B-cell malignancies and autoimmune disorders has been revolutionized by Bruton's tyrosine kinase (BTK) inhibitors, creating an urgent demand for reliable supply chains of their complex heteroaromatic intermediates. Patent CN116425664A discloses a groundbreaking preparation method for a specific heteroaromatic BTK inhibitor intermediate, designated as Formula (IA), which serves as a critical precursor in the synthesis of next-generation therapeutics. This technical disclosure addresses the longstanding challenges in constructing the sterically hindered ether linkage between the pyrrole core and the 2,6-difluorophenyl moiety, a structural motif essential for kinase selectivity and clinical efficacy. By leveraging a novel copper-catalyzed coupling strategy, this methodology offers a robust alternative to traditional palladium-mediated processes, thereby enhancing the feasibility of large-scale manufacturing. For global pharmaceutical developers, securing a supply of this intermediate through such an optimized route ensures not only chemical integrity but also strategic independence in the production of life-saving medications targeting chronic lymphocytic leukemia and rheumatoid arthritis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of diaryl ether bonds within complex heteroaromatic systems has relied heavily on transition metal-catalyzed cross-coupling reactions that often necessitate the use of precious metals like palladium. These conventional methodologies frequently suffer from significant drawbacks, including the requirement for elevated temperatures that can compromise the stability of sensitive functional groups such as esters and nitriles present in the BTK inhibitor scaffold. Furthermore, the removal of residual heavy metal catalysts to meet stringent pharmaceutical purity specifications adds costly and time-consuming purification steps to the manufacturing workflow. Traditional routes may also exhibit poor atom economy and generate substantial amounts of hazardous waste, conflicting with modern green chemistry principles and increasing the environmental compliance burden for manufacturers. The reliance on expensive ligands and inert atmosphere conditions in older protocols further exacerbates the operational expenditure, making the cost of goods sold (COGS) prohibitively high for widespread commercial adoption.
The Novel Approach
The innovative process detailed in the patent data introduces a highly efficient copper-catalyzed system that operates under remarkably mild reaction conditions, effectively overcoming the thermal sensitivity issues associated with previous methods. By utilizing readily available copper salts such as cuprous bromide or cuprous iodide in conjunction with a specialized diketopromide ligand, the reaction achieves high conversion rates without the need for extreme thermal energy input. This approach significantly simplifies the reaction setup and reduces the dependency on scarce precious metal resources, thereby stabilizing the supply chain against market volatility in metal prices. The compatibility of this catalytic system with diverse protecting groups allows for greater flexibility in synthetic design, enabling chemists to optimize the route for maximum yield and minimal impurity generation. Ultimately, this novel methodology represents a paradigm shift towards more sustainable and economically viable manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Etherification
The core of this technological advancement lies in the precise orchestration of the copper catalytic cycle, which facilitates the nucleophilic substitution of the leaving group on the heteroaromatic ring by the 2,6-difluorophenol nucleophile. The presence of the specific diketopromide ligand is crucial as it stabilizes the copper center, preventing aggregation and maintaining the active catalytic species throughout the reaction duration. This ligand-metal interaction enhances the electron density at the copper atom, promoting the oxidative addition step which is often the rate-determining factor in such cross-coupling transformations. The steric environment created by the ligand also plays a pivotal role in discriminating against potential side reactions, ensuring that the coupling occurs selectively at the desired position on the pyrrole ring. Such mechanistic control is essential for maintaining the structural integrity of the chiral centers and other sensitive moieties within the molecule, which are critical for the biological activity of the final BTK inhibitor drug substance.
Impurity control is inherently built into this mechanistic design through the suppression of homocoupling and dehalogenation pathways that typically plague copper-catalyzed reactions. The optimized molar ratios of the metal catalyst to the ligand, ranging from 1:1 to 1:10 as disclosed, ensure that the catalytic turnover is maximized while minimizing the formation of metal-associated impurities that are difficult to remove downstream. The mild basic conditions employed, utilizing reagents like potassium phosphate, further contribute to a clean reaction profile by avoiding the harsh alkaline environments that could lead to ester hydrolysis or nitrile degradation. This high level of chemoselectivity translates directly into a simplified workup procedure, where the crude product often requires minimal purification to achieve the necessary purity standards for subsequent synthetic steps. Consequently, the overall process mass intensity is reduced, aligning with the industry's push towards more efficient and environmentally responsible chemical manufacturing practices.
How to Synthesize Heteroaromatic BTK Inhibitor Intermediate Efficiently
The synthesis of the target compound involves a meticulously designed sequence of transformations that prioritize yield and operational simplicity at every stage. Starting from readily available pyrrole derivatives, the route incorporates strategic protection and deprotection steps to manage the reactivity of the nitrogen atoms, ensuring that the coupling reactions proceed with high regioselectivity. The key etherification step, which forms the bond between the heteroaromatic core and the difluorophenyl ring, is executed under nitrogen protection to maintain catalyst activity and prevent oxidation of sensitive intermediates. Detailed standardized synthetic steps see the guide below.
- Protect the pyrrole nitrogen of the starting material using a sulfonyl group to prevent side reactions during subsequent functionalization.
- Perform lithiation followed by esterification to introduce the carboxyl group at the desired position on the heteroaromatic ring.
- Execute a copper-catalyzed coupling reaction with 2,6-difluorophenol using a specific diketopromide ligand to form the key ether linkage.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility into the realm of cost optimization and risk mitigation. The shift from precious metal catalysts to abundant copper-based systems fundamentally alters the cost structure of the intermediate, removing exposure to the volatile pricing of palladium and rhodium markets. This transition not only lowers the direct material costs but also simplifies the regulatory documentation required for metal residue clearance, accelerating the release of batches for further processing. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in operating parameters, thereby enhancing the reliability of supply deliveries to downstream API manufacturers. Furthermore, the high yields reported in the experimental examples suggest a more efficient utilization of starting materials, reducing the overall volume of waste that requires treatment and disposal.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the use of cost-effective copper salts result in a substantial decrease in raw material expenditures per kilogram of produced intermediate. Additionally, the mild reaction temperatures reduce energy consumption requirements for heating and cooling, contributing to lower utility costs over the lifecycle of the product. The simplified purification process, driven by the high selectivity of the new method, reduces the consumption of chromatography media and solvents, which are often significant cost drivers in fine chemical manufacturing. These cumulative savings allow for a more competitive pricing structure without compromising on the quality or purity of the final pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as cuprous bromide and potassium phosphate, the manufacturing process is insulated from the supply disruptions that frequently affect specialized organometallic reagents. The availability of these reagents from multiple global suppliers ensures that production schedules can be maintained even in the face of regional logistical challenges or geopolitical tensions. The stability of the intermediates under the described reaction conditions also allows for longer holding times if necessary, providing greater flexibility in production planning and inventory management. This resilience is critical for maintaining continuous supply to pharmaceutical clients who operate on tight just-in-time manufacturing schedules.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, with reaction parameters that translate seamlessly from laboratory scale to multi-ton commercial reactors without the need for complex engineering modifications. The reduction in hazardous waste generation and the use of less toxic metal catalysts align with increasingly stringent environmental regulations, minimizing the risk of compliance violations and associated fines. The ability to scale up efficiently ensures that the supply can grow in tandem with the clinical and commercial demand for the BTK inhibitor, preventing bottlenecks that could delay patient access to new therapies. This scalability makes the technology an attractive option for long-term partnerships focused on bringing new drugs to market rapidly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this BTK inhibitor intermediate, derived directly from the patent specifications and experimental data. These insights are intended to clarify the capabilities of the manufacturing process and its alignment with industry standards for quality and safety. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader drug development pipelines.
Q: What is the primary advantage of this BTK inhibitor intermediate synthesis method?
A: The primary advantage is the use of mild reaction conditions and a copper-based catalytic system, which avoids the need for expensive palladium catalysts and harsh temperatures, significantly lowering production costs and environmental impact while maintaining high purity.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes specific protecting group strategies and optimized ligand systems that minimize byproduct formation. The mild conditions prevent thermal degradation of sensitive heteroaromatic structures, ensuring the impurity profile meets stringent pharmaceutical standards.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production. The reagents are commercially available, the reaction steps are robust, and the avoidance of extreme conditions facilitates safe scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BTK Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of oncology and immunology therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of BTK inhibitor intermediate meets the exacting standards required by global regulatory agencies. We understand the complexities of heteroaromatic chemistry and are prepared to implement the advanced copper-catalyzed processes described in recent patents to deliver superior value to our clients.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this optimized synthetic route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and transparency. Let us collaborate to secure a stable and cost-effective supply of this vital pharmaceutical building block for your next breakthrough therapy.
