Advanced Synthesis of Atracurium Besilate Intermediates for Commercial Scale-Up
Advanced Synthesis of Atracurium Besilate Intermediates for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust and efficient pathways for producing critical active pharmaceutical ingredient (API) precursors, particularly for neuromuscular blocking agents used in anesthesia. Patent CN101845017A introduces a significant breakthrough in the preparation of atracurium besilate intermediates, specifically targeting the synthesis of (1R, 1'R), (1S, 1'S), and (1R, 1'S)-4,10-dioxy-3,11-dioxo tridecylene-1,13-bi-[1-(3,4-dimethoxy benzyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline] dioxalate. This innovation addresses long-standing challenges in the supply chain of cisatracurium besylate, a drug renowned for its organ-independent metabolism via Hofmann elimination. By re-engineering the synthetic route to eliminate the most problematic intermediate, this technology offers a pathway that is not only chemically superior but also commercially viable for high-volume manufacturing. For R&D directors and procurement specialists, understanding this shift from complex diene synthesis to a streamlined Michael addition and transesterification sequence is crucial for securing a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates for atracurium besilate has been plagued by significant technical hurdles that impact both cost and timeline. Traditional routes, as referenced in prior art such as U.S. Patents 4,179,507 and 5,454,510, rely heavily on the preparation of a specific diene intermediate known as 4,10-dioxy-3,11-dioxo-1,12-tridecylene. The synthesis of this particular compound is notoriously difficult, requiring multi-step sequences that often involve harsh reaction conditions and sensitive reagents. These legacy methods result in extended production cycles, increased labor intensity due to complex post-reaction treatments, and stringent equipment requirements to handle unstable intermediates. Furthermore, the low yields associated with the formation of the diene backbone lead to substantial raw material waste, driving up the overall cost of goods sold (COGS) and creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN101845017A revolutionizes the process by completely bypassing the synthesis of the difficult diene intermediate. Instead, the novel approach initiates with a direct Michael addition reaction between tetrahydropapaverine and methyl or ethyl acrylate in an alcoholic solvent. This is followed by a base-catalyzed transesterification with 1,5-pentanediol, utilizing potassium tert-butoxide in toluene. This strategic pivot simplifies the entire workflow, replacing harsh conditions with mild reflux processes that are far easier to control and monitor. The result is a dramatic reduction in operational complexity, allowing for higher product purity and yield without the need for exotic reagents. For procurement managers, this translates to cost reduction in pharmaceutical intermediates manufacturing through simplified logistics and reduced waste disposal costs.
Mechanistic Insights into Michael Addition and Transesterification
The core of this technological advancement lies in the precise execution of two fundamental organic transformations: Michael addition and transesterification. The process begins with the nucleophilic attack of the secondary amine in tetrahydropapaverine on the beta-carbon of the acrylate ester. This Michael addition is conducted under reflux in methanol or ethanol, ensuring complete conversion to the mono-acrylate ester intermediate. The choice of solvent and temperature is critical here, as it prevents over-alkylation while maintaining the stereochemical integrity required for the final chiral drug substance. Following isolation, the intermediate undergoes transesterification where the alkoxide generated from 1,5-pentanediol attacks the carbonyl of the acrylate ester. The use of potassium tert-butoxide as a catalyst ensures rapid kinetics, while the simultaneous removal of the generated alcohol (methanol or ethanol) via azeotropic distillation with toluene drives the equilibrium towards the desired diester product.
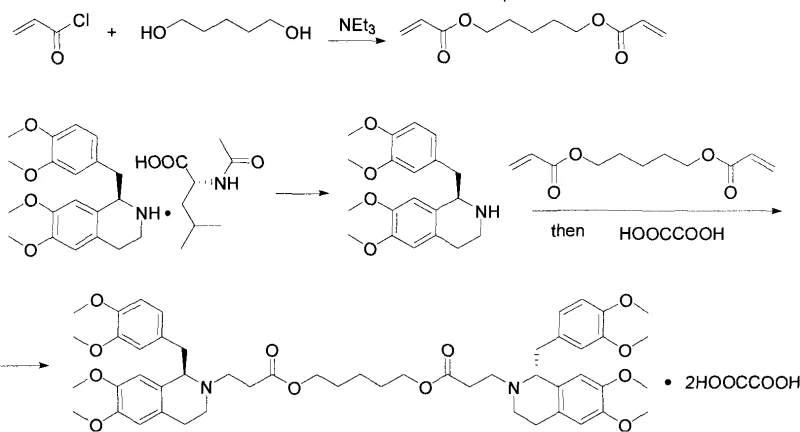
Controlling impurities in this sequence is paramount for meeting the rigorous standards of a reliable agrochemical intermediate supplier or pharma partner. The avoidance of the diene intermediate inherently eliminates a major source of polymeric byproducts and degradation compounds that typically arise from the instability of conjugated systems in the old route. Furthermore, the final salt formation step with oxalic acid in acetone serves as a powerful purification mechanism. By carefully controlling the addition of saturated oxalic acid solution, the process ensures the precipitation of the specific stereoisomers ((1R, 1'R), (1S, 1'S), and (1R, 1'S)) while leaving soluble impurities in the mother liquor. This crystallization-induced purity enhancement is a key factor in achieving the high-quality specifications demanded for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Atracurium Besilate Intermediates Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and process parameters to maximize efficiency. The patent outlines a clear protocol where the molar ratio of tetrahydropapaverine to acrylate is maintained between 1:2.0 and 1:4.0 to ensure full conversion. Subsequently, the transesterification step utilizes a molar ratio of ester intermediate to pentanediol of approximately 1.8-2.2:1, with a catalytic loading of potassium tert-butoxide at 0.08-0.12 equivalents relative to the diol. These optimized ratios are essential for minimizing side reactions and ensuring high throughput. For detailed standard operating procedures and safety guidelines regarding the handling of potassium tert-butoxide and solvent recovery, please refer to the specific technical documentation provided below.
- Perform a Michael addition reaction between tetrahydropapaverine and methyl or ethyl acrylate in an alcohol solvent under reflux conditions to form the acrylate ester intermediate.
- Conduct a transesterification reaction using the obtained ester intermediate and 1,5-pentanediol in toluene, catalyzed by potassium tert-butoxide, while removing the alcohol byproduct via azeotropic distillation.
- React the resulting diester with oxalic acid in acetone to precipitate and isolate the final dioxalate salt product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound benefits for supply chain stability and cost management. By eliminating the most challenging step of the traditional synthesis, manufacturers can significantly reduce the risk of batch failures and production delays. The reliance on commodity chemicals such as methyl acrylate, 1,5-pentanediol, and tetrahydropapaverine derivatives ensures a robust raw material supply base, reducing vulnerability to market fluctuations. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production schedules with greater confidence and accuracy.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for the expensive and difficult-to-synthesize diene intermediate, which traditionally accounted for a significant portion of the raw material costs. By shortening the synthetic route and utilizing common solvents like toluene and methanol, the overall consumption of energy and resources is drastically lowered. Additionally, the high yield and purity achieved reduce the need for extensive downstream purification steps, such as repeated recrystallizations or chromatography, leading to substantial cost savings in labor and materials.
- Enhanced Supply Chain Reliability: The use of stable, commercially available starting materials mitigates the risk of supply disruptions often associated with custom-synthesized specialty reagents. The mild reaction conditions (reflux temperatures) allow for the use of standard glass-lined or stainless steel reactors, meaning that production can be easily transferred between different manufacturing sites without requiring specialized equipment. This flexibility enhances the resilience of the supply chain, ensuring consistent availability of critical intermediates for global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with simple work-up procedures involving filtration and solvent evaporation that are easily adapted to large-scale industrial reactors. Furthermore, the avoidance of harsh reagents and the ability to recover and recycle solvents like toluene and acetone contribute to a greener manufacturing profile. This alignment with environmental compliance standards reduces the burden of waste treatment and supports sustainable manufacturing practices, a key consideration for modern supply chain heads.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of atracurium besilate intermediates using this patented method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply networks.
Q: What is the primary technical advantage of this new synthesis route?
A: The primary advantage is the avoidance of the synthetically difficult intermediate 4,10-dioxy-3,11-dioxo-1,12-tridecylene. By bypassing this complex step, the new method utilizes readily available starting materials and milder reaction conditions, significantly simplifying the overall process flow and reducing operational hazards.
Q: Which catalyst is employed in the transesterification step?
A: The process utilizes potassium tert-butoxide as the catalyst for the transesterification reaction. This strong base effectively facilitates the exchange between the acrylate ester intermediate and 1,5-pentanediol in a toluene solvent system, ensuring high conversion rates under reflux conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial suitability. It features simple operational steps, mild reaction temperatures (reflux in common solvents like methanol and toluene), and avoids harsh conditions, making it highly scalable for commercial manufacturing of neuromuscular blocking agent intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atracurium Besilate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates. Our team of expert chemists has extensively analyzed the methodology described in CN101845017A and possesses the technical capability to implement this advanced process immediately. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of atracurium besilate intermediate meets the highest international standards.
We invite you to collaborate with us to leverage this cost-effective and robust technology for your neuromuscular blocking agent projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing process is seamless and commercially advantageous.
