Advanced Synthesis of Hydroxy Pinacolone Retinoic Acid Ester for Commercial Scale-Up
The landscape of vitamin A derivative synthesis is undergoing a significant transformation driven by the urgent need for greener, safer, and more efficient manufacturing protocols, particularly for high-value cosmetic actives. Patent CN115260071A introduces a groundbreaking preparation method for hydroxy pinacolone retinoic acid ester (HPR), a next-generation retinoid known for its superior stability and reduced skin irritation compared to traditional tretinoin. This innovation addresses critical bottlenecks in the existing supply chain by replacing hazardous coupling reagents and complex enzymatic processes with a direct, robust nucleophilic substitution strategy. By utilizing tretinoin and halogenated pinacolone as primary starting materials in the presence of specific acid-binding agents, the process achieves exceptional molar yields exceeding 80 percent while maintaining liquid phase purity levels greater than 99.8 percent. For R&D directors and procurement specialists seeking a reliable functional active ingredients supplier, this technology represents a pivotal shift towards sustainable chemistry that minimizes environmental footprint without compromising on the stringent quality standards required for topical pharmaceutical and cosmetic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxy pinacolone retinoic acid ester has been plagued by severe technical and environmental drawbacks that hindered large-scale adoption. Traditional routes often relied on condensation reactions using dicyclohexylcarbodiimide (DCC) as a dehydrating agent, a practice that generates substantial quantities of dicyclohexylurea (DCU) solid waste, estimated at nearly one ton of waste for every ton of product produced. Furthermore, the removal of DCU and catalysts like DMAP is notoriously difficult, frequently necessitating extensive column chromatography or acidic washing steps that generate massive volumes of wastewater and increase production costs exponentially. Alternative methods employing phosphorus trichloride introduce significant safety hazards and environmental pollution due to the generation of corrosive byproducts, while enzymatic approaches, though selective, suffer from high enzyme costs, strict pH requirements, and poor adaptability to industrial reactor conditions. These legacy processes not only inflate the cost of goods sold but also create complex supply chain vulnerabilities related to waste disposal compliance and raw material availability.
The Novel Approach
In stark contrast, the methodology disclosed in CN115260071A circumvents these obstacles by employing a direct substitution mechanism that is both atom-economical and operationally simple. The core innovation lies in the complete substitution of halogenated pinacolone with tretinoin under the mediation of an acid-binding agent, effectively eliminating the risk of residual halogenated alkanes which are strictly forbidden in cosmetic formulations due to genotoxicity concerns. This approach allows for the use of common, recyclable solvents such as ethyl acetate, acetonitrile, or toluene, and produces only inorganic salts as byproducts, which are easily removed via simple filtration. The reaction conditions are remarkably mild, operating effectively between -10°C and 60°C without the need for energy-intensive cryogenic cooling or inert atmosphere complexities beyond standard nitrogen protection. This streamlined workflow not only enhances cost reduction in personal care manufacturing by removing purification bottlenecks but also ensures a consistent supply of high-purity hydroxy pinacolone retinoic acid ester suitable for sensitive skin applications.
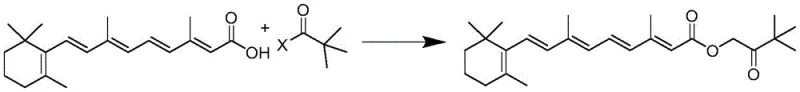
Mechanistic Insights into Nucleophilic Substitution Esterification
The chemical elegance of this synthesis lies in its straightforward nucleophilic substitution mechanism, where the carboxyl group of tretinoin acts as the nucleophile attacking the electrophilic carbon of the halogenated pinacolone. In the presence of a base such as triethylamine, sodium carbonate, or potassium bicarbonate, the carboxylic acid proton is scavenged, generating a carboxylate anion that is significantly more reactive towards the alkyl halide. This acid-binding step is crucial as it drives the equilibrium forward by neutralizing the hydrogen halide byproduct, preventing the degradation of the acid-sensitive polyene chain of the vitamin A moiety. The choice of solvent plays a pivotal role in stabilizing the transition state; polar aprotic solvents like acetonitrile or moderately polar esters like ethyl acetate facilitate the dissolution of both organic reactants while allowing the resulting inorganic salts to precipitate out of the solution, simplifying downstream processing. This mechanistic pathway avoids the formation of activated intermediates that could lead to cis-trans isomerization, thereby keeping cis-isomer impurities below 500ppm, a critical specification for maintaining biological efficacy.
From an impurity control perspective, the reaction design inherently mitigates the formation of genotoxic impurities that have historically plagued retinoid synthesis. By ensuring a molar excess of the acid-binding agent and carefully controlling the dropwise addition of the halogenated precursor, the process guarantees complete consumption of the halogenated starting material, leaving no detectable traces of chloro-pinacolone in the final matrix. The absence of heavy metal catalysts or coupling reagents means that the final product requires minimal purification, often needing only a recrystallization step to achieve pharmaceutical-grade specifications. This level of control is essential for meeting the rigorous regulatory standards of global markets, where residual solvents and toxic byproducts are scrutinized heavily. The robustness of this mechanism allows for real-time monitoring via liquid chromatography, enabling operators to adjust feed rates dynamically to ensure reaction completion before proceeding to workup, thus maximizing yield consistency across different batch sizes.
How to Synthesize Hydroxy Pinacolone Retinoic Acid Ester Efficiently
Implementing this synthesis route in a pilot or production facility requires adherence to specific operational parameters to maximize the benefits of the patented technology. The process begins with the dissolution of tretinoin in a selected solvent under dark conditions to prevent photo-degradation of the conjugated polyene system, followed by the addition of the acid-binding agent to prepare the reactive medium. The subsequent dropwise addition of monohalogen pinacolone must be controlled to manage the exotherm and ensure uniform mixing, with reaction progress tracked via HPLC to determine the precise endpoint. Once the substitution is complete, the workup involves a simple filtration to remove the precipitated inorganic salts, followed by vacuum concentration to isolate the crude product which can be further purified if necessary.
- Dissolve tretinoin in a suitable organic solvent such as ethyl acetate or acetonitrile under inert gas protection in a dark environment.
- Add an acid-binding agent like triethylamine or sodium carbonate to the solution to scavenge generated acids during the reaction.
- Dropwise add monohalogen pinacolone to the mixture, maintain temperature between -10°C and 60°C, and stir until reaction completion monitored by HPLC.
- Filter the reaction mixture to remove inorganic salts, then concentrate the filtrate under reduced pressure to crystallize the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers profound strategic advantages that extend beyond mere technical feasibility. The elimination of expensive coupling reagents like DCC and the avoidance of chromatographic purification steps result in a drastically simplified production workflow that lowers the overall cost of goods sold. The ability to recycle solvents such as ethyl acetate or toluene multiple times without significant loss of efficiency contributes to substantial cost savings and aligns with corporate sustainability goals by reducing chemical consumption. Furthermore, the generation of only inorganic salt waste simplifies effluent treatment protocols, reducing the burden on waste management infrastructure and lowering compliance costs associated with hazardous waste disposal. These factors combined create a more resilient supply chain capable of responding to market demand fluctuations without the bottlenecks associated with complex purification or scarce reagent availability.
- Cost Reduction in Manufacturing: The replacement of stoichiometric coupling agents with catalytic or stoichiometric acid-binding agents significantly reduces raw material costs, as bases like sodium carbonate are vastly cheaper than carbodiimides. Additionally, the removal of column chromatography from the purification train eliminates the need for large volumes of silica gel and eluents, which are major cost drivers in fine chemical production. The simplified workup procedure reduces labor hours and equipment occupancy time, allowing for higher throughput in existing facilities. This economic efficiency makes the production of high-purity hydroxy pinacolone retinoic acid ester commercially viable even in competitive markets where price sensitivity is high.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as tretinoin, halogenated pinacolone, and common organic solvents, the process mitigates the risk of supply disruptions associated with specialized reagents. The robustness of the reaction conditions, which tolerate a wide temperature range and do not require exotic catalysts, ensures that production can be maintained consistently across different manufacturing sites. This reliability is crucial for securing long-term contracts with major cosmetic and pharmaceutical clients who demand uninterrupted supply of critical active ingredients. The scalability of the process from gram to multi-ton scales ensures that commercial scale-up of complex vitamin derivatives can be achieved without re-engineering the core chemistry.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by low E-factors and the absence of persistent organic pollutants, facilitates easier regulatory approval in environmentally stringent regions. The ability to operate without phosphorus trichloride removes the need for specialized corrosion-resistant equipment and scrubbing systems for acidic gases, lowering capital expenditure for new production lines. Waste streams consisting primarily of inorganic salts are easier to treat and dispose of compared to the complex organic sludge generated by DCC methods. This environmental compatibility not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand value of the end products by associating them with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing portfolios. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does this method eliminate gene-toxic impurities compared to prior art?
A: Unlike conventional methods using chloro-pinacolone which leave toxic residues, this process ensures complete substitution via an acid-binding agent, generating only harmless inorganic salts and ensuring residual halogenated alkanes are below detection limits suitable for cosmetics.
Q: What are the waste management advantages of this synthesis route?
A: This protocol avoids the use of DCC coupling agents that generate massive amounts of DCU solid waste (approx. 900kg per ton of product in older methods). Instead, it produces only filterable inorganic salts, and the organic solvents can be recycled repeatedly, drastically reducing three-waste treatment costs.
Q: Is the process scalable for industrial production without cryogenic conditions?
A: Yes, the reaction operates efficiently within a broad temperature range of -10°C to 60°C, eliminating the need for energy-intensive deep cryogenic cooling or ice baths required by phosphorus trichloride methods, thereby facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxy Pinacolone Retinoic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN115260071A and are fully equipped to leverage this technology for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle light-sensitive and air-sensitive chemistries with precision, utilizing rigorous QC labs to verify that every batch meets stringent purity specifications, including the critical limits for tretinoin residue and cis-isomers. We are committed to delivering high-purity hydroxy pinacolone retinoic acid ester that empowers your formulations with superior stability and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through this advanced synthetic route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate the superior quality of our HPR produced via this green and efficient methodology. Together, we can drive the next generation of anti-aging solutions to market faster and more sustainably.
