Advanced Synthesis of Hydroxy Pinacolone Retinoic Acid Ester for Commercial Skincare Applications
Advanced Synthesis of Hydroxy Pinacolone Retinoic Acid Ester for Commercial Skincare Applications
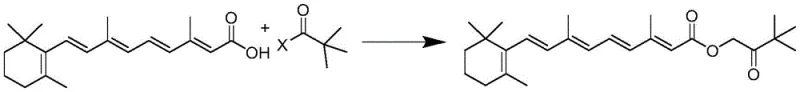
The pharmaceutical and cosmetic industries are constantly seeking more efficient and environmentally sustainable pathways for producing high-value active ingredients. Patent CN115260071A introduces a groundbreaking preparation method for hydroxy pinacolone retinoic acid ester (HPR), a next-generation vitamin A derivative renowned for its stability and low irritation profile. This innovation addresses critical bottlenecks in traditional retinoid synthesis by utilizing a direct nucleophilic substitution strategy between tretinoin and halogenated pinacolone. By bypassing complex condensation agents and harsh activation steps, this technology offers a robust solution for manufacturers aiming to secure a reliable cosmetic active ingredient supplier status. The process operates under mild conditions, typically ranging from -10°C to 60°C, and achieves exceptional purity levels exceeding 99.8%, making it highly attractive for large-scale commercial adoption in the competitive skincare market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vitamin A ester derivatives has been plagued by significant technical and environmental challenges that hinder cost-effective manufacturing. Traditional approaches, such as those disclosed in prior art document CN113149880A, rely heavily on dicyclohexylcarbodiimide (DCC) as a dehydrating agent and 4-dimethylaminopyridine (DMAP) as a catalyst. While effective for small-scale laboratory synthesis, these reagents generate substantial amounts of dicyclohexylurea (DCU) solid waste, with estimates suggesting nearly 900 kilograms of waste per ton of product. Furthermore, removing residual DCC and DMAP from the final product is notoriously difficult, often requiring extensive acid washing or chromatographic purification, which drastically increases production costs and wastewater generation. Other methods involving phosphorus trichloride introduce severe environmental pollution risks, while enzymatic routes suffer from high catalyst costs and strict environmental sensitivity, limiting their viability for industrial scale-up.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in patent CN115260071A employs a direct substitution mechanism that fundamentally simplifies the reaction workflow. By selecting halogenated pinacolone as the electrophile and tretinoin as the nucleophile in the presence of a simple acid-binding agent, the reaction proceeds to completion without the need for expensive coupling reagents. This method effectively overcomes the technical prejudice against using halogenated pinacolone due to fears of residual halogenated alkanes; the new protocol ensures complete substitution, leaving no forbidden halogenated alkane residues in the final cosmetic grade product. The result is a cleaner reaction profile where the only by-products are inorganic salts that can be easily removed via filtration. This shift not only streamlines the downstream processing but also aligns perfectly with modern green chemistry principles, offering a pathway for significant cost reduction in skincare intermediate manufacturing.
Mechanistic Insights into Nucleophilic Substitution Esterification
The core of this technological advancement lies in the precise control of the nucleophilic substitution reaction between the carboxyl group of tretinoin and the halogenated carbon of the pinacolone derivative. Under the protection of an inert gas like nitrogen and in a light-proof environment to prevent retinoid degradation, the tretinoin is first deprotonated by an acid-binding agent such as triethylamine or sodium carbonate. This generates a reactive carboxylate anion which then attacks the electrophilic carbon center of the monohalogen pinacolone. The reaction kinetics are carefully managed by controlling the dropwise addition of the halogenated species and maintaining temperatures between -10°C and 60°C, ensuring that the polyene chain of the retinoid remains intact while facilitating the displacement of the halide ion. This mechanistic precision is crucial for minimizing side reactions that could lead to the formation of cis-isomers or degradation products.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the stringent requirements for cosmetic safety. The process is designed to limit residual tretinoin to less than 50ppm and cis-isomers to less than 500ppm, parameters that are vital for minimizing skin irritation. The use of specific solvents like ethyl acetate, acetonitrile, or butyl acetate plays a dual role: they solubilize the reactants effectively and allow for easy recovery and reuse after the reaction is complete. By avoiding the formation of stubborn organic by-products like DCU, the purification process is reduced to simple filtration of inorganic salts followed by concentration and crystallization. This high level of control over the reaction environment and workup procedure ensures that the final high-purity hydroxy pinacolone retinoic acid ester meets the rigorous quality standards demanded by global regulatory bodies for topical applications.
How to Synthesize Hydroxy Pinacolone Retinoic Acid Ester Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize yield and purity while maintaining operational safety. The process begins with the dissolution of tretinoin in a selected organic solvent under inert atmosphere, followed by the addition of a stoichiometric amount of acid-binding agent to activate the carboxylic acid. The subsequent dropwise addition of monohalogen pinacolone must be monitored closely, with reaction progress tracked via liquid chromatography to ensure complete conversion before proceeding to workup.
- Dissolve tretinoin in a suitable organic solvent such as ethyl acetate or acetonitrile under inert gas protection in a dark environment.
- Add an acid-binding agent like triethylamine or sodium carbonate to the solution to facilitate the deprotonation of the carboxylic acid.
- Dropwise add monohalogen pinacolone to the mixture, maintain temperature between -10°C and 60°C, and stir until reaction completion monitored by HPLC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis method represents a strategic opportunity to optimize both cost structures and operational reliability. The elimination of expensive coupling agents like DCC and the removal of complex purification steps such as column chromatography directly translate to lower raw material costs and reduced processing time. Furthermore, the ability to recycle solvents repeatedly creates a closed-loop system that minimizes waste disposal fees and reduces the overall environmental footprint of the manufacturing facility. This efficiency gain is not merely theoretical; it is built into the fundamental chemistry of the process, which generates only benign inorganic salts as waste, thereby simplifying compliance with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the reaction workflow and the removal of high-cost reagents. By replacing expensive dehydrating agents and catalysts with inexpensive acid-binding agents like sodium carbonate or triethylamine, the direct material cost per kilogram of product is significantly lowered. Additionally, the avoidance of chromatographic purification steps reduces the consumption of silica gel and solvents, further driving down operational expenditures. The simplified workup, which involves only filtration and concentration, also lowers energy consumption and labor hours, contributing to a more lean and cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the use of readily available and stable raw materials such as tretinoin and halogenated pinacolone. Unlike enzymatic processes that require specialized storage conditions and have limited shelf lives, the reagents in this chemical synthesis are robust and easy to source from multiple suppliers. The mild reaction conditions, which do not require extreme cryogenic temperatures or high-pressure equipment, reduce the risk of batch failures due to equipment malfunction. This robustness ensures consistent production schedules and reduces lead time for high-purity vitamin A esters, allowing manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous by-products and the simplicity of the unit operations involved. The generation of only inorganic salt waste eliminates the need for complex hazardous waste treatment facilities, making it easier to obtain environmental permits and maintain compliance. The solvent recovery capability further enhances the sustainability profile of the operation, aligning with corporate social responsibility goals. This scalability ensures that the commercial scale-up of complex retinoid derivatives can be achieved without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of hydroxy pinacolone retinoic acid ester based on the patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield, purity, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this synthesis route for their own manufacturing lines.
Q: What are the primary impurities controlled in this HPR synthesis?
A: The process strictly controls residual tretinoin to less than 50ppm and cis-isomers to less than 500ppm, ensuring high safety profiles for cosmetic use.
Q: How does this method improve environmental compliance compared to DCC coupling?
A: Unlike traditional DCC methods that generate nearly 1:1 solid waste ratios (DCU), this substitution method produces only removable inorganic salts, drastically reducing waste treatment burdens.
Q: Is the solvent reusable in this manufacturing process?
A: Yes, the patent explicitly states that the organic solvents used, such as ethyl acetate or acetonitrile, can be recovered and reused after concentration, enhancing process sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxy Pinacolone Retinoic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis technology in the realm of anti-aging skincare formulations. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical limits for residual tretinoin and cis-isomers defined in the patent. We are committed to delivering high-quality intermediates that empower your R&D teams to develop safer and more effective cosmetic products.
We invite you to collaborate with us to leverage this innovative manufacturing route for your next product launch. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener process can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for premium skincare actives.
