Industrial Synthesis of 3-Cephem Compounds: A Breakthrough in Antibiotic Intermediate Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical antibiotic intermediates, and Patent CN1155603C presents a transformative approach to the preparation of 3-cephem compounds. This technology addresses long-standing inefficiencies in the production of key precursors for broad-spectrum antibiotics like Cefazolin and Ceftriaxone. By shifting away from moisture-sensitive silyl chemistry toward a robust phenol-mediated deprotection strategy, this method offers a compelling solution for manufacturers aiming to optimize their supply chains. The core innovation lies in the use of phenol derivatives to selectively remove the 4-position carboxy-protective group from 7-amino-3-halomethylcephem compounds, enabling a seamless transition to high-value 3-thiomethylcephem derivatives. This technical advancement is not merely a laboratory curiosity but a validated industrial process that promises to redefine cost structures and operational reliability for global reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-halomethylcephem compounds has been plagued by significant operational hurdles that inflate production costs and complicate quality control. The conventional route typically relies on 7-aminocephalosporanic acid (7-ACA) as the starting material, necessitating the protection of both amino and carboxyl groups before halogenation can occur. This multi-step protection-deprotection sequence demands the use of stoichiometric amounts of expensive silyl reagents, such as trimethylsilyl iodide, which are not only costly but also extremely sensitive to moisture. Consequently, the entire reaction system must be maintained under rigorous anhydrous conditions, requiring specialized equipment and increasing energy consumption. Furthermore, the direct reaction of thiols with 7-ACA to form 3-thiomethylcephem compounds often suffers from poor reactivity at the acetoxy group, forcing operators to employ high temperatures that degrade yield and generate complex impurity profiles difficult to separate.
The Novel Approach
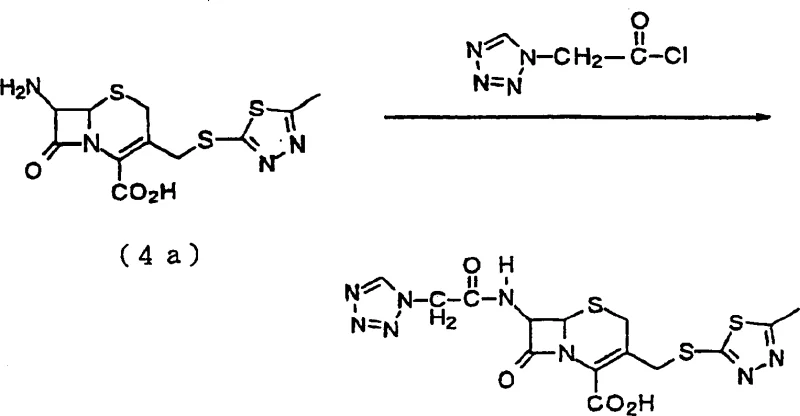
In stark contrast, the methodology disclosed in CN1155603C introduces a streamlined pathway that bypasses these bottlenecks by utilizing phenol derivatives for deprotection. This novel approach allows for the conversion of 7-amino-3-halomethylcephem compounds into reactive 3-halomethylcephem intermediates under mild conditions, typically between 15°C and 50°C. The elimination of strict anhydrous requirements and expensive silyl agents represents a paradigm shift in process chemistry. As illustrated in the reaction scheme below, the process flows logically from the protected starting material to the final thio-substituted product with remarkable efficiency. This structural clarity enables manufacturers to achieve cost reduction in pharmaceutical intermediate manufacturing by simplifying the unit operations and reducing the reliance on exotic reagents.
Mechanistic Insights into Phenol-Mediated Deprotection and Substitution
The chemical elegance of this process lies in the specific reactivity of the phenol derivative towards the 4-position carboxy-protective group. Unlike traditional acidic or basic hydrolysis which might compromise the delicate beta-lactam ring, the phenol-mediated mechanism proceeds with high chemoselectivity. The phenol acts as a nucleophile or a proton donor depending on the specific derivative used, facilitating the cleavage of the protecting group without inducing ring opening or epimerization at the chiral centers. This mechanistic precision is crucial for maintaining the stereochemical integrity of the cephem nucleus, which is vital for the biological activity of the final antibiotic. Following deprotection, the resulting 3-halomethylcephem intermediate possesses a highly reactive halogen atom at the C-3' position, which serves as an excellent leaving group for subsequent nucleophilic attack by thiol compounds.
Impurity control is inherently built into this mechanism due to the mild reaction conditions. By avoiding the high temperatures required for direct 7-ACA substitution, the formation of degradation products and polymeric by-products is significantly suppressed. The patent data indicates that the reaction can proceed smoothly at ambient temperatures, which kinetically favors the desired substitution over competing decomposition pathways. Furthermore, the final 3-thiomethylcephem compounds often precipitate directly from the reaction mixture upon pH adjustment, allowing for purification via simple crystallization rather than resource-intensive chromatography. This ease of isolation ensures that the high-purity pharmaceutical intermediates produced meet the stringent specifications required for downstream API synthesis, minimizing the risk of toxic impurities carrying over into the final drug product.
How to Synthesize 3-Thiomethylcephem Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these results in a pilot or production setting. The process begins with the preparation of the 3-halomethylcephem precursor, followed by the critical phenol deprotection step which sets the stage for high-yield thiolation. Operators should note that the choice of phenol derivative and solvent system can be tuned to optimize solubility and reaction rate, with water and polar aprotic solvents like DMF showing particular promise. The detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- React 7-amino-3-halomethylcephem compound (Formula 1) with a phenol derivative at 15-50°C to remove the 4-position carboxy-protective group, yielding the 3-halomethylcephem intermediate (Formula 2).
- Without isolation, react the resulting 3-halomethylcephem compound with a thiol compound or its salt (Formula 3) in a suitable solvent system such as water or DMF.
- Adjust pH to precipitate the final 3-thiomethylcephem compound (Formula 4), followed by filtration and drying to achieve high purity suitable for antibiotic synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic advantages beyond mere technical feasibility. The primary driver of value is the drastic simplification of the raw material portfolio. By eliminating the need for moisture-sensitive silyl reagents and the associated infrastructure for handling them, facilities can reduce their inventory complexity and lower the total cost of ownership for chemical storage. The ability to run reactions at ambient temperatures also decouples production from expensive cryogenic cooling systems, leading to substantial energy savings and increased throughput capacity. These factors combine to create a more resilient supply chain capable of responding quickly to market demand fluctuations for critical antibiotic ingredients.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive silyl protecting groups with commodity phenol derivatives cannot be overstated. This substitution removes a major cost center from the bill of materials while simultaneously reducing waste disposal costs associated with silicon-containing by-products. The high yields reported in the patent examples suggest that material utilization is maximized, further driving down the cost per kilogram of the active intermediate. Additionally, the simplified purification process reduces solvent consumption and processing time, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on readily available phenol derivatives and standard solvents mitigates the risk of supply disruptions often associated with specialty reagents. The robustness of the reaction conditions means that production is less susceptible to variations in environmental humidity or minor deviations in temperature control. This operational stability ensures consistent output quality and volume, allowing supply chain planners to forecast with greater confidence and maintain optimal inventory levels without the need for excessive safety stock.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on gram scales with straightforward workup procedures. The absence of hazardous silyl reagents simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. Waste streams are easier to treat, and the overall process aligns better with green chemistry principles, which is increasingly important for maintaining corporate sustainability goals and meeting the auditing standards of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 3-cephem synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: Why is the phenol-based deprotection method superior to traditional silyl reagent methods?
A: Traditional methods require expensive silyl reagents like trimethylsilyl iodide and strict anhydrous conditions. The phenol-based method operates at ambient temperatures, eliminates moisture sensitivity, and significantly reduces raw material costs while simplifying purification.
Q: What represents the key impurity control advantage in this process?
A: By avoiding harsh halogenation conditions and utilizing mild phenol deprotection, the formation of side products is drastically suppressed. The final product can often be purified simply by crystallization, ensuring high chemical purity essential for API manufacturing.
Q: Can this process be scaled for commercial production of Cefazolin intermediates?
A: Yes, the patent explicitly demonstrates industrial applicability with high yields (up to 99% in deprotection steps) and robust reaction conditions that do not require cryogenic cooling or specialized anhydrous equipment, making it ideal for large-scale commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cephem Compound Supplier
The technological potential of Patent CN1155603C represents a significant opportunity for optimizing the production of cephalosporin intermediates, and realizing this potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to support your development goals with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of intermediate we deliver meets the exacting standards required for global pharmaceutical markets. We understand the critical nature of antibiotic supply chains and are committed to providing consistent, high-quality materials that enable our partners to focus on their core drug development activities.
We invite you to engage with our technical team to explore how this novel synthesis route can be integrated into your existing manufacturing framework. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the specific economic benefits applicable to your operation. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project needs. Together, we can drive efficiency and innovation in the production of life-saving antibiotics.