Revolutionizing 2-Chloro-5-nitropyridine Production via Safe Photocatalytic Chlorination Technology
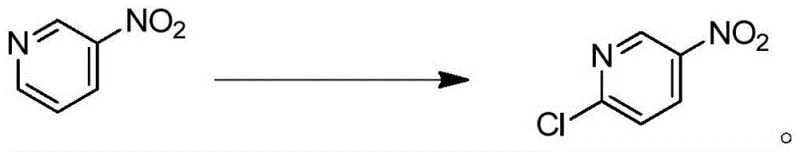
The pharmaceutical and agrochemical industries are constantly seeking safer, more efficient pathways for synthesizing critical heterocyclic intermediates, and patent CN116217469A presents a transformative solution for the production of 2-chloro-5-nitropyridine. This vital building block, essential for synthesizing androgen receptor antagonists, antimalarial drugs like pyronaridine, and various fungicides, has historically been produced through hazardous and environmentally taxing processes. The disclosed invention introduces a novel photocatalytic chlorination strategy that utilizes 3-nitropyridine as a starting material, reacting it with dilute hydrochloric acid in the presence of a chloride salt and a photocatalyst under UV irradiation. This approach not only eliminates the need for cryogenic conditions and explosive reagents but also achieves exceptional yields and purity profiles that meet the stringent demands of modern GMP manufacturing. By shifting from thermal or harsh chemical activation to photo-driven catalysis, this technology represents a significant leap forward in green chemistry, offering a robust platform for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-5-nitropyridine has been plagued by severe safety hazards and environmental burdens associated with traditional chlorinating agents. One common prior art method relies on dichlorine monoxide (Cl2O), a highly unstable oxidant that poses a catastrophic explosion risk when concentrations exceed 30%, necessitating storage at extremely low temperatures below -80°C and creating immense operational risks for large-scale production facilities. Another prevalent route involves the nitration of 2-aminopyridine followed by diazotization and chlorination with phosphorus oxychloride (POCl3), a multi-step process that generates substantial amounts of acidic, phosphorus-containing wastewater requiring complex and costly treatment protocols. Furthermore, alternative cyclization routes using expensive precursors like methyl chloroacrylate and triethyl orthoformate drive up raw material costs significantly, while still suffering from the pollution issues inherent to POCl3 usage. These conventional methodologies create bottlenecks in supply chain continuity due to safety shutdowns, regulatory scrutiny on waste disposal, and the high capital expenditure required for specialized corrosion-resistant and cryogenic equipment.
The Novel Approach
In stark contrast to these perilous legacy methods, the technology described in patent CN116217469A leverages visible or UV light to drive the chlorination reaction under remarkably mild and safe conditions. By employing 3-nitropyridine dissolved in dilute hydrochloric acid (10% solution) along with inexpensive chloride salts like ferric chloride or copper chloride, the process avoids the handling of explosive gases or corrosive chlorinating liquids entirely. The reaction proceeds efficiently at ambient temperatures ranging from 25°C to 30°C, eliminating the need for energy-intensive heating or deep-freeze cooling systems, which drastically simplifies the engineering requirements for reactor design. This photocatalytic protocol not only streamlines the operational workflow by reducing the number of unit operations but also inherently enhances workplace safety by removing the most dangerous steps from the manufacturing sequence. The result is a streamlined, cost-effective synthesis that maintains high throughput while adhering to increasingly strict global environmental and safety regulations.
Mechanistic Insights into Photocatalytic Chlorination
The core innovation of this process lies in the synergistic interaction between the photocatalyst, typically diphenylanthracene (DPA), and the chloride source under UV irradiation at a wavelength of approximately 360nm. Upon absorption of photon energy, the photocatalyst enters an excited state that facilitates the generation of reactive chlorine species from the chloride salts present in the acidic medium. These reactive intermediates then selectively attack the electron-deficient pyridine ring at the 2-position, driven by the electronic directing effects of the nitrogen atom and the nitro group at the 5-position. This regioselective functionalization is crucial for minimizing the formation of isomeric byproducts, such as 4-chloro or 6-chloro derivatives, which are difficult to separate and can compromise the quality of downstream API synthesis. The mild thermal conditions further suppress thermal degradation pathways and polymerization side reactions that often occur in high-temperature chlorinations, ensuring a cleaner reaction profile.
Furthermore, the choice of catalyst and solvent system plays a pivotal role in controlling the impurity profile of the final product. The use of dilute hydrochloric acid serves a dual purpose: it acts as the proton source to solubilize the pyridine substrate and provides the chloride ions necessary for the substitution reaction, thereby avoiding the introduction of extraneous counterions that could complicate purification. The photocatalytic cycle allows for the regeneration of the active catalytic species, meaning that only catalytic amounts of the metal salt and organic photocatalyst are required, which reduces the metal load in the crude product and simplifies the subsequent workup procedures. This mechanistic elegance translates directly into a superior impurity spectrum, with HPLC analysis confirming purity levels consistently above 99%, a critical metric for R&D directors evaluating the feasibility of this route for clinical trial material production.
How to Synthesize 2-Chloro-5-nitropyridine Efficiently
Implementing this photocatalytic synthesis requires precise control over reaction parameters to maximize yield and reproducibility, starting with the dissolution of 3-nitropyridine in 10% hydrochloric acid at a molar ratio of 2:1 to 3:1. The addition of the catalyst system, comprising ferric chloride or copper chloride alongside diphenylanthracene, must be followed by immediate exposure to UV light sources, preferably emitting at 360nm, while maintaining the reaction temperature between 25°C and 30°C using a circulating water bath. The reaction progress is monitored until completion, typically within 5 to 24 hours depending on the light intensity and scale, after which the mixture is neutralized with sodium carbonate to pH 8 to facilitate the extraction of the product into an organic solvent like ethyl acetate. For a detailed breakdown of the standardized operating procedures and specific equipment configurations required for this transformation, please refer to the technical guide below.
- Dissolve 3-nitropyridine in dilute hydrochloric acid (10% solution) with a molar ratio of acid to substrate between 2: 1 and 3:1.
- Add a chloride salt catalyst (FeCl3 or CuCl2) and a photocatalyst (Diphenylanthracene/DPA), then irradiate with UV light (360nm) at 25-30°C for 5-24 hours.
- Extract the reaction mixture with ethyl acetate, adjust pH to 8 with sodium carbonate, wash organic layers, and evaporate to dryness to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this photocatalytic methodology offers profound advantages by fundamentally altering the cost structure and risk profile of 2-chloro-5-nitropyridine manufacturing. The elimination of hazardous reagents like dichlorine monoxide and phosphorus oxychloride removes the need for specialized storage infrastructure and expensive waste treatment protocols, leading to substantial operational expenditure savings. Additionally, the use of commodity chemicals such as dilute hydrochloric acid and common metal salts ensures a stable and resilient supply chain, insulating production from the volatility often seen with specialty reagent markets. The simplicity of the workup procedure, involving standard extraction and evaporation techniques, further reduces labor costs and processing time, enabling faster turnaround times for custom synthesis orders.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive and dangerous chlorinating agents with inexpensive chloride salts and the removal of cryogenic cooling requirements. By operating at near-ambient temperatures, the process significantly lowers energy consumption associated with heating and cooling, while the high selectivity reduces the loss of valuable starting materials to byproducts. The avoidance of phosphorus-based reagents also eliminates the high costs associated with treating phosphorus-contaminated wastewater, resulting in a leaner and more profitable manufacturing model that passes savings down to the customer.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain reliability, and by removing explosive and highly toxic reagents from the process, the risk of unplanned plant shutdowns due to safety incidents is drastically minimized. The raw materials required, including 3-nitropyridine and iron or copper salts, are widely available bulk chemicals with robust global supply networks, ensuring consistent availability even during market disruptions. This stability allows for more accurate production planning and inventory management, guaranteeing on-time delivery for downstream pharmaceutical and agrochemical clients who depend on uninterrupted material flow.
- Scalability and Environmental Compliance: The scalability of this photocatalytic route is supported by its simple reaction engineering, which does not require complex pressure vessels or exotic materials of construction, making it easier to scale from pilot plant to commercial tonnage. The environmental footprint is significantly reduced due to the absence of heavy metal waste and acidic phosphorus effluents, aligning perfectly with modern green chemistry principles and facilitating easier regulatory approval in jurisdictions with strict environmental laws. This compliance advantage future-proofs the supply chain against tightening regulations, ensuring long-term viability for the production of this key intermediate.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this advanced synthesis method, we have compiled a set of frequently asked questions based on the specific data and claims found in the underlying patent literature. These answers provide clarity on the safety profile, efficiency metrics, and catalyst selection criteria that are critical for decision-makers evaluating this technology for adoption. Understanding these details is essential for assessing the feasibility of integrating this photocatalytic route into existing manufacturing portfolios or for sourcing high-quality intermediates produced via this superior method.
Q: What are the safety advantages of this photocatalytic method over traditional chlorination?
A: Unlike traditional methods using dichlorine monoxide (which is explosive above 30% concentration) or phosphorus oxychloride (which generates hazardous acidic wastewater), this photocatalytic method operates at mild temperatures (25-30°C) using stable chloride salts and dilute hydrochloric acid, significantly reducing explosion risks and environmental pollution.
Q: What yields and purity levels can be achieved with this novel synthesis route?
A: According to the experimental data in patent CN116217469A, this method consistently achieves high yields ranging from 93% to 96.5% and high purity levels exceeding 99% as measured by HPLC, demonstrating excellent efficiency for industrial scale-up.
Q: Which catalysts are suitable for this photocatalytic chlorination process?
A: The patent specifies the use of inexpensive transition metal chloride salts such as ferric chloride (FeCl3) or copper chloride (CuCl2) in combination with diphenylanthracene (DPA) as the photocatalyst, ensuring low raw material costs and easy availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-nitropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic routes like the photocatalytic chlorination described in CN116217469A to maintain a competitive edge in the global fine chemicals market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-chloro-5-nitropyridine delivered meets the highest standards required for pharmaceutical and agrochemical applications.
We invite potential partners to engage with our technical team to explore how this cost-effective and safe manufacturing technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener synthesis route. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and sustainability.
